BCTC is a selective and orally bioactive vanilloid receptor 1 (TRPV1 receptor) antagonist. Inhibits acid- and capsaicin-induced activation of rat TRPV1 receptors (IC50 are 6.0 and 35 nM respectively). BCTC significantly reduced the accompanying thermal and mechanical hyperalgesia (3 mg/kg and 10 mg/kg p.o., respectively). BCTC also reduced mechanical hyperalgesia and tactile allodynia 2 weeks after partial sciatic nerve injury (10 and 30 mg/kg p.o.). BCTC did not affect motor performance on the rotarod after administration of doses up to 50 mg/kg p.o.
Physicochemical Properties
| Exact Mass | 372.171 |
| CAS # | 393514-24-4 |
| Related CAS # | 393514-24-4; |
| PubChem CID | 9929425 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 561.6±50.0 °C at 760 mmHg |
| Flash Point | 293.4±30.1 °C |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.610 |
| LogP | 4.08 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 26 |
| Complexity | 465 |
| Defined Atom Stereocenter Count | 0 |
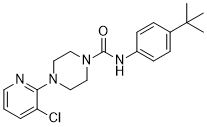
| SMILES | O=C(N1CCN(C2=NC=CC=C2Cl)CC1)NC3=CC=C(C(C)(C)C)C=C3 |
| InChi Key | ROGUAPYLUCHQGK-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H25ClN4O/c1-20(2,3)15-6-8-16(9-7-15)23-19(26)25-13-11-24(12-14-25)18-17(21)5-4-10-22-18/h4-10H,11-14H2,1-3H3,(H,23,26) |
| Chemical Name | N-(4-tert-butylphenyl)-4-(3-chloropyridin-2-yl)piperazine-1-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | On DU145 cells, BCTC (20-100 μM; 72 h) exhibits very specific anti-tumor action [1]. By selectively controlling the expression levels of a handful of cell cycle regulatory proteins, BCTC (20-100 μM; 48 h) produces cell cycle arrest in the G0/G1 phase without inducing apoptosis (10 μM and 100 μM; 72 h). Demise [1]. Cell migration and invasion are inhibited by BCTC (10μM and 100μM; 48 h) [1]. Rat spinal cord TRPV1 function is effectively inhibited by BCTC (3-300 nM), which also reduces capsaicin (300 nM)-induced calcitonin gene-related peptide-like immunoreactivity (CGRP-LI) (IC50=37.0 nM) and p-like substance immunoreactivity in rat spinal cord slices (SP-LI) (IC50=36.0 nM) [3]. |
| ln Vivo | By targeting VR1, BCTC (1-30 mg/kg; oral administration; single dosage) has analgesic effects and can reduce neuropathic and inflammatory thermal allodynia as well as mechanical hyperalgesia in Sprague-Dawley rats[2]. In diabetic ob/ob mice, BCTC (10–100 mg/kg; oral dosing, twice daily for 4 weeks) can enhance insulin production and improve systemic glucose and lipid metabolism as well as insulin resistance [5]. |
| Cell Assay |
Western Blot Analysis[1] Cell Types: DU145 Tested Concentrations: 20 μM, 40 μM, 60 μM, 80 μM, 100 μM Incubation Duration: 48 h Experimental Results: Down-regulates p-Akt, while p-GSK-3β is up-regulated leaving their unphosphorylated form unchanged. Dramatically down-regulated Cyclin D1(20), the most relevant protein in the cell cycle, without affecting cyclin-B1. decreased the expression of CDK2 and CDK6, but without affecting the expression level of CDK4. Downregulates MMP2 and p-FAK levels. Cell Viability Assay[1] Cell Types: DU145, PNT1A Tested Concentrations: 20 μM, 40 μM, 60 μM, 80 μM, 100 μM Incubation Duration: 72 h Experimental Results: diminished the growth of DU145 cells in a concentration -dependent manner, with 12.03% and 50.69% growth inhibition at 10 µM and 100 µM, respectively, but had little effect on normal prostate PNT1A cells. |
| Animal Protocol |
Animal/Disease Models: Capsaicin-induced SD (Sprague-Dawley) rats model[2] Doses: 1 mg/kg, 3 mg/kg, 10 mg/kg, 30 mg/kg Route of Administration: po (oral gavage), Single dose. Before capsaicin treatment ( 30 μg; intraplantar injection; Single dose) Experimental Results: Inhibited capsaicin-mediated thermal hyperalgesia in a dose-dependent manner. Animal/Disease Models: Freund's complete adjuvant (FCA) SD (Sprague-Dawley) rats model[2] Doses: 1 mg/kg, 3 mg /kg, 10 mg/kg, 30 mg/kg Route of Administration: po (oral gavage), Single dose. After 100 % FAC treatment (50 μL; intraplantar injection; Single dose) Experimental Results: Dramatically decreased FAC-induced inflammation-related thermal pain and mechanical hyperalgesia, and extended the inhibitory effect of mechanical hyperalgesia to 6 h at high doses (10 mg/kg, 30 mg/kg). Animal/Disease Models: Partial sciatic nerve ligation SD (Sprague-Dawley) rats model[2] Doses: 1 mg/ kg, 3 mg/kg, 10 mg/kg, 30 mg/kg Route of Administration: po (oral gavage), Single dose. After partial sciatic nerve ligation. Experimental Results: decreased post-operative ab |
| References |
[1]. Liu T, et al. Anti-tumor activity of the TRPM8 inhibitor BCTC in prostate cancer DU145 cells. Oncol Lett. 2016 Jan;11(1):182-188. [2]. Pomonis JD, et al. N-(4-Tertiarybutylphenyl)-4-(3-cholorphyridin-2-yl)tetrahydropyrazine -1(2H)-carbox-amide (BCTC), a novel, orally effective vanilloid receptor 1 antagonist with analgesic properties: II. in vivo characterization in rat models of inflamm [3]. Valenzano KJ, et al. N-(4-tertiarybutylphenyl)-4-(3-chloropyridin-2-yl)tetrahydropyrazine -1(2H)-carbox-amide (BCTC), a novel, orally effective vanilloid receptor 1 antagonist with analgesic properties: I. in vitro characterization and pharmacokinetic pro |
| Additional Infomation | 1-piperazinecarboxamide, 4-(3-chloro-2-pyridinyl)-n-[4-(1,1-dimethylethyl)phenyl]- is a member of piperazines and a member of pyridines. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~134.09 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.70 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.70 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |