Physicochemical Properties
| Molecular Formula | C23H14CLF3N4 |
| Molecular Weight | 438.832273960114 |
| Exact Mass | 438.085 |
| CAS # | 2018282-44-3 |
| PubChem CID | 129012190 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 6.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 31 |
| Complexity | 699 |
| Defined Atom Stereocenter Count | 0 |
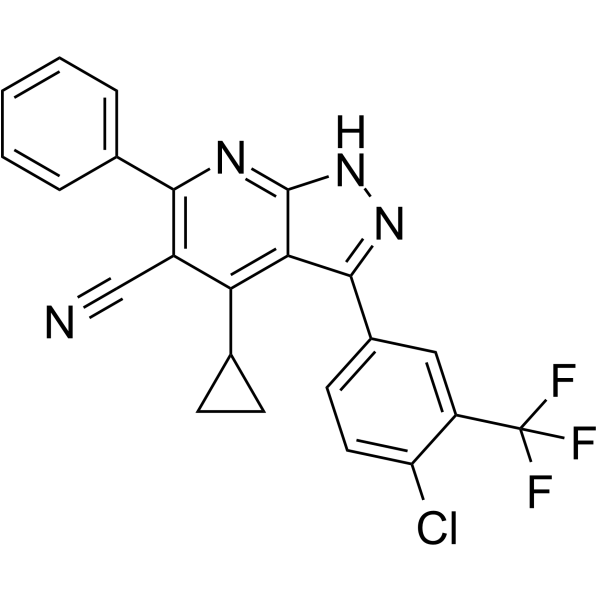
| SMILES | C12NN=C(C3=CC=C(Cl)C(C(F)(F)F)=C3)C1=C(C1CC1)C(C#N)=C(C1=CC=CC=C1)N=2 |
| InChi Key | UXLUNMKSQUMWLH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H14ClF3N4/c24-17-9-8-14(10-16(17)23(25,26)27)21-19-18(12-6-7-12)15(11-28)20(29-22(19)31-30-21)13-4-2-1-3-5-13/h1-5,8-10,12H,6-7H2,(H,29,30,31) |
| Chemical Name | 3-[4-chloro-3-(trifluoromethyl)phenyl]-4-cyclopropyl-6-phenyl-2H-pyrazolo[3,4-b]pyridine-5-carbonitrile |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Metabotropic Glutamate Receptor 5 (mGluR5) (EC₅₀ = 0.45 μM in calcium flux assay; EC₅₀ = 0.32 μM in IP1 accumulation assay) [1] |
| ln Vitro |
BMT-145027 is a molecule that exhibits a moderate reduction in planarity (Fsp3 = 0.17), adequate potency (EC50 = 47 nM), and strong MsLM stability (85% remaining). Crucially, at doses as high as 16 μM, BMT-145027 has no intrinsic mGluR5 agonist action [1]. BMT-145027 is a potent and selective positive allosteric modulator (PAM) of human metabotropic Glutamate Receptor 5 (mGluR5). [1] In calcium flux assays using HEK293 cells stably expressing human mGluR5, BMT-145027 dose-dependently enhanced glutamate (1 μM)-induced mGluR5 activation, with an EC₅₀ value of 0.45 μM and a maximum effect (Emax) of 226% relative to the glutamate-alone control. [1] In IP1 accumulation assays (a measure of Gq-mediated signaling) in the same cell line, BMT-145027 exhibited an EC₅₀ of 0.32 μM and an Emax of 218% when co-administered with 1 μM glutamate. [1] BMT-145027 showed high subtype selectivity: it did not exhibit significant modulatory activity (EC₅₀ > 10 μM) against other mGluR subtypes (mGluR1, mGluR2, mGluR3, mGluR4, mGluR6, mGluR7, mGluR8) in calcium flux or IP1 assays. [1] The compound acts through an allosteric binding site (distinct from the glutamate orthosteric site), as it did not compete with [³H]-quisqualate (a selective orthosteric ligand) for binding to mGluR5 in radioligand binding assays. [1] |
| ln Vivo | Mice given drugs and mice given no drugs were shown two identical things. Mice were concurrently presented with novel objects and then reintroduced to known objects after a 24-hour period of natural forgetting. The amount of time mice spend investigating novel objects can be used to gauge memory enhancements because they explore them more than humans do. At a dose of 30 mg/kg, BMT-145027 led to a considerable increase in the amount of time spent on novel objects, and at a dose of 10 mg/kg, there was a definite tendency toward novel object preference. In satellite animals, the total plasma concentration is 2800 nM at 30 mg/kg [1]. |
| Enzyme Assay |
Calcium flux assay for mGluR5 functional activity: HEK293 cells stably expressing human mGluR5 were seeded in 96-well plates and cultured to confluence. Cells were loaded with a cell-permeable calcium-sensitive fluorescent dye and incubated for a specified period. Serial dilutions of BMT-145027 (0.01 μM to 10 μM) were prepared and added to the cells, followed by stimulation with 1 μM glutamate (a submaximal concentration of the orthosteric agonist). Fluorescence intensity was measured in real time using a microplate reader to monitor intracellular calcium mobilization. The EC₅₀ value was calculated by plotting the percentage of maximum glutamate-induced response against the logarithm of compound concentration. [1] IP1 accumulation assay for Gq signaling activation: HEK293 cells stably expressing human mGluR5 were seeded in 96-well plates and cultured overnight. Cells were pre-incubated with serial dilutions of BMT-145027 for 30 minutes, then stimulated with 1 μM glutamate for 1 hour. After stimulation, cells were lysed, and intracellular IP1 levels (a downstream metabolite of IP3) were quantified using a homogeneous time-resolved fluorescence (HTRF)-based assay. The EC₅₀ value was determined by fitting the dose-response data to a four-parameter logistic model. [1] Radioligand binding assay for orthosteric site competition: Membrane preparations from HEK293 cells expressing human mGluR5 were incubated with a fixed concentration of [³H]-quisqualate (a radiolabeled orthosteric ligand) and serial dilutions of BMT-145027 at 4°C for 60 minutes. Bound and free radioligands were separated by filtration, and the radioactivity of the bound fraction was measured. The absence of displacement of [³H]-quisqualate confirmed that the compound binds to an allosteric site. [1] mGluR subtype selectivity assay: Using the same calcium flux or IP1 accumulation assay protocols described above, BMT-145027 (up to 10 μM) was tested against HEK293 cells stably expressing individual human mGluR subtypes (mGluR1, mGluR2-mGluR4, mGluR6-mGluR8) to evaluate subtype selectivity. [1] |
| References |
[1]. Development of 1H-Pyrazolo[3,4-b]pyridines as Metabotropic Glutamate Receptor 5 Positive Allosteric Modulators. ACS Med Chem Lett. 2016 Oct 3;7(12):1082-1086. |
| Additional Infomation |
BMT-145027 belongs to the 1H-pyrazolo[3,4-b]pyridine chemical class, developed through structure-activity relationship (SAR) optimization of lead compounds targeting mGluR5. [1] Metabotropic glutamate receptor 5 (mGluR5) is a G protein-coupled receptor (Gq-coupled) primarily expressed in the central nervous system (CNS), and its dysfunction is associated with neuropsychiatric and neurodegenerative disorders such as schizophrenia, Alzheimer's disease, and depression; mGluR5 positive allosteric modulators (PAMs) are potential therapeutic agents for these conditions. [1] The structural optimization of BMT-145027 focused on improving potency, selectivity, and drug-like properties, with the 1H-pyrazolo[3,4-b]pyridine scaffold identified as a promising core for mGluR5 PAM development due to its favorable binding interactions with the mGluR5 allosteric site. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~2.4 mg/mL (~5.47 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2788 mL | 11.3939 mL | 22.7879 mL | |
| 5 mM | 0.4558 mL | 2.2788 mL | 4.5576 mL | |
| 10 mM | 0.2279 mL | 1.1394 mL | 2.2788 mL |