BMS-935177 is a potent, selective, reversible, and 2nd generation inhibitor of Bruton’s Tyrosine Kinase (BTK) with an IC50 of 3 nM. It outperforms other kinases, including those of the Tec family (TEC, BMX, ITK, and TXK, among others), in terms of kinase selectivity (5- and 67-fold selectivity) and activity against BTK. Clinical applications for BMS-935177 include the treatment of autoimmune diseases. BMS-935177 inhibited CD69 surface expression in peripheral B cells stimulated with anti-IgM and anti-IgG, and it also inhibited calcium flux in human Ramos B cells with an IC50 of 27 nM. BMS-935177 was chosen to proceed into clinical development due to its notable in vitro potency, strong in vivo activity, good pharmacokinetic profile, and tolerable safety profile.
Physicochemical Properties
| Molecular Formula | C31H26N4O3 | |
| Molecular Weight | 502.56 | |
| Exact Mass | 502.2 | |
| Elemental Analysis | C, 74.09; H, 5.21; N, 11.15; O, 9.55 | |
| CAS # | 1231889-53-4 | |
| Related CAS # |
|
|
| PubChem CID | 70647728 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 805.4±75.0 °C at 760 mmHg | |
| Flash Point | 440.9±37.1 °C | |
| Vapour Pressure | 0.0±3.0 mmHg at 25°C | |
| Index of Refraction | 1.707 | |
| LogP | 3.98 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 4 | |
| Heavy Atom Count | 38 | |
| Complexity | 951 | |
| Defined Atom Stereocenter Count | 0 | |
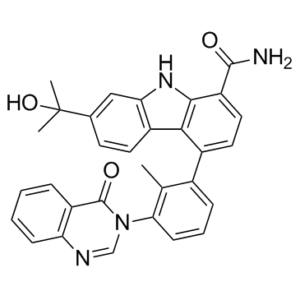
| SMILES | OC(C)(C)C1C=CC2=C(C=1)NC1C(C(N)=O)=CC=C(C3C=CC=C(C=3C)N3C=NC4C=CC=CC=4C3=O)C2=1 |
|
| InChi Key | TVJRDCQUZMGBAB-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C31H26N4O3/c1-17-19(8-6-10-26(17)35-16-33-24-9-5-4-7-22(24)30(35)37)20-13-14-23(29(32)36)28-27(20)21-12-11-18(31(2,3)38)15-25(21)34-28/h4-16,34,38H,1-3H3,(H2,32,36) | |
| Chemical Name | 7-(2-hydroxypropan-2-yl)-4-[2-methyl-3-(4-oxoquinazolin-3-yl)phenyl]-9H-carbazole-1-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | BTK (IC50 = 2.8 nM); TEC (IC50 = 13 nM); BLK (IC50 = 20 nM); BMX (IC50 = 24 nM); TrkA (IC50 = 30 nM) | |
| ln Vitro |
|
|
| ln Vivo |
|
|
| Enzyme Assay | BMS-935177 is tested at 11 different concentrations after being dissolved at 10 mM in DMSO. BMS-935177, 1 nM of human recombinant BTK, 1.5 μM of fluoresceinated peptide, 20 μM (Km app) of ATP, and 20 mM HEPES, pH 7.4, 10 mM MgCl2, 0.015% Brij 35 surfactant, and 4 mM DTT in 1.6% DMSO) are added to V-bottom 384-well plates, with a final volume of 30 μL of assay buffer. 45 μL of 35 mM EDTA is added to each sample to stop the reaction after it has been incubated for 60 minutes at room temperature. By electrophoretic separation of the fluorescent substrate and phosphorylated product (excitation, 488 nm; emission, 530 nm), the reaction mixture is examined. | |
| Animal Protocol |
Rats: In the PK studies, male Sprague-Dawley rats weighing 255-298 g are employed. Rats are given T99.5% 10 mM citrate buffer, pH 4, 0.02% DOSS, Methocel A4M, and BMS-935177 by oral gavage (1, 5, and 20 mg/kg) in order to examine the oral bioavailability of the drug following crystalline microsuspension doses. Following oral dosage, serial blood samples are taken at 0.25, 0.5, 0.75, 1, 2, 4, 6, 8, and 24 hours later. After centrifugation at 4 °C (1500-2000g), plasma samples are kept at -20 °C until analysis. Mice: BMS-935177 (10 and 30 mg/kg) is tested in a mouse anti-collagen antibody-induced arthritis (CAIA) inflammation model to compare its effectiveness with vehicle and dexamethasone (Dex). Four different combinations of monoclonal antimouse type II collagen antibodies—one milligram of each—are injected intraperitoneally (ip) into mice. The vehicle (EtOH:TPGS:PEG300, 5:5:90), BMS-935177 (10 or 30 mg/kg), or dexamethasone (dex, 1 mg/kg) is immediately started as a daily oral dose. The mice receive an intraperitoneal injection of 1.25 mg/kg lipopolysaccharide (LPS) after three days. The mice are then observed three times a week to observe the progression and intensity of paw inflammation. |
|
| References |
[1]. Small Molecule Reversible Inhibitors of Bruton's Tyrosine Kinase (BTK): Structure-Activity Relationships Leading to the Identification of 7-(2-Hydroxypropan-2-yl)-4-[2-methyl-3-(4-oxo-3,4-dihydroquinazolin-3-yl)phenyl]-9H-carbazole-1-c. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: 3.75 mg/mL (7.46 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 37.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 3.75 mg/mL (7.46 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 37.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9898 mL | 9.9491 mL | 19.8981 mL | |
| 5 mM | 0.3980 mL | 1.9898 mL | 3.9796 mL | |
| 10 mM | 0.1990 mL | 0.9949 mL | 1.9898 mL |