Physicochemical Properties
| Molecular Weight | 139.15516 |
| Exact Mass | 139.075 |
| Elemental Analysis | C, 51.79; H, 6.52; N, 30.20; O, 11.50 |
| CAS # | 654666-65-6 |
| PubChem CID | 55285412 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 0.089 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 10 |
| Complexity | 209 |
| Defined Atom Stereocenter Count | 0 |
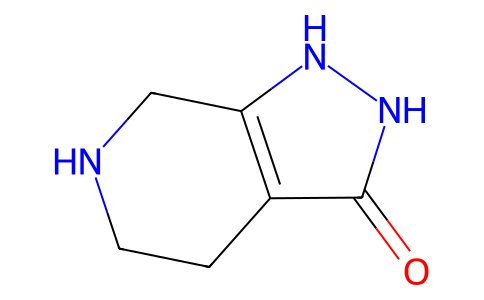
| SMILES | C1CNCC2=C1C(=O)NN2 |
| InChi Key | UPMMUUFLHUEUKP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H9N3O/c10-6-4-1-2-7-3-5(4)8-9-6/h7H,1-3H2,(H2,8,9,10) |
| Chemical Name | 1,2,4,5,6,7-hexahydropyrazolo[3,4-c]pyridin-3-one |
| Synonyms | aza-THIP; 654666-65-6; 1H,4H,5H,6H,7H-pyrazolo[3,4-c]pyridin-3-ol; 1H,4H,5H,6H,7H-pyrazolo(3,4-c)pyridin-3-ol; 1,2,4,5,6,7-Hexahydro-3H-pyrazolo[3,4-c]pyridin-3-one; 4,5,6,7-tetrahydro-2H-pyrazolo[3,4-c]pyridin-3-ol; 4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridin-3-ol; 4,5,6,7-tetrahydropyrazolo[5,4-c]pyridin-3-ol; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | GABA-C receptor |
| ln Vitro | The potency of a series of eight compounds structurally related with 4,5,6,7-tetrahydroisoxazolo[5,4-c]pyridin-3-ol (THIP), a potent GABA(A) partial agonist exhibiting GABA(C) rho(1) antagonist effect (K(i)=25 microM), was determined electrophysiologically using homomeric human GABA(C) rho(1) receptors expressed in Xenopus oocytes. Protolytic properties (pK(a) values for the acidic bioisosteric groups) and the presence of steric bulk in the molecules appear to be structural parameters of importance for blockade of the GABA(C) rho(1) receptor. Within this series of moderately potent GABA(C) antagonists, only 4,5,6,7-tetrahydropyrazolo[5,4-c]pyridin-3-ol (Aza-THIP) does not interact detectably with GABA(A) receptors, and Aza-THIP has the potential of being a useful tool for molecular and behavioural pharmacological studies. [1] |
| Enzyme Assay |
Electrophysiological recording [1] Xenopus laevis was anaesthesized with 0.17% ethyl 3-aminobenzoate and a lobe of the ovaries was carefully removed. The lobe of the ovary was placed in oocyte releasing buffer 2 (OR2) (82.5 mM NaCl, 2 mM KCl, 1 mM MgCl2 · 6H2O, 5 mM HEPES, pH 7.5) with 2 mg/mL Collagenase A for 2 h. Defolliculated oocytes were then rinsed with frog Ringer solution (96 mM NaCl, 2 mM KCl, 1 mM MgCl2 · 6H2O, 1.8 mM CaCl2, 5 mM HEPES, pH 7.5) supplemented with 2.5 mM pyruvate, 0.5 mM theophylline and 50 μg/mL gentamycin. Stage V–VI oocytes were collected. Receptor activity was measured by two electrode voltage clamp recording using a Geneclamp 500 amplifier, a MacLab 2e recorder and Chart version 3.5 program. Oocytes were voltage clamped at −60 mV and continuously superfused with frog Ringer solution (96 mM NaCl, 2 mM KCl, 1 mM MgCl2 · 2H2O, 1.8 mM CaCl2 · 2H2O, 5 mM HEPES). For receptor activation measurements, the indicated concentrations of drug were added to the buffer solution. Antagonist activity was measured as a pKi or an IC50 value. The pKi value was determined on the basis of a GABA concentration-response curve as a control followed by a GABA concentration-response curve in the presence of a fixed antagonist concentration on the same oocyte (n≥3). The IC50 value was determined by measuring the activation of the receptor by GABA in the presence of different antagonist concentrations. The GABA concentration was chosen as the concentration which produces 50% of max response, e.g., EC50, based on a standard GABA concentration–response curve for each oocyte prior to the inhibition curve, thereby using a fixed relative response level of GABA. |
| References | [1]. Aza-THIP and related analogues of THIP as GABA C antagonists. Bioorg Med Chem. 2003 Nov 17;11(23):4891-6. |
| Additional Infomation | TPMPA has so far been the most useful antagonist for studying GABAC receptors. However, Aza-THIP, which has now been shown to be a specific competitive GABAC antagonist, may be a supplement to TPMPA as a reference GABAC compound. TPMPA shows strong antagonist activity at GABAC receptors and, in contrast to Aza-THIP, weak activity at both GABAA (Table 1) and metabotropic GABAB receptors.6 Using a series of compounds related to THIP, we have shown that only minor structural changes affect their potency as GABAC antagonists. The acidic character of the 3-isoxazolol ring of Iso-THIP (pKa 3.0) is more pronounced than that of the 3-isoxazolol ring of THIP (pKa 4.4) (Table 1), suggesting that this difference in potency may be explained by a stronger electrostatic interaction of Iso-THIP than of THIP with the receptor. The structural and protolytic similarity between THIP and Aza-THIP suggests that the latter compound, like THIP, is capable of penetrating the blood-brain-barrier. Thus, although Aza-THIP is only moderately potent as a GABAC antagonist, it may be a useful compound for behavioural pharmacological studies. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.1860 mL | 35.9299 mL | 71.8597 mL | |
| 5 mM | 1.4372 mL | 7.1860 mL | 14.3719 mL | |
| 10 mM | 0.7186 mL | 3.5930 mL | 7.1860 mL |