Physicochemical Properties
| Molecular Formula | C27H22F3N5O2 |
| Molecular Weight | 505.491096019745 |
| Exact Mass | 505.172 |
| CAS # | 2499499-26-0 |
| PubChem CID | 155400340 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 4.7 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 37 |
| Complexity | 867 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | JKZZCHSVFXZBAI-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C27H22F3N5O2/c28-27(29,30)21-16-19(9-10-23(21)35-11-13-37-14-12-35)26(36)32-20-5-1-3-17(15-20)7-8-18-4-2-6-22-24(18)25(31)34-33-22/h1-6,9-10,15-16H,11-14H2,(H,32,36)(H3,31,33,34) |
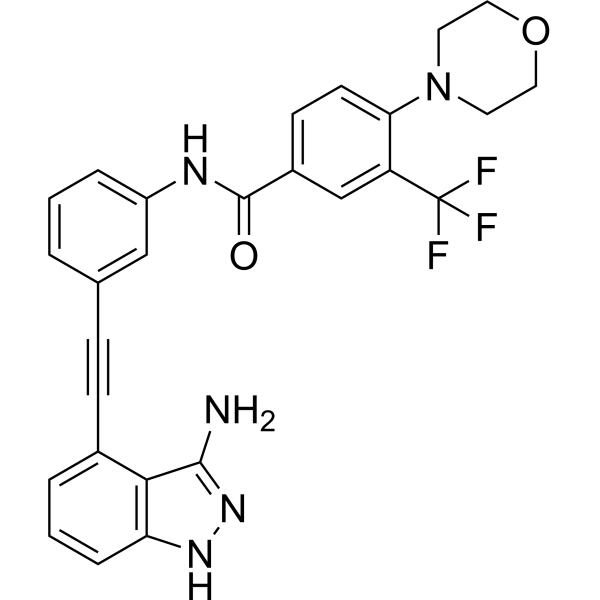
| Chemical Name | N-[3-[2-(3-amino-1H-indazol-4-yl)ethynyl]phenyl]-4-morpholin-4-yl-3-(trifluoromethyl)benzamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | When bound to c-Src, a closely related kinase domain of Bcr-Abl, BCR-ABL-IN-6 (10 μM; 1 hour) exhibits greater cellular efficacy than imatinib [1]. Bcr-Abl phosphorylation is dose-dependently inhibited by BCR-ABL-IN-6 (10 μM; 1 hour), which has a specific antiproliferative impact on Bcr-Abl [1]. Good selective cytotoxicity between K562 and L132 cells is demonstrated by BCR-ABL-IN-6 (10 μM; 1 h) [1]. Strong cytostatic action is shown by BCR-ABL-IN-6 (10 μM) against K562 and HL60 cells [1]. Excellent selective antiproliferative activities against Bcr-Abl-positive leukemia cells K562 are demonstrated by BCR-ABL-IN-6 (10 μM) [1]. |
| ln Vivo | BCR-ABL-IN-6 (5 and 10 mg/kg; male ICR mice; 9 h) required 0.6 h to reach maximum concentration (Cmax). The AUClast values of BCR-ABL-IN-6 were 14018.7 ng·h/mL and 174.7 ng·h/mL when administered intravenously and orally. BCR-ABL-IN-6 is more effective when administered intravenously, but less effective when administered orally [1]. |
| Cell Assay |
Western Blot Analysis[1] Cell Types: K562 Cell Line Tested Concentrations: 0.003, 0.01, 0.03, 0.1 and 0.3 μM Incubation Duration: 1 hour Experimental Results: Demonstrated dose-dependent inhibition of Bcr-Abl phosphorylation. Cell viability assay [1] Cell Types: K562 and L132 cell lines Tested Concentrations: 10 μM Incubation Duration: 1 h Experimental Results: The cell activity of Bcr-Abl positive leukemia K562 cells demonstrated a GI50 of less than 160 nM, and it was better than imatin. The cellular potency GI50 of nitrate is 0.02 μM. It exhibits a selective cytotoxic effect on normal cells L132, with a GI50 of 9.27 μM. |
| Animal Protocol |
Animal/Disease Models: Male ICR mouse[1] Doses: 5 mg/kg and 10 mg/kg Route of Administration: intravenous (iv) (iv)injection and oral administration; 5 and 10 mg/kg; 9h Experimental Results: Oral administration is not as effective as intravenous (iv) (iv)injection, and intravenous (iv) (iv)injection is more effective better. |
| References |
[1]. Design, synthesis, and biological evaluations of novel 3-amino-4-ethynyl indazole derivatives as Bcr-Abl kinase inhibitors with potent cellular antileukemic activity. European Journal of Medicinal Chemistry, 2020, 207:112710. [2]. Design, synthesis, and biological evaluations of novel 3-amino-4-ethynyl indazole derivatives as Bcr-Abl kinase inhibitors with potent cellular antileukemic activity. European Journal of Medicinal Chemistry, 2020, 207:112710. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9783 mL | 9.8914 mL | 19.7828 mL | |
| 5 mM | 0.3957 mL | 1.9783 mL | 3.9566 mL | |
| 10 mM | 0.1978 mL | 0.9891 mL | 1.9783 mL |