Physicochemical Properties
| Molecular Formula | C15H14FN3O2 |
| Molecular Weight | 287.2944 |
| Exact Mass | 287.106 |
| Elemental Analysis | C, 62.71; H, 4.91; F, 6.61; N, 14.63; O, 11.14 |
| CAS # | 1550053-02-5 |
| PubChem CID | 72734382 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 449.9±45.0 °C at 760 mmHg |
| Flash Point | 225.9±28.7 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.689 |
| LogP | 1.81 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 21 |
| Complexity | 383 |
| Defined Atom Stereocenter Count | 0 |
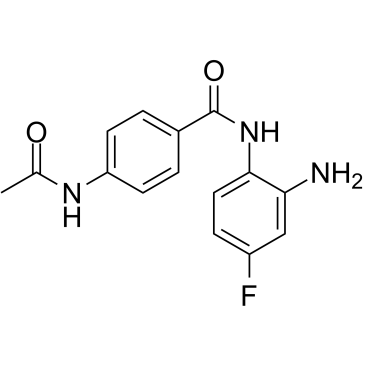
| SMILES | FC1C([H])=C([H])C(=C(C=1[H])N([H])[H])N([H])C(C1C([H])=C([H])C(=C([H])C=1[H])N([H])C(C([H])([H])[H])=O)=O |
| InChi Key | RRJDFENBXIEAPD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C15H14FN3O2/c1-9(20)18-12-5-2-10(3-6-12)15(21)19-14-7-4-11(16)8-13(14)17/h2-8H,17H2,1H3,(H,18,20)(H,19,21) |
| Chemical Name | 4-acetamido-N-(2-amino-4-fluorophenyl)benzamide |
| Synonyms | BRD-3308; BRD3308; BRD 3308 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | HIV-1; HDAC2 ( Ki = 6300 nM ); HDAC2 ( IC50 = 1340 nM ); HDAC1 ( Ki = 5100 nM ); HDAC1 ( IC50 = 1260 nM ); HDAC3 ( Ki = 29 nM ); HDAC3 ( Ki = 54 nM ) |
| ln Vitro | BRD3308 is a derivative of the ortho-aminoanilide HDAC inhibitor CI-994. Its IC50 value is 23 times lower for HDAC3 than it is for HDAC1 or 22, indicating that it is highly selective on HDAC3 inhibition. Exposure to BRD3308 facilitates the recovery of latent HIV-1 from patient cells and selective inhibition of HDAC3 induces HIV expression.[1] |
| ln Vivo | BRD3308 selectively inhibits HDAC3 to stop diabetes in female NOD mice from developing. In vivo administration of HDAC3 treatment inhibits pancreatic islet infiltration and shields β-cells from apoptosis. Animals treated with BRD3308 exhibit increased proliferation of β-cells. In NOD mice, HDAC3 treatment stops white adipose tissue infiltration in vivo.[2] |
| Cell Assay | Aliquots of resting CD4+ T cells are mixed with 15 μM of BRD3308, and plated in limiting dilution replicates of 2.5 or 1 million, 0.5 million, and 0.1 million cells. The cells are then incubated at 37°C with 5% CO2. The cultures of BRD3308 are withdrawn after a 24-hour period. |
| Animal Protocol |
Female NOD/ShiLTJ (NOD) mice 0.1 mg/kg, 1 mg/kg, 10 mg/kg IP |
| References |
[1]. PLoS One . 2014 Aug 19;9(8):e102684. [2]. J Biol Chem . 2017 Oct 27;292(43):17598-17608. |
Solubility Data
| Solubility (In Vitro) | DMSO : 57~250 mg/mL (198.4~870.2 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.24 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (7.24 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4808 mL | 17.4040 mL | 34.8080 mL | |
| 5 mM | 0.6962 mL | 3.4808 mL | 6.9616 mL | |
| 10 mM | 0.3481 mL | 1.7404 mL | 3.4808 mL |