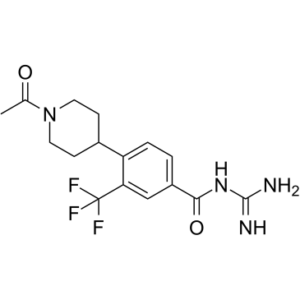
BI-9627 is a novel and highly potent NHE1 (sodium-hydrogen exchanger isoform 1) inhibitor with with an EC50 of 31 nM. It has good drug like properties such as high oral bioavailability, low DDI (drug drug interaction) potential, excellent pharmacokinetics, and good selectivity against NHE2 and NHE3. Sodium-hydrogen exchanger isoform 1 (NHE1) is a ubiquitously expressed transmembrane ion channel responsible for intracellular pH regulation. During myocardial ischemia, low pH activates NHE1 and causes increased intracellular calcium levels and aberrant cellular processes, leading to myocardial stunning, arrhythmias, and ultimately cell damage and death. The role of NHE1 in cardiac injury has prompted interest in the development of NHE1 inhibitors for the treatment of heart failure.
Physicochemical Properties
| Molecular Formula | C16H19F3N4O2 |
| Molecular Weight | 356.342873811722 |
| Exact Mass | 356.146 |
| CAS # | 1204329-34-9 |
| Related CAS # | BI-9627 hydrochloride;1422252-46-7 |
| PubChem CID | 51031001 |
| Appearance | White to yellow solid powder |
| LogP | 1.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 25 |
| Complexity | 538 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | C(NC(N)=N)(=O)C1=CC=C(C2CCN(C(C)=O)CC2)C(C(F)(F)F)=C1 |
| InChi Key | QMHRLXNEGYTSRV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H19F3N4O2/c1-9(24)23-6-4-10(5-7-23)12-3-2-11(14(25)22-15(20)21)8-13(12)16(17,18)19/h2-3,8,10H,4-7H2,1H3,(H4,20,21,22,25) |
| Chemical Name | 4-(1-acetylpiperidin-4-yl)-N-(diaminomethylidene)-3-(trifluoromethyl)benzamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Sodium-hydrogen exchanger isoform 1 (NHE1) inhibitor (IC50 = 45 nM in pH change assay; EC50 = 241 nM in human platelet swelling assay) with >30-fold selectivity against NHE2 and no measurable inhibition of NHE3 up to 16 μM [1] |
| ln Vitro |
In the Langendorff isolated perfused rat heart model, BI-9627 significantly prevented ischemia-reperfusion injury, recovering left ventricular developed pressure (LVDP) to 100% and 36% of baseline at perfusate concentrations of 100 nM and 10 nM, respectively, measured 30 minutes post-reperfusion. It also significantly attenuated the increase in left ventricular end-diastolic pressure (LVEDP) compared to vehicle control [1] |
| Enzyme Assay |
NHE1 inhibition was assessed using a pH change assay in PS120 cells stably expressing human NHE1. Cells were loaded with the pH-sensitive dye BCECF-AM, acid-loaded with an NH₄Cl-containing buffer, and then exposed to compound-containing buffer lacking NH₄Cl. The recovery of intracellular pH was monitored fluorometrically, and IC₅₀ values were calculated as the concentration causing 50% inhibition of pH recovery [1] |
| Cell Assay |
Functional inhibition of NHE1 was evaluated using a human platelet swelling assay (hPSA). Platelet-rich plasma was incubated with compound and then exposed to a propionate medium to induce osmotic swelling. Swelling was measured as a decrease in optical density at 680 nm over time, and EC₅₀ values were derived from the inhibition of swelling [1] |
| Animal Protocol |
Pharmacokinetic studies in rats: BI-9627 was dissolved in 20% hydroxypropyl-β-cyclodextrin for intravenous dosing (1 mg/kg) and suspended in 0.015% Tween 80 + 0.5% aqueous methylcellulose for oral dosing (5 mg/kg) to fasted male Sprague-Dawley rats. Serial blood samples were collected up to 24 hours for plasma analysis by LC/MS/MS [1] Pharmacokinetic studies in dogs: Similar formulations were used in male Beagle dogs following 1 mg/kg iv or 5 mg/kg po dosing, with blood sampling up to 72 hours [1] |
| ADME/Pharmacokinetics |
In rats, BI-9627 showed low clearance (5.7% of hepatic blood flow), a volume of distribution of 0.76 L/kg, mean residence time of 3.2 hours, and oral bioavailability of 73% [1] In dogs, clearance was 36.2% of hepatic blood flow, volume of distribution was 1.4 L/kg, mean residence time was 2.1 hours, and oral bioavailability was 33% [1] It exhibited low CYP inhibition (IC₅₀ > 30 µM for 2C19, 2C9, 2D6, and 3A4), high metabolic stability in human and rat liver microsomes (<11% and 11% QH, respectively), and high membrane permeability in PAMPA assay [1] |
| Toxicity/Toxicokinetics |
BI-9627 showed no mutagenicity in the Ames test up to 5000 µg/plate with or without S9 activation, no cytotoxicity up to 100 µM in HeLa cells, and no phospholipidosis potential up to 50 µM in the Nile Red assay [1] It did not inhibit hERG channels (IC₅₀ > 30 µM in PatchXpress assay) and showed no time-dependent CYP3A4 inactivation up to 50 µM [1] |
| References |
[1]. Identification of a potent sodium hydrogen exchanger isoform 1 (NHE1) inhibitor with a suitableprofile for chronic dosing and demonstrated cardioprotective effects in a preclinical model of myocardial infarction in the rat. J Med Chem. 2012 Aug 23;55(16):7114-40. |
| Additional Infomation |
BI-9627 is an optimized NHE1 inhibitor derived from sabiporide, designed for once-daily oral dosing with low drug-drug interaction potential and improved pharmacokinetic profile for chronic heart failure treatment [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~280.63 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.84 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.84 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (5.84 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8063 mL | 14.0315 mL | 28.0631 mL | |
| 5 mM | 0.5613 mL | 2.8063 mL | 5.6126 mL | |
| 10 mM | 0.2806 mL | 1.4032 mL | 2.8063 mL |