Physicochemical Properties
| Molecular Formula | C32H30F2N4O4 | |
| Molecular Weight | 572.61 | |
| Exact Mass | 572.223 | |
| Elemental Analysis | C, 67.12; H, 5.28; F, 6.64; N, 9.78; O, 11.18 | |
| CAS # | 1643368-58-4 | |
| Related CAS # |
|
|
| PubChem CID | 86582336 | |
| Appearance | White to off-white solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 746.5±70.0 °C at 760 mmHg | |
| Flash Point | 405.3±35.7 °C | |
| Vapour Pressure | 0.0±2.6 mmHg at 25°C | |
| Index of Refraction | 1.667 | |
| LogP | 4.51 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 4 | |
| Heavy Atom Count | 42 | |
| Complexity | 1090 | |
| Defined Atom Stereocenter Count | 1 | |
| SMILES | FC1C=C(C(N)=O)C2=C(C=1C1C=CC=C(C=1C)N1C(N(C)C3C(=CC=CC=3C1=O)F)=O)C1=C(C[C@@H](C(C)(C)O)CC1)N2 |
|
| InChi Key | ZRYMMWAJAFUANM-INIZCTEOSA-N | |
| InChi Code | InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | |
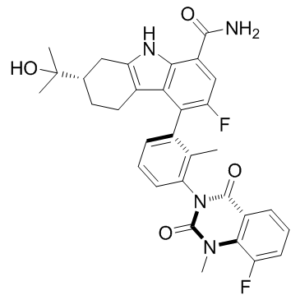
| Chemical Name | (7S)-3-fluoro-4-[3-(8-fluoro-1-methyl-2,4-dioxoquinazolin-3-yl)-2-methylphenyl]-7-(2-hydroxypropan-2-yl)-6,7,8,9-tetrahydro-5H-carbazole-1-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
BTK (IC50 = 0.5 nM); TEC (IC50 = 10 nM); ITK (IC50 = 15 nM); BLK (IC50 = 23 nM); TXK (IC50 = 28 nM); BMX (IC50 = 32 nM); LCK (IC50 = 71 nM); SRC (IC50 = 1100 nM) BMS-986142 potently inhibits human recombinant BTK with an IC50 of 0.5 nM in enzymatic assays. BMS-986142 exhibits high selectivity against a panel of 384 kinases; only five other kinases (Tec, ITK, BLK, Txk, and BMX) are inhibited with selectivity for BTK that is less than 100 times. Out of these kinases, only Tec (IC50=10 nM) is inhibited with a selectivity that is less than thirty times when compared to BTK. These four kinases are members of the Tec family of kinases. BMS-986142 does not prevent peripheral blood B cells from expressing CD86 or CD69 when stimulated by CD40L (IC50>10,000 nM for both). With an IC50 of 9 nM, BMS-986142 inhibits BTK-dependent calcium flux in response to anti-IgM treatment of Ramos B cells, activating BCR[2]. |
| ln Vitro |
BMS-986142 potently inhibits human recombinant BTK with an IC50 of 0.5 nM in enzymatic assays. BMS-986142 exhibits high selectivity against a panel of 384 kinases; only five other kinases (Tec, ITK, BLK, Txk, and BMX) are inhibited with selectivity for BTK that is less than 100 times. Out of these kinases, only Tec (IC50=10 nM) is inhibited with a selectivity that is less than thirty times when compared to BTK. These four kinases are members of the Tec family of kinases. BMS-986142 does not prevent peripheral blood B cells from expressing CD86 or CD69 when stimulated by CD40L (IC50>10,000 nM for both). With an IC50 of 9 nM, BMS-986142 inhibits BTK-dependent calcium flux in response to anti-IgM treatment of Ramos B cells, activating BCR[2]. BMS-986142 potently inhibited human recombinant BTK with an IC50 of 0.5 nM in enzymatic assays and showed high selectivity against a panel of 384 kinases, primarily inhibiting other Tec family kinases.[2] In primary human B cells stimulated through the B-cell receptor (BCR), BMS-986142 inhibited BCR-stimulated calcium flux in Ramos B cells (IC50 = 9 nM), proliferation of tonsillar B cells (IC50 = 3 nM), expression of CD86 on peripheral blood B cells (IC50 = 4 nM) and memory B cells (IC50 = 3 nM), and production of TNF-α (IC50 = 3 nM) and IL-6 (IC50 = 5 nM) from tonsillar B cells. It did not inhibit CD40L-induced CD86 expression (IC50 >10,000 nM).[2] In Ramos B cells, BMS-986142 inhibited anti-IgM-stimulated phosphorylation of phospholipase C-γ2 (IC50 ≈20 nM) and BTK autophosphorylation at Y551.[2] In human peripheral blood mononuclear cells (PBMCs), BMS-986142 inhibited immune complex (IC)-stimulated, FcγR-dependent production of TNF-α (IC50 = 3 nM) and IL-6 (IC50 = 4 nM), but not LPS (TLR4)-stimulated TNF-α production (IC50 >30,000 nM).[2] In human whole blood assays, BMS-986142 inhibited BCR-stimulated CD69 expression on B cells (IC50 = 90 nM) and FcεRI-stimulated CD63 expression on basophils (IC50 = 89 nM).[2] BMS-986142 dose-dependently inhibited RANK ligand (RANK-L)-induced osteoclastogenesis from human monocytic precursor cells, with inhibition evident at concentrations as low as 15 nM.[2] |
| ln Vivo |
BMS-986142 at 4, 10, and 30 mg/kg causes dose-dependent reductions in clinically evident disease of 26%, 43%, and 79%, respectively at the end of the study. It's interesting to note that when administered in addition to MTX, 4 mg/kg BMS-986142 offers an additive benefit in clinical scores (54% inhibition) as opposed to 19% inhibition with MTX alone. In comparison to 24% and 10%, respectively, with either medication alone, co-administration of BMS-986142 at 4 mg/kg with MTX results in a 53% reduction in inflammation and bone resorption. Moreover, 10 and 30 mg/kg BMS-986142 significantly inhibit serum anti-collagen II IgG titers. If BMS-986142 is not administered until day 21, the collagen booster, there are also dose-dependent reductions in clinical scores. At the conclusion of the trial, BMS-986142 doses of 2, 4, and 25 mg/kg produce clinical score reductions of 17%, 37%, and 67%, respectively[2]. In a primary anti-keyhole limpet hemocyanin (KLH) antibody response model in mice, prophylactic oral administration of BMS-986142 (3, 10, 30 mg/kg QD) dose-dependently inhibited anti-KLH IgM (day 7) and IgG (day 14) antibody titers, with significant reductions at 10 and 30 mg/kg.[2] In the collagen antibody-induced arthritis (CAIA) mouse model, prophylactic oral administration of BMS-986142 (5, 20 mg/kg QD) reduced clinical arthritis scores (72% and >90% inhibition, respectively) and histological scores for inflammation and bone resorption.[2] In the collagen-induced arthritis (CIA) mouse model, prophylactic oral administration of BMS-986142 (4, 10, 30 mg/kg QD) dose-dependently reduced clinical scores (26%, 43%, 79% inhibition), histological joint damage, serum anti-collagen II IgG titers, and disease-associated increases in splenic plasma cells.[2] In the CIA model, co-administration of a suboptimal dose of BMS-986142 (4 mg/kg QD) with methotrexate (MTX, 0.25 mg/kg QD) resulted in greater inhibition of clinical scores and histology than either agent alone.[2] Therapeutic oral administration of BMS-986142 (2, 4, 25 mg/kg QD) starting at disease onset in the CIA model dose-dependently reduced clinical scores (17%, 37%, 67% inhibition) and protected against bone loss.[2] In the therapeutic CIA model, co-administration of low-dose BMS-986142 (2 or 4 mg/kg QD) with etanercept (15 mg/kg BIW, IP) resulted in significantly greater efficacy than either treatment alone, improving clinical scores, bone mineral density, and bone surface area.[2] In the prophylactic CIA model, co-administration of BMS-986142 (10 mg/kg QD) with murine CTLA-4-Ig (0.05 or 0.2 mg/kg BIW, IP) provided additive benefits in reducing clinical scores and histological damage compared to monotherapy.[2] |
| Enzyme Assay |
Test compounds, fluoresceinated peptide (1.5 μM), ATP (20 μM), human recombinant BTK (1 nM), and assay buffer (20 mM HEPES at pH 7.4, 10 mM MgCl2, 0.015% Brij 35 surfactant, and 4 mM DTT in 1.6% DMSO) were added to V-bottom 384-well plates, with a final volume of 30 μL. 45 μL of 35 mM EDTA was added to each sample to stop the reaction after it had been incubated for 60 minutes at room temperature. The fluorescent substrate and phosphorylated product were separated electrophoretically to analyze the reaction mixture. By comparing the inhibition data to both controls without an inhibitor (for 0% inhibition) and controls with no enzyme (for 100% inhibition), the inhibition levels were computed. The concentration needed to inhibit 50% of BTK activity (IC50) was found by creating dose-response curves. In DMSO, compounds were dissolved at a concentration of 10 mM and assessed at 11 concentrations. For primary kinase assays, human recombinant BTK was incubated at room temperature with a fluorescent peptide substrate, ATP, and BMS-986142 in assay buffer. Product turnover was quantitated using a microfluidic mobility shift assay system. Similar assays were used for other kinases (Tec, ITK, BLK, Txk, BMX) with ATP concentrations set at the apparent Km for each kinase.[2] The interaction of BMS-986142 at a high concentration (1000 nM) with a broad panel of 384 protein and lipid kinases was examined using competition binding assays. Additionally, its interaction at 200 nM with a panel of 337 kinases was assessed using enzymatic assays where ³²P-ATP incorporation into peptide or protein substrates was measured.[2] |
| Cell Assay |
Ramos B cells phosphorylate phospholipase C (PLC)-γ2 in response to stimulation from the B-cell receptor (BCR): After pre-incubating Ramos B cells for one hour at 37°C in media containing 10% fetal bovine serum (FBS) with different concentrations of BMS-986142, the cells are stimulated for precisely two minutes at 37°C with AffiniPure F(ab')2 fragment goat anti-human immunoglobulin (Ig)M at 50 μg/mL. Ice-cold phosphate-buffered saline is then added to quench the cells. After the cells are lysed and pelleted, the levels of PLCγ2 are determined by immunoblotting with rabbit anti-human phosphoY759-PLCγ2, and the Odyssey Infrared Imaging System is used to analyze the results. To guarantee uniform loading, actin control is normalized. For stimulated expression of CD86 and CD69 on human peripheral blood B cells, the E-negative fraction of PBMCs was stimulated with anti-human IgG+IgM F(ab')2 fragments in the presence of BMS-986142 for 18 hours. Cells were stained with antibodies against CD20 and either CD86 or CD69, and expression was quantified by median fluorescence intensity (MFI) using flow cytometry after gating on CD20+ B cells. Similar setups were used for CD40 or TLR4 stimulation.[2] For whole blood assays of BCR-stimulated CD69 expression on B cells, human whole blood was incubated with BMS-986142 and stimulated with anti-human IgM F(ab')2 fragments and IL-4 for 18 hours. Cells were stained for CD20 and CD69, lysed, fixed, and analyzed by flow cytometry.[2] For BCR-stimulated IL-6 and TNF-α production from human tonsillar B cells, isolated cells were pre-incubated with BMS-986142, then stimulated with anti-human IgM F(ab')2 fragments and IL-4. Supernatants were collected after 4.5 hours, and cytokine levels were measured by enzyme immunoassay.[2] For BCR-stimulated human tonsillar B-cell proliferation, cells were cultured with BMS-986142 and stimulated with anti-human IgG+IgM F(ab')2 fragments for 72 hours, followed by [³H]-thymidine incorporation measurement using liquid scintillation counting.[2] For BCR-stimulated calcium flux in Ramos B cells, dye-loaded cells were plated, treated with BMS-986142, and stimulated with anti-human IgM. Calcium flux was measured using a fluorometric imaging plate reader.[2] For BCR-stimulated PLCγ2 phosphorylation in Ramos B cells, cells pre-incubated with BMS-986142 were stimulated with anti-human IgM F(ab')2 fragments for exactly 2 minutes, then lysed. Phospho-PLCγ2 levels were measured by immunoblot.[2] For BCR-stimulated BTK phosphorylation in Ramos B cells, cells treated similarly were stimulated, fixed, stained with an Alexa647-conjugated anti-phospho-BTK (Y551) antibody, and analyzed by flow cytometry.[2] For IC-stimulated TNF-α and IL-6 production in PBMCs, cells were treated with BMS-986142 and stimulated with pre-formed IgG immune complexes for 7 hours. Cytokine levels in supernatants were measured by ELISA.[2] For RANK-L-induced osteoclastogenesis, human monocytic precursor cells were cultured with macrophage colony-stimulating factor, RANK-L, and BMS-986142 for 9 days. Cells were then fixed, stained for tartrate-resistant acid phosphatase (TRAP), and the number of TRAP-positive multinucleated cells was counted.[2] |
| Animal Protocol |
Mice: In male DBA/1 mice, 200 μg of admixed bovine type II collagen is injected subcutaneously at the base of the tail. Similar stimulation is given to the mice 21 days later. BMS-986142 in EtOH: TPGS: PEG300 (5:5:90) is used to initiate PO QD dosing for preventative administration right away; however, for therapeutic administration, dosing is postponed until day 21 of the booster immunization. In mice used for BMS-986142 plus MTX preventative studies, the doses are as follows: vehicle; 4, 10, or 30 mg/kg of BMS-986142; 4 mg/kg of BMS-986142 plus MTX 0.25 mg/kg; or MTX at 0.25 mg/kg per day. Mice are given vehicle daily, BMS-986142 at 2, 4, or 25 mg/kg daily, BMS-986142 at 2 or 4 mg/kg daily plus etanercept at 15 mg/kg IP twice weekly (BIW), or etanercept at 15 mg/kg IP BIW for therapeutic studies involving BMS-986142 plus etanercept. Mice are given vehicle daily, BMS-986142 at 10 or 30 mg/kg daily, murine CTLA-4-Ig at 0.05 or 0.2 mg/kg IP BIW, or BMS-986142 at 10 mg/kg daily plus murine CTLA-4-Ig at 0.05 or 0.2 mg/kg IP BIW for the preventative studies involving BMS-986142 plus murine cytotoxic T lymphocyte-associated protein 4 immunoglobulin (CTLA-4-Ig). Dosing is done starting on day 0 and continuing for the full 36-day study [2]. For the primary anti-KLH antibody response study, female BALB/c mice were immunized intraperitoneally (IP) with KLH on day 0. Mice were dosed once daily (QD) by oral gavage (PO) with BMS-986142 (3, 10, 30 mg/kg) formulated in PEG 400:water (80:20 v/v). Blood was collected on days 7 and 14 for anti-KLH antibody titer analysis by ELISA.[2] For the collagen antibody-induced arthritis (CAIA) model, a mixture of anti-collagen II monoclonal antibodies was administered IP to female BALB/c mice. Oral QD dosing with BMS-986142 (3, 10, 30 mg/kg) formulated in ethanol:tocopherol PEG 1000 succinate:PEG300 (5:5:90) began immediately. Three days later, LPS was injected IP to boost arthritis. Mice were monitored daily for paw inflammation.[2] For the collagen-induced arthritis (CIA) model, male DBA/1 mice were immunized subcutaneously with bovine type II collagen emulsified in adjuvant on day 0 and boosted on day 21. For preventative studies, oral QD dosing with BMS-986142 formulated in ethanol:TPGS:PEG300 (5:5:90) began on day 0. For therapeutic studies, dosing began on day 21 (booster). Doses varied by experiment (e.g., 4, 10, 30 mg/kg for monotherapy; 2, 4, 25 mg/kg for combination with etanercept). Mice were monitored for paw inflammation several times per week after boosting. Blood was collected at the end for drug level and anti-collagen II antibody measurement. Spleens were processed for flow cytometry. Hind paws were analyzed by micro-CT and histology.[2] In combination studies within the CIA model, BMS-986142 was administered orally QD. Co-administered agents included: methotrexate (MTX, 0.25 mg/kg, daily PO), etanercept (15 mg/kg, twice weekly IP), or murine CTLA-4-Ig (0.05 or 0.2 mg/kg, twice weekly IP).[2] |
| ADME/Pharmacokinetics |
In the KLH study, plasma drug levels of BMS-986142 were measured on day 14. At doses of 3, 10, and 30 mg/kg PO QD, drug levels were maintained above the mouse whole blood BCR-stimulated CD69 IC50 (140 nM) for less than 11, 14, and 17 hours per day, respectively. Levels at 24 hours for the 3 mg/kg dose were below the limit of detection.[2] In the CAIA model, a dose of 5 mg/kg PO QD provided approximately 16 hours of daily coverage above the mouse whole blood BCR CD69 IC50, while 20 mg/kg provided near-complete (24-hour) coverage.[2] In the preventative CIA model, doses of 4, 10, and 30 mg/kg PO QD provided 9, 14, and 21 hours of daily coverage above the mouse whole blood BCR CD69 IC50, respectively.[2] In the therapeutic CIA model, doses of 2, 4, and 25 mg/kg PO QD provided less than 10, 11, and 22 hours of daily coverage above the mouse whole blood BCR CD69 IC50, respectively.[2] |
| References |
[1]. Discovery of 6-Fluoro-5-(R)-(3-(S)-(8-fluoro-1-methyl-2,4-dioxo-1,2-dihydroquinazolin-3(4H)-yl)-2-methylphenyl)-2-(S)-(2-hydroxypropan-2-yl)-2,3,4,9-tetrahydro-1H-carbazole-8-carboxamide (BMS-986142): A Reversible Inhibitor of Bruton' [2]. Bruton'styrosine kinase inhibitor BMS-986142 in experimental models of rheumatoid arthritis enhances efficacy of agents representing clinical standard-of-care. PLoS One. 2017; 12(7): e0181782. |
| Additional Infomation |
BMS-986142 is under investigation in clinical trial NCT02880670 (Pharmacokinetics and Metabolism Study of Radiolabeled BMS-986142 in Healthy Male Subjects). BMS-986142 is a novel, oral, reversible, small-molecule inhibitor of Bruton's tyrosine kinase (BTK). BTK is a key intracellular enzyme expressed in B cells and myeloid cells, regulating critical pathways in rheumatoid arthritis (RA) pathobiology, including B-cell receptor (BCR) signaling, Fcγ receptor (FcγR)-dependent cytokine production, and RANK ligand (RANK-L)-induced osteoclastogenesis.[2] The drug demonstrated efficacy in murine models of RA (CIA and CAIA) both as monotherapy and in combination with standard-of-care agents (methotrexate, etanercept, CTLA-4-Ig), suggesting its potential for clinical investigation in RA.[2] Efficacy in these models was observed without requiring continuous, complete inhibition of BTK, as drug levels providing only partial daily coverage of the target IC50 were effective.[2] The reversible nature of BMS-986142 may allow for precise titration and potentially mitigate risks associated with prolonged immunosuppression, especially in combination therapies.[2] The safety and efficacy of BMS-986142 were being evaluated in clinical trials for rheumatoid arthritis and primary Sjögren's syndrome at the time of publication.[2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.63 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (3.63 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (3.63 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7464 mL | 8.7319 mL | 17.4639 mL | |
| 5 mM | 0.3493 mL | 1.7464 mL | 3.4928 mL | |
| 10 mM | 0.1746 mL | 0.8732 mL | 1.7464 mL |