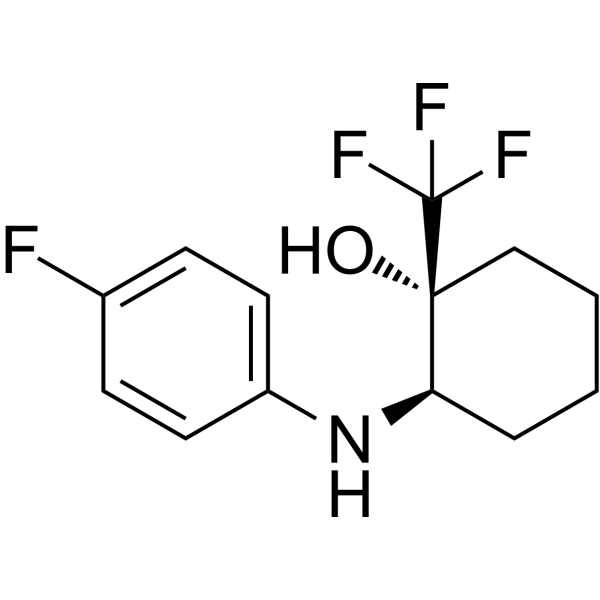
Physicochemical Properties
| Molecular Formula | C13H15F4NO |
| Molecular Weight | 277.26 |
| Exact Mass | 277.108 |
| CAS # | 2741956-55-6 |
| PubChem CID | 155539293 |
| Appearance | Yellow to brown ointment |
| LogP | 3.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 19 |
| Complexity | 302 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | C1CC[C@@]([C@@H](C1)NC2=CC=C(C=C2)F)(C(F)(F)F)O |
| InChi Key | IESAJAZKMLPVIB-VXGBXAGGSA-N |
| InChi Code | InChI=1S/C13H15F4NO/c14-9-4-6-10(7-5-9)18-11-3-1-2-8-12(11,19)13(15,16)17/h4-7,11,18-19H,1-3,8H2/t11-,12-/m1/s1 |
| Chemical Name | (1R,2R)-2-(4-fluoroanilino)-1-(trifluoromethyl)cyclohexan-1-ol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | IC50: 16 nM (hTRPA1 FLIPR), 82 nM (hTRPA1 Ephys), 63 nM (rTRPA1 FLIPR), 35 nM (rDRG Ephys), 73 nM (mTRPA1), 68 nM (gpTRPA1), 81 nM (dogTRPA1)m, 19 nM (monkeyTRPA1)[1] |
| ln Vitro | hTRPA1 FLIPR, hTRPA1 Ephys, rTRPA1 FLIPR, and rDRG Ephys are all inhibited by BAY-390, with IC50 values of 16, 82, 63, and 35 nM, respectively[1]. The inhibitory concentrations of BAY-390 are 73, 68, 81, and 19 nM for mTRPA1, gpTRPA1, dogTRPA1, and monkeyTRPA1, respectively[1]. |
| ln Vivo | Neuropathic pain is affected in vivo by BAY-390 (30 and 90 mg/kg; po; BID for 10 days)[1]. In rat models of cystitis produced by cyclophosphamide, BAY-390 decreases visceral pain[1]. In models of neurogenic inflammation and inflammatory pain, BAY-390 is effective[1]. |
| Animal Protocol |
Animal/Disease Models: Nrodent animals with neuropathic pain[1] Doses: 30 and 90 mg/kg Route of Administration: Oral gavage; 30 and 90 mg/kg; twice (two times) daily for 10 days Experimental Results: Effectively decreased the neuropathic pain in rodent neuropathic pain model. |
| References | [1]. |
Solubility Data
| Solubility (In Vitro) | DMSO: 100 mg/mL (360.67 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.02 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (9.02 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6067 mL | 18.0336 mL | 36.0672 mL | |
| 5 mM | 0.7213 mL | 3.6067 mL | 7.2134 mL | |
| 10 mM | 0.3607 mL | 1.8034 mL | 3.6067 mL |