BD-AcAc 2, usually added in diet and given orally, can elevate mean blood ketone bodies of 3.5 mm and lowered plasma glucose, insulin, and leptin in animals.
Physicochemical Properties
| Molecular Formula | C8H16O4 |
| Molecular Weight | 176.21024 |
| Exact Mass | 176.105 |
| CAS # | 1208313-97-6 |
| Related CAS # | (S,S)-BD-AcAc 2;1251829-99-8 |
| PubChem CID | 44631890 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.102 |
| Boiling Point | 269 ºC |
| Flash Point | 101 ºC |
| LogP | 0.071 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 12 |
| Complexity | 135 |
| Defined Atom Stereocenter Count | 2 |
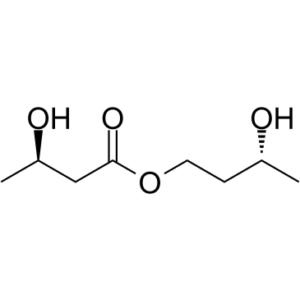
| SMILES | C[C@@H](O)CC(OCC[C@H](O)C)=O |
| InChi Key | AOWPVIWVMWUSBD-RNFRBKRXSA-N |
| InChi Code | InChI=1S/C8H16O4/c1-6(9)3-4-12-8(11)5-7(2)10/h6-7,9-10H,3-5H2,1-2H3/t6-,7-/m1/s1 |
| Chemical Name | [(3R)-3-hydroxybutyl] (3R)-3-hydroxybutanoate |
| Synonyms | BD-AcAc-2; BD-AcAc 2; BD AcAc 2 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo |
(R,R)-BD-AcAc 2 (D-3HHB) enhances blood Na+ and creatinine levels, decreases Aldh3b2 gene expression, increases blood glucose concentration moderately, boosts particular muscle strength, and lowers plasma free fatty acid concentration [1]. Racemic 1,3-butanediol acetoacetate diester was shown to increase ketone body concentrations in the blood of both a pig and a dog. [2] More recent studies in rats indicate it induces a rapid increase in (R)-β-hydroxybutyric acid concentration in the blood and a decrease in blood glucose concentration, indicative of ketosis. [2] (±)-1,3-Butanediol acetoacetate diester has been shown to suppress seizure activity in rats. [2] Combining (±)-1,3-butanediol acetoacetate diester with hyperbaric oxygen therapy was shown to slow metastatic cancer growth in mice. [2] |
| Animal Protocol |
Animal/Disease Models: Septic mice [1] Doses: 10, 20, 40 and 80 mmol/kg/day Route of Administration: PO Experimental Results: Specific muscle strength increased to 40 mmol/kg/day compared to placebo Healthy 93% of control levels. There was a modest increase in blood glucose concentrations of 40 mmol/kg/day compared to placebo. A dose of 40 mmol/kg/day diminished Aldh3b2 gene expression compared with placebo. Caused a further moderate increase in blood Na+ levels and an increase in blood creatinine levels to 20 mmol/kg/day. Plasma free fatty acid concentrations decrease by 10 or 20 mmol/kg/day. Liver gene expression levels of Aldh1a7 were also diminished by sepsis but increased by D-3HHB. 28-Day Oral Toxicity Study: Male and female CrI:WI (Wistar) rats (approximately 9 weeks old) were used. The test group received a diet formulated to contain 30% of calories from (R)-3-hydroxybutyl (R)-3-hydroxybutyrate (ketone monoester), which resulted in daily intakes of 12.0 g/kg body weight/day for males and 15.1 g/kg body weight/day for females. The diet was prepared by mixing rodent chow powder with sugar-free jelly crystals, then adding a heated mixture of water and the ketone monoester to form pellets. Control groups received isocaloric diets where the ketone monoester calories were replaced with either carbohydrate (corn starch) or fat (palm oil). All diets were matched for protein content. Animals were fed ad libitum for 28 days. Body weights and food consumption were recorded regularly. At termination (day 29), blood was collected for hematology, coagulation, and clinical chemistry analysis. Urine was collected for urinalysis. A full gross necropsy was performed, and organs were weighed. Liver, kidneys, gastrointestinal tract sections, brain, heart, and skeletal muscle were preserved for histopathological examination. [3] Developmental Toxicity Study: Pregnant CrI:WI (Han) rats were administered (R)-3-hydroxybutyl (R)-3-hydroxybutyrate (neat, undiluted) at a dosage of 2000 mg/kg body weight/day (2 g/kg/day) via oral gavage from gestation days (DG) 6 through 20. The control group received an equivalent volume of reverse osmosis (RO) water. Dams were observed for clinical signs, and body weights and food consumption were recorded. On DG 21, dams were euthanized, subjected to Caesarean section, and examined. Maternal blood was collected for hematology and clinical chemistry. The uterus was examined for implantation sites, resorptions, and live fetuses. Fetuses were examined for external, visceral, and skeletal alterations. [3] |
| ADME/Pharmacokinetics |
(±)-1,3-Butanediol acetoacetate diester is metabolically hydrolyzed to generate two equivalents of acetoacetic acid and (±)-1,3-butanediol. [2] The (±)-1,3-butanediol can be oxidized to generate β-hydroxybutyric acid. The oxidation of the (R)-enantiomer occurs in the liver. (S)-β-Hydroxybutyric acid is not naturally occurring, but its metabolism in perfused rat livers has been shown to generate ketone bodies through an unknown pathway. [2] |
| Toxicity/Toxicokinetics |
28-Day Study: Rats fed the ketone monoester diet consumed less food and gained significantly less body weight than control groups fed carbohydrate or fat diets. Hematology showed significantly higher red blood cell (RBC) count, hemoglobin (Hb), and hematocrit (Hct) in both sexes, and higher reticulocytes in males compared to controls. All values remained within normal physiological ranges. Activated partial thromboplastin time (APTT) was slightly shorter in the ketone ester group but within normal range. Clinical chemistry showed significantly higher creatine kinase (CK), albumin, and alanine aminotransferase (ALT) in males, and higher lactate dehydrogenase (LDH) in both sexes compared to controls. Most values were within normal ranges, though LDH was slightly above the historical upper limit in some cases. Cholesterol levels were significantly higher in the ketone ester-fed rats compared to controls. No treatment-related abnormalities were found in urinalysis. Absolute uterine weight was lower in females fed the ketone ester diet, but this was attributed to their lower body weight, with relative uterine weight being comparable. Histopathology revealed liver vacuolation (lipid accumulation) in all female rats across all diet groups (test and controls) and in a few male controls. Minor necroinflammatory foci in the liver and minimal myocyte necrosis/focal histiocytosis in skeletal muscle were observed in some animals across all groups. These findings were considered background or diet-dilution related, not specifically due to the ketone monoester. No other toxicologically significant histopathological lesions were attributed to the test article. [3] Developmental Toxicity Study: Dams administered 2 g/kg/day of ketone monoester showed reduced body weight gain (after correction for gravid uterine weight) and reduced food consumption compared to controls. Maternal hematology showed higher segmented neutrophils and lower basophils in the test group, considered normal biological variation. Maternal clinical chemistry showed significantly lower ALT and ALP in the test group. No gross lesions were observed at necropsy. No dead fetuses were observed. Caesarean-sectioning and litter parameters (e.g., number of corpora lutea, implantations, live fetuses) were not affected by treatment. Male fetal body weights were significantly lower in the test group, but the combined fetal weight was not significantly different, and the difference was less than 5%. The overall incidence of fetal alterations (any observed) was significantly higher in the test group (8% of fetuses) compared to controls (4%). This increase was driven by skeletal variations, but the incidence of skeletal variations itself was not statistically different between groups. One fetus in the test group had a medial cleft palate (incompletely ossified). No specific pattern of teratogenicity was attributable to the test substance. [3] |
| References |
[1]. Efficacy and safety of ketone ester infusion to prevent muscle weakness in a mouse model of sepsis-induced critical illness. Sci Rep. 2022 Jun 22;12(1):10591. [2]. The Chemistry of the Ketogenic Diet: Updates and Opportunities in Organic Synthesis. Int J Mol Sci. 2021 May 15;22(10):5230. [3]. Oral 28-day and developmental toxicity studies of (R)-3-hydroxybutyl (R)-3-hydroxybutyrate. Regul Toxicol Pharmacol. 2012 Jul;63(2):196-208. |
| Additional Infomation |
(±)-1,3-Butanediol acetoacetate diester is a synthetic ketogenic compound. [2] It was synthesized from (±)-1,3-butanediol and t-butyl acetoacetate via a transesterification reaction, published in 1995. [2] The use of synthetic ketogenic compounds like (±)-1,3-butanediol acetoacetate diester to supplement a normal diet has shown the capability to induce ketosis and decrease blood glucose concentration without affecting triglyceride or cholesterol levels. [2] It represents a potential alternative to the strict ketogenic diet for inducing nutritional ketosis. [2] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~100 mg/mL (~567.50 mM) DMSO : ~100 mg/mL (~567.50 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5.5 mg/mL (31.21 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 55.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 5.5 mg/mL (31.21 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 55.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 5.5 mg/mL (31.21 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 55.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (567.50 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.6750 mL | 28.3752 mL | 56.7505 mL | |
| 5 mM | 1.1350 mL | 5.6750 mL | 11.3501 mL | |
| 10 mM | 0.5675 mL | 2.8375 mL | 5.6750 mL |