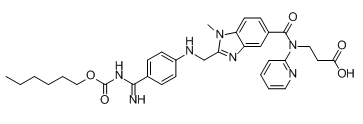
BIBR 1087 SE (BIBR-1087SE; Desethyl Dabigatran Etexilate) is a metabolite of dabigatran etexilate (BIBR-1048), which is the prodrug of dabigatran (an anticoagulant drug and blood thinner).
Physicochemical Properties
| Molecular Formula | C₃₂H₃₇N₇O₅ |
| Molecular Weight | 599.68 |
| Exact Mass | 599.285 |
| CAS # | 212321-78-3 |
| Related CAS # | Dabigatran etexilate;211915-06-9 |
| PubChem CID | 135565549 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 853.4±75.0 °C at 760 mmHg |
| Flash Point | 469.9±37.1 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.636 |
| LogP | 4.14 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 44 |
| Complexity | 959 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCCCCOC(=O)NC(=N)c1ccc(NCc2nc3cc(ccc3n2C)C(=O)N(CCC(O)=O)c2ccccn2)cc1 |
| InChi Key | UGEWTLXHMYKLCO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H37N7O5/c1-3-4-5-8-19-44-32(43)37-30(33)22-10-13-24(14-11-22)35-21-28-36-25-20-23(12-15-26(25)38(28)2)31(42)39(18-16-29(40)41)27-9-6-7-17-34-27/h6-7,9-15,17,20,35H,3-5,8,16,18-19,21H2,1-2H3,(H,40,41)(H2,33,37,43) |
| Chemical Name | 3-[[2-[[4-(N-hexoxycarbonylcarbamimidoyl)anilino]methyl]-1-methylbenzimidazole-5-carbonyl]-pyridin-2-ylamino]propanoic acid |
| Synonyms | Desethyl Dabigatran Etexilate; BIBR 1087 SE; 212321-78-3; Desethyl Dabigatran Etexilate; BIBR-1087SE; Desethyldabigatran Etexilate; BIBR 1087; BIBR-1087; 441X78E2Z0; (E)-3-(2-(((4-(N'-((Hexyloxy)carbonyl)carbamimidoyl)phenyl)amino)methyl)-1-methyl-N-(pyridin-2-yl)-1H-benzo[d]imidazole-5-carboxamido)propanoic acid; BIBR-1087 SE |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Dabigatran etexilate metabolite/intermediate |
| Additional Infomation | The impact of moderate hepatic impairment on the pharmacokinetics (PK) and pharmacodynamics (PD) of dabigatran etexilate was evaluated in an open, parallel-group study. Healthy volunteers (n = 12) and patients with hepatic impairment (Child-Pugh classification B; n = 12) received a single oral dose of 150 mg dabigatran etexilate. The mean values for area under the concentration-time curve, terminal half-life, and renal clearance of dabigatran were comparable between patients with hepatic impairment and healthy volunteers. Conversion of the dabigatran intermediate BIBR1087 to active dabigatran was slower in patients with hepatic impairment, indicating that the liver is partly involved in bioconversion of dabigatran etexilate. However, total drug exposure was comparable between groups; therefore, this observation is of no clinical relevance with respect to the anticoagulant activity of dabigatran. The extent of dabigatran glucuronidation was unchanged by liver disease; glucuronidation capacity was maintained in moderate liver disease. The activated partial thromboplastin time, ecarin clotting time, and thrombin time relationships were essentially identical in both groups. This study shows that moderate hepatic impairment does not affect the PK/PD or safety profile of dabigatran. Therefore, patients with moderate hepatic impairment can be given dabigatran etexilate without the need for dose adjustment. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~416.89 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.47 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (3.47 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6676 mL | 8.3378 mL | 16.6756 mL | |
| 5 mM | 0.3335 mL | 1.6676 mL | 3.3351 mL | |
| 10 mM | 0.1668 mL | 0.8338 mL | 1.6676 mL |