Description: BI-671800 is a novel, potent and highly specific antagonist of chemoattractant receptor-homologous molecule on Th2 cells (DP2/CRTH2), with IC50 values of 4.5 nM and 3.7 nM for PGD2 binding to CRTH2 in hCRTH2 and mCRTH2 transfected cells, respectively. BI-671800 can be potentially used for the treatment of poorly controlled asthma.
Physicochemical Properties
| Molecular Formula | C25H26F3N5O3 |
| Molecular Weight | 501.500855922699 |
| Exact Mass | 501.199 |
| Elemental Analysis | C, 59.87; H, 5.23; F, 11.36; N, 13.96; O, 9.57 |
| CAS # | 1093108-50-9 |
| PubChem CID | 45270144 |
| Appearance | White to off-white solid powder |
| LogP | 4.481 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 36 |
| Complexity | 717 |
| Defined Atom Stereocenter Count | 0 |
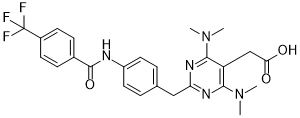
| SMILES | FC(C1C=CC(=CC=1)C(NC1C=CC(=CC=1)CC1=NC(=C(CC(=O)O)C(=N1)N(C)C)N(C)C)=O)(F)F |
| InChi Key | XEOSTBFUCNZKGS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C25H26F3N5O3/c1-32(2)22-19(14-21(34)35)23(33(3)4)31-20(30-22)13-15-5-11-18(12-6-15)29-24(36)16-7-9-17(10-8-16)25(26,27)28/h5-12H,13-14H2,1-4H3,(H,29,36)(H,34,35) |
| Chemical Name | 5-Pyrimidineacetic acid, 4,6-bis(dimethylamino)-2-((4-((4-(trifluoromethyl)benzoyl)amino)phenyl)methyl)- |
| Synonyms | BI-671800; BI 671800; BI671800. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Compound A (Cmpd A) is a potent and selective antagonist of the prostaglandin D₂ receptor–chemoattractant receptor-homologous molecule expressed on Tₕ₂ cells (CRTH2). The IC₅₀ values for inhibiting PGD₂ binding to CRTH2 are 3.7 nM for murine CRTH2 and 4.5 nM for human CRTH2. The IC₅₀ values for other prostanoid, thromboxane, or cysteinyl leukotriene receptors (DP1, BLT1, CysLT1, CysLT2, EP1, EP2, EP3, EP4, FP, IP, TP) are all >10 μM. Additionally, Cmpd A showed no activity on a broad panel of G protein-coupled receptors in a screening assay. [1] |
| ln Vitro |
In transfected cells, BI-671800 (Compound A) has modest nM efficacy as a human or mouse CRTH2 antagonist [1]. Cmpd A down-regulated the transcription of a wide range of pro-inflammatory genes in FITC-challenged mouse ears, including cytokines (IL-1β, IL-4, IL-13), chemokines (CCL7/MCP-3, CCL4/MIP-1β, CXCL9/MIG), and atopy-associated genes (mucin-1, trefoil factors 1–3). [1] |
| ln Vivo |
BI-671800 (compound A, 0.1-10 mg/kg, ig) has a strong inhibitory effect on mice AHR [1]. BI-671800 (Compound A) substantially inhibits edema formation and greatly lowers inflammatory infiltrates and skin pathology observed in drug vehicle-treated mice [3]. In a FITC-induced allergic contact hypersensitivity model in BALB/c mice, oral administration of Cmpd A (0.1, 1, or 10 mg/kg) significantly inhibited ear swelling in a dose-dependent manner, with peak inhibition observed at 10 mg/kg. Histological analysis showed reduced leukocyte infiltration (lymphocytes, eosinophils, neutrophils) and absence of epidermal microabscesses in treated mice. Cmpd A also reduced protein levels of pro-inflammatory cytokines (IL-4, TNF-α, TGF-β) and chemokines (MIP-2, GRO-α) in challenged ears. [1] |
| Enzyme Assay |
A radioligand binding assay was performed to characterize Compound A. CRTH2-transfected cells were suspended in binding buffer. Cell suspensions were incubated with tritiated PGD₂ and varying concentrations of the test compound for 1 hour at room temperature. After incubation, the mixtures were transferred to filtration plates, washed, and the bound radioactivity was measured using a scintillation counter. Competition binding curves were analyzed to determine IC₅₀ values. [3] A separate panel screening for binding affinity against a broad set of prostanoid, thromboxane, and cysteinyl leukotriene receptors was conducted using standard protocols. [3] |
| Cell Assay |
Protein lysates from FITC-challenged mouse ears were analyzed by ELISA for cytokine and chemokine levels. Ears were homogenized in protein isolation buffer with protease and phosphatase inhibitors, centrifuged to remove debris, and supernatants were quantified for total protein. Cytokine/chemokine levels were measured using commercial ELISA kits according to the manufacturer’s protocols. [1] |
| Animal Protocol |
Animal/Disease Models: 6-8 weeks old, age- and sex-matched balb/c (Bagg ALBino) mouse (mouse sensitized for 14 days, intranasally challenged) [1]. Doses: 10-0.1 mg/kg Route of Administration: po (oral gavage) for 4 weeks. Experimental Results: It has a significant inhibitory effect on AHR in mice. A 1-week acute FITC-induced allergic inflammation model was used. Female BALB/c mice were shaved on the belly and sensitized with 0.5% FITC solution on days 1 and 2. On day 6, mice were challenged with 0.5% FITC on the right ear and vehicle on the left ear. Cmpd A was administered orally 1 hour before challenge and again 7 hours post-challenge. Ear thickness was measured at various time points using digital calipers. For cytokine analysis, ears were harvested, snap-frozen, homogenized, and protein lysates were prepared for ELISA. [1] |
| References |
[1]. A small molecule CRTH2 antagonist inhibits FITC-induced allergic cutaneous inflammation. Int Immunol. 2009 Jan;21(1):81-93. [2]. A randomized study of BI 671800, a CRTH2 antagonist, as add-on therapy in poorly controlled asthma. Allergy Asthma Proc. 2017 Mar 1;38(2):157-164. [3]. CRTH2 antagonism significantly ameliorates airway hyperreactivity and downregulates inflammation-induced genes in a mouse model of airway inflammation. Am J Physiol Lung Cell Mol Physiol. 2008 Nov;295(5):L767-79. |
| Additional Infomation |
Bi 671800 has been used in trials studying the treatment of Asthma and Rhinitis, Allergic, Perennial. Cmpd A is a proprietary small-molecule CRTH2 antagonist developed by Actimis Pharmaceuticals. In the FITC-induced contact hypersensitivity model, CRTH2 antagonism inhibited multiple inflammatory pathways, including TSLP and IL-1β production, and reduced recruitment of neutrophils and eosinophils. The study suggests that CRTH2 antagonism may be a potential therapeutic strategy for allergic skin diseases such as atopic dermatitis. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~135 mg/mL (~269.19 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.25 mg/mL (4.49 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 22.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.25 mg/mL (4.49 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 22.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9940 mL | 9.9701 mL | 19.9402 mL | |
| 5 mM | 0.3988 mL | 1.9940 mL | 3.9880 mL | |
| 10 mM | 0.1994 mL | 0.9970 mL | 1.9940 mL |