BMS-751324 is a novel prodrug of BMS-582949 (also known as PS540446) which is a potent and highly selective p38 mitogen-activated protein kinase (p38 MAPK) inhibitor with IC50 of 13nM and is currently in phase II clinical trials for the treatment of rheumatoid arthritis. BMS-751324 was discovered in search for prodrugs to address the issue of pH-dependent solubility and exposure associated with BMS-582949, a previously disclosed phase II clinical p38α MAP kinase inhibitor, a structurally novel clinical prodrug. BMS-751324 has a carbamoylmethylene linked promoiety containing hydroxyphenyl acetic acid (HPA) derived ester and phosphate functionalities. BMS-751324 was not only stable but also water-soluble under both acidic and neutral conditions. It was effectively bioconverted into parent drug BMS-582949 in vivo by alkaline phosphatase and esterase in a stepwise manner, providing higher exposure of BMS-582949 compared to its direct administration, especially within higher dose ranges. In a rat LPS-induced TNFα pharmacodynamic model and a rat adjuvant arthritis model, BMS-751324 demonstrated similar efficacy to BMS-582949. Most importantly, it was shown in clinical studies that BMS-751324 was indeed effective in addressing the pH-dependent absorption issue associated with BMS-582949.
Physicochemical Properties
| Molecular Formula | C32H35N6O10P |
| Molecular Weight | 694.628268480301 |
| Exact Mass | 694.215 |
| Elemental Analysis | C, 55.33; H, 5.08; N, 12.10; O, 23.03; P, 4.46 |
| CAS # | 948842-66-8 |
| PubChem CID | 44540113 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.5±0.1 g/cm3 |
| Index of Refraction | 1.676 |
| LogP | 1.22 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 15 |
| Heavy Atom Count | 49 |
| Complexity | 1230 |
| Defined Atom Stereocenter Count | 0 |
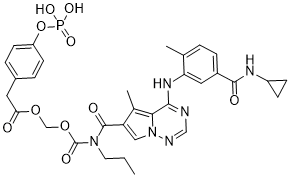
| SMILES | O=C(N(CCC)C(C1C(C)=C2N(N=CN=C2NC2C(C)=CC=C(C(NC3CC3)=O)C=2)C=1)=O)OCOC(CC1C=CC(OP(O)(O)=O)=CC=1)=O |
| InChi Key | XAYQDTPEOFCYIG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H35N6O10P/c1-4-13-37(32(42)47-18-46-27(39)14-21-6-11-24(12-7-21)48-49(43,44)45)31(41)25-16-38-28(20(25)3)29(33-17-34-38)36-26-15-22(8-5-19(26)2)30(40)35-23-9-10-23/h5-8,11-12,15-17,23H,4,9-10,13-14,18H2,1-3H3,(H,35,40)(H,33,34,36)(H2,43,44,45) |
| Chemical Name | [[4-[5-(cyclopropylcarbamoyl)-2-methylanilino]-5-methylpyrrolo[2,1-f][1,2,4]triazine-6-carbonyl]-propylcarbamoyl]oxymethyl 2-(4-phosphonooxyphenyl)acetate |
| Synonyms | BMS-751324; 948842-66-8; 976Z3162LI; UNII-976Z3162LI; Benzeneacetic acid, 4-(phosphonooxy)-, 1-((((((4-((5-((cyclopropylamino)carbonyl)-2-methylphenyl)amino)-5-methylpyrrolo(2,1-f)(1,2,4)triazin-6-yl)carbonyl)propylamino)carbonyl)oxy)methyl) ester; (((4-((5-(cyclopropylcarbamoyl)-2-methylphenyl)amino)-5-methylpyrrolo[2,1-f][1,2,4]triazine-6-carbonyl)(propyl)carbamoyl)oxy)methyl 2-(4-(phosphonooxy)phenyl)acetate; [[4-[5-(cyclopropylcarbamoyl)-2-methylanilino]-5-methylpyrrolo[2,1-f][1,2,4]triazine-6-carbonyl]-propylcarbamoyl]oxymethyl 2-(4-phosphonooxyphenyl)acetate; [[4-[5-(cyclopropylcarbamoyl)-2-methylanilino]-5-methylpyrrolo[2,1-f][1,2,4]triazine-6-carbonyl]-propylcarbamoyl]oxymethyl 2-(4-phosphonooxyphenyl)acetate;BMS-751324 (BMS751324);Bms751324; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | p38α MAPK |
| ln Vitro |
BMS-751324 is featured with a carbamoylmethylene linked promoiety containing hydroxyphenyl acetic acid (HPA) derived ester and phosphate functionalities. BMS-751324 was not only stable but also water-soluble under both acidic and neutral conditions. The alkaline phosphatase human placental ALP processes BMS-751324 (10 μM; 0-60 min), whereas the chemical enzyme factory does not [1]. |
| ln Vivo | The drug BMS-751324 (1 mg/kg, 3 mg/kg; elbow; twice daily for one week) is used as a topological adjuvant for arthritis-related foot swelling. It is biotransformed in animals to produce BMS-582949 (suspension: 1 mg/kg-100 mg/kg, monkey: 10 or 30 mg/kg, 5 mL/kg methylcellulose suspension; and inhibits lipopolysaccharide-induced TNFα production [1]. Oral ; single dose) is administered to patients. |
| Animal Protocol | In search for prodrugs to address the issue of pH-dependent solubility and exposure associated with 1 (BMS-582949), a previously disclosed phase II clinical p38α MAP kinase inhibitor, a structurally novel clinical prodrug, 2 (BMS-751324), featuring a carbamoylmethylene linked promoiety containing hydroxyphenyl acetic acid (HPA) derived ester and phosphate functionalities, was identified. Prodrug 2 was not only stable but also water-soluble under both acidic and neutral conditions. It was effectively bioconverted into parent drug 1 in vivo by alkaline phosphatase and esterase in a stepwise manner, providing higher exposure of 1 compared to its direct administration, especially within higher dose ranges. In a rat LPS-induced TNFα pharmacodynamic model and a rat adjuvant arthritis model, 2 demonstrated similar efficacy to 1. Most importantly, it was shown in clinical studies that prodrug 2 was indeed effective in addressing the pH-dependent absorption issue associated with 1.[2] |
| References |
[1]. Discovery of 4-(5-(cyclopropylcarbamoyl)-2-methylphenylamino)-5-methyl-N-propylpyrrolo [1, 2-f][1, 2, 4] triazine-6-carboxamide (BMS-582949), a clinical p38α MAP kinase inhibitor for the treatment of inflammatory diseases. Journal of Medicinal Chemistry, 2010, 53(18): 6629-6639. [2]. Discovery of ((4-(5-(Cyclopropylcarbamoyl)-2-methylphenylamino)-5-methylpyrrolo[1,2-f][1,2,4]triazine-6-carbonyl)(propyl)carbamoyloxy)methyl-2-(4-(phosphonooxy)phenyl)acetate (BMS-751324), a Clinical Prodrug of p38α MAP Kinase Inhibitor. J Med Chem . 2015 Oct 8;58(19):7775-84. |
| Additional Infomation |
The discovery and characterization of 7k (BMS-582949), a highly selective p38α MAP kinase inhibitor that is currently in phase II clinical trials for the treatment of rheumatoid arthritis, is described. A key to the discovery was the rational substitution of N-cyclopropyl for N-methoxy in 1a, a previously reported clinical candidate p38α inhibitor. Unlike alkyl and other cycloalkyls, the sp2 character of the cyclopropyl group can confer improved H-bonding characteristics to the directly substituted amide NH. Inhibitor 7k is slightly less active than 1a in the p38α enzymatic assay but displays a superior pharmacokinetic profile and, as such, was more effective in both the acute murine model of inflammation and pseudoestablished rat AA model. The binding mode of 7k with p38α was confirmed by X-ray crystallographic analysis [1]. Weakly basic compounds which have pH dependent solubility are liable to exhibit pH dependent absorption. In some cases, a subtle change in gastric pH can significantly modulate the plasma concentration of the drug and can lead to sub-therapeutic exposure of the drug. Evaluating the risk of pH dependent absorption and potential drug-drug interaction with pH modulators are important aspects of drug discovery and development. In order to assess the risk around the extent of decrease in the systemic exposure of drugs co-administered with pH modulators in the clinic, a pH effect study is carried out, typically in higher species, mostly dog. The major limitation of a higher species pH effect study is the resource and material requirement to assess this risk. Hence, these studies are mostly restricted to promising or advanced leads. In our current work, we have used in vitro aqueous solubility, in silico simulations using GastroPlus™ and an in vivo rat pH effect model to provide a qualitative assessment of the pH dependent absorption liability. Here, we evaluate ketoconazole and atazanavir with different pH dependent solubility profiles and based on in vitro, in silico and in vivo results, a different extent of gastric pH effect on absorption is predicted. The prediction is in alignment with higher species and human pH effect study results. This in vitro, in silico and in vivo (IVISIV) correlation is then extended to assess pH absorption mitigation strategy. The IVISIV predicts pH dependent absorption for BMS-582949 whereas its solubility enhancing prodrug, BMS-751324 is predicted to mitigate this liability. Overall, the material requirement for this assessment is substantially low which makes this approach more practical to screen multiple compounds during lead optimization. https://pubmed.ncbi.nlm.nih.gov/25960252/ |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4396 mL | 7.1981 mL | 14.3962 mL | |
| 5 mM | 0.2879 mL | 1.4396 mL | 2.8792 mL | |
| 10 mM | 0.1440 mL | 0.7198 mL | 1.4396 mL |