Physicochemical Properties
| Molecular Formula | C15H14BRNO3S2 |
| Molecular Weight | 400.3106 |
| Exact Mass | 398.96 |
| Elemental Analysis | C, 45.01; H, 3.53; Br, 19.96; N, 3.50; O, 11.99; S, 16.02 |
| CAS # | 300817-68-9 |
| Related CAS # | 300817-68-9 |
| PubChem CID | 5720188 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 3.697 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 22 |
| Complexity | 513 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | BrC1C([H])=C([H])C(=C([H])C=1[H])C([H])=C1C(N(C(=S)S1)C([H])(C(=O)O[H])C([H])(C([H])([H])[H])C([H])([H])[H])=O |
| InChi Key | COHIEJLWRGREHV-YRNVUSSQSA-N |
| InChi Code | InChI=1S/C15H14BrNO3S2/c1-8(2)12(14(19)20)17-13(18)11(22-15(17)21)7-9-3-5-10(16)6-4-9/h3-8,12H,1-2H3,(H,19,20)/b11-7+ |
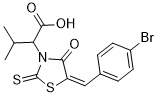
| Chemical Name | 2-[(5E)-5-[(4-bromophenyl)methylidene]-4-oxo-2-sulfanylidene-1,3-thiazolidin-3-yl]-3-methylbutanoic acid |
| Synonyms | BH3I1; BH 3I1; BH-3I1; BH3I-1; BH 3I-1; BH-3I-1 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | RIP1 (Kd = 5.3 μM); Bcl-2; Bcl-xL; Bim; Bak |
| ln Vitro | In vitro, BH3I-1 prevents Bcl-xL heterodimerization. It also causes the release of cytochrome c[1]. While inhibiting its reported targets Bcl-2/Bim and Bcl-xL/Bim, BH3I-1 also significantly inhibits the interactions between p53/hDM2 and p300/Hif-1a[2]. |
| Cell Assay | Cells (5×104 cells per well) are seeded into white 96-well plates (Costar) and treated with various concentrations of the compounds for 48 h. Cells are preincubated with 100 µM zVAD-FMK for 1 hour prior to the addition of chemicals for zVAD-FMK protection experiments. An MTS assay is used to gauge cell viability. Cells are expanded in 24-well plates for PI staining experiments before being incubated with 2 g ml/L PI. FACS analysis is used to identify cell death. |
| References |
[1]. Nat Cell Biol . 2001 Feb;3(2):173-82. [2]. Chem Commun (Camb) . 2010 Nov 14;46(42):8020-2. |
| Additional Infomation | 2-[5-[(4-bromophenyl)methylidene]-4-oxo-2-sulfanylidene-3-thiazolidinyl]-3-methylbutanoic acid is a valine derivative. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 64~100 mg/mL (159.9~249.8 mM) Ethanol: ~13 mg/mL (~32.5 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.25 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.25 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4981 mL | 12.4903 mL | 24.9806 mL | |
| 5 mM | 0.4996 mL | 2.4981 mL | 4.9961 mL | |
| 10 mM | 0.2498 mL | 1.2490 mL | 2.4981 mL |