Physicochemical Properties
| Molecular Formula | C24H22CL2N6O2 |
| Molecular Weight | 497.3765 |
| Exact Mass | 496.12 |
| Elemental Analysis | C, 57.96; H, 4.46; Cl, 14.25; N, 16.90; O, 6.43 |
| CAS # | 2290660-61-4 |
| PubChem CID | 137333984 |
| Appearance | Solid powder |
| LogP | 2.1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 34 |
| Complexity | 785 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | BMOUOZDRMBLNSI-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H22Cl2N6O2/c25-20-2-1-15(11-21(20)26)13-31-14-19(23(33)30-16-3-7-27-8-4-16)22-18(24(31)34)12-29-32(22)17-5-9-28-10-6-17/h1-4,7-8,11-12,14,17,28H,5-6,9-10,13H2,(H,27,30,33) |
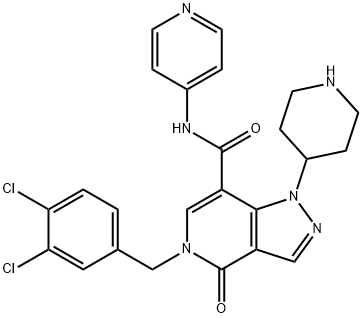
| Chemical Name | 5-[(3,4-dichlorophenyl)methyl]-4-oxo-1-piperidin-4-yl-N-pyridin-4-ylpyrazolo[4,3-c]pyridine-7-carboxamide |
| Synonyms | BDP 13176; BDP-13176; BDP13176; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | The fascin-1 bundling activity is inhibited by BDP-13176 (0-1 μM) [1]. |
| ln Vitro |
The fascin-1 bundling activity is inhibited by BDP-13176 (0-1 μM) [1]. BDP-13176 inhibited fascin-mediated bundling of filamentous actin (F-actin) in a biochemical assay with an IC50 of 240 ± 0.01 nM.[1] The compound exhibited favorable physicochemical properties: low lipophilicity (LogD7.4 = 1.8) and reasonable kinetic solubility (midpoint = 65 µM).[1] It demonstrated moderate stability in human and mouse liver microsomes (Human Clint = 6.4 µL/min/mg protein; Mouse Clint = 11.8 µL/min/mg protein).[1] Caco-2 permeability assay showed low permeability (A-B Papp = 0.15 x 10-6 cm/s) and high efflux (efflux ratio = 37.8).[1] |
| Enzyme Assay |
The binding affinity of BDP-13176 for fascin was determined using Surface Plasmon Resonance (SPR). Serial dilutions of the compound were injected over a fascin-immobilized chip, and the equilibrium dissociation constant (Kd) was calculated from the sensorgram data, yielding a Kd of 85 ± 0.02 nM.[1] Isothermal Titration Calorimetry (ITC) was also used to confirm binding, where aliquots of the compound were titrated into a fascin solution, and the heat change was measured to determine the Kd, which was 50 nM.[1] A fluorescence-based in vitro F-actin bundling assay was developed to measure functional inhibition. Fascin was incubated with pre-polymerized, pyrene-labeled F-actin in the presence of varying concentrations of BDP-13176. The decrease in fluorescence due to bundling (and subsequent sedimentation) was monitored, and the concentration causing 50% inhibition (IC50) was calculated.[1] |
| ADME/Pharmacokinetics |
BDP-13176 exhibited low lipophilicity with a measured LogD7.4 of 1.8.[1] Its kinetic solubility was determined to be 65 µM (midpoint).[1] In vitro metabolic stability was assessed in human and mouse liver microsomes, showing moderate intrinsic clearance (Human Clint = 6.4 µL/min/mg protein; Mouse Clint = 11.8 µL/min/mg protein).[1] Permeability was evaluated using a Caco-2 cell monolayer model. The apparent permeability from apical to basolateral side (A-B Papp) was low at 0.15 x 10-6 cm/s, and the compound showed high efflux with an efflux ratio of 37.8.[1] |
| References |
[1]. Structure-based design, synthesis and biological evaluation of a novel series of isoquinolone and pyrazolo[4,3-c]pyridine inhibitors of fascin 1 as potential anti-metastatic agents. Bioorg Med Chem Lett. 2019;29(8):1023-1029. |
| Additional Infomation |
Fascin is an actin-bundling protein overexpressed in invasive cancers and promotes cell migration and invasion. Inhibiting fascin is a potential anti-metastatic strategy.[1] BDP-13176 was developed through structure-based design starting from a fragment hit. It binds in an induced pocket between the first two β-trefoil domains of fascin, causing a substantial conformational change in domain 1. This change is proposed to deform fascin's actin-binding regions, thereby disrupting its bundling activity.[1] The compound represents a promising starting point for developing fascin-targeted therapies.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~125 mg/mL (~251.32 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (4.18 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0105 mL | 10.0527 mL | 20.1054 mL | |
| 5 mM | 0.4021 mL | 2.0105 mL | 4.0211 mL | |
| 10 mM | 0.2011 mL | 1.0053 mL | 2.0105 mL |