Physicochemical Properties
| Exact Mass | 458.21 |
| Elemental Analysis | C, 49.72; H, 6.07; Cl, 13.34; N, 15.81; O, 9.03; S, 6.03 |
| CAS # | 170956-82-8 |
| Related CAS # | 191588-96-2 (HCl);170956-82-8 (2HCl);151140-97-5 (3HCl);151140-96-4; 171171-42-9 (fumarate); 171171-42-9; |
| Appearance | Solid powder |
| LogP | 1.8 |
| InChi Key | LXDKLZKTSZKRLF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H30N6O3S.2ClH/c1-23-32(29,30)15-17-5-6-20-19(12-17)18(13-25-20)4-3-7-27-8-10-28(11-9-27)22-21(31-2)14-24-16-26-22;;/h5-6,12-14,16,23,25H,3-4,7-11,15H2,1-2H3;2*1H |
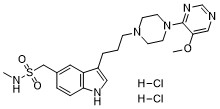
| Chemical Name | 1H-Indole-5-methanesulfonamide, 3-(3-(4-(5-methoxy-4-pyrimidinyl)-1-piperazinyl)propyl)-N-methyl-, dihydrochloride |
| Synonyms | Avitriptan HCl; Avitriptan hydrochloride; Avitriptan dihydrochloride; Avitriptan; BMS 180048; BMS-180048; Avitriptan; 151140-96-4; Avitriptan [INN]; UNII-6RS056L04P; 1-[3-[3-[4-(5-methoxypyrimidin-4-yl)piperazin-1-yl]propyl]-1H-indol-5-yl]-N-methylmethanesulfonamide; 6RS056L04P; 1H-Indole-5-methanesulfonamide, 3-(3-(4-(5-methoxy-4-pyrimidinyl)-1-piperazinyl)propyl)-N-methyl-; 3-(3-(4-(5-Methoxy-4-pyrimidinyl)-1-piperazinyl)propyl)-N-methylindole-5-methanesulfonamide; BMS180048; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 5-HT1 receptor |
| ln Vitro |
Activation of AhR in reporter gene assays was observed for Avitriptan and to a lesser extent for Donitriptan, while other triptans were very weak or no activators of AhR. Using competitive binding assay and by homology docking, we identified Avitriptan as a low-affinity ligand of AhR. Avitriptan triggered nuclear translocation of AhR and increased binding of AhR in CYP1A1 promotor DNA, as revealed by immune-fluorescence microscopy and chromatin immune-precipitation assay, respectively. Strong induction of CYP1A1 mRNA was achieved by Avitriptan in wild type but not in AhR-knockout, immortalized human hepatocytes, implying that induction of CYP1A1 is AhR-dependent. Increased levels of CYP1A1 mRNA by Avitriptan were observed in human colon carcinoma cells LS180 but not in primary cultures of human hepatocytes. Collectively, we show that Avitriptan is a weak ligand and activator of human AhR, which induces the expression of CYP1A1 in a cell-type specific manner. Our data warrant the potential off-label therapeutic application of Avitriptan as an AhR-agonist drug [1].
Triptans Are Activators of Human AhR. Avitriptan and Donitriptan induce CYP1A1 in Hepatic and Intestinal Cells via AhR. Avitriptan Is a Low-Affinity Ligand of AhR. Avitriptan and Donitriptan Trigger Nuclear Translocation of AhR. Formation of AhR-ARNT Heterodimer by Avitriptan and Donitriptan. Avitriptan and Donitriptan Enhance the Recruitment of AhR into CYP1A1 Promotor[1]. |
| Enzyme Assay |
Radio-Ligand Binding Assay [1] Cytosolic protein from murine hepatoma Hepa1c1c7 cells (2 mg/mL) was incubated for 2 h at room temperature in the presence of 2 nM [3H]-TCDD with Avitriptan (1–1000 µM), Donitriptan (1–1000 µM), FICZ (10 nM; positive control), dexamethasone (100 nM; negative control) or vehicle (DMSO; 0.1% v/v; corresponds to specific binding of [3H]-TCDD = 100%). Ligand binding to the cytosolic proteins was determined by the hydroxyapatite binding protocol and scintillation counting. Specific binding of [3H]-TCDD was determined as a difference between total and non-specific (TCDF; 200 nM) reactions. Three independent experiments were performed, and the incubations and measurements were done in triplicates in each experiment (technical replicates). Chromatin Immunoprecipitation (ChIP) [1] The assay was performed as per the manufacturer recommendations for SimpleChIP Plus Enzymatic Chromatin IP kit (Magnetic Beads), with minor modifications, as recently described. Briefly, LS174T cells were seeded in a 60-mm dish and the following day they were incubated with Donitriptan (100 µM), Avitriptan (100 µM), TCDD (10 nM) and/or vehicle (0.1% DMSO) 90 min and 18 h at 37 °C. Experiments were performed in two consecutive cell passages. |
| Cell Assay |
Cytotoxicity Assay [1] Cells were incubated for 24 h with tested compounds, vehicle (DMSO; 0.1% v/v) and Triton X-100 (1%, v/v), using multi-well culture plates of 96 wells. MTT test was performed and absorbance was measured spectrophotometrically at 540 nm on Infinite M200. The data were expressed as the percentage of cell viability, where 100% and 0% represent the treatments with vehicle and Triton X-100, respectively. Reporter Gene Assay [1] The stably transfected human hepatoma gene reporter cells AZ-AHR [36] were seeded at 96-well culture plates and incubated with test compounds as indicated in detail in figure legends. Thereafter, the cells were lysed, and luciferase activity was measured on a Tecan Infinite M200 Pro plate reader. Measurements were carried out in quadruplicates (technical replicates). Isolation of RNA and qRT-PCR [1] The total RNA was isolated by TRI Reagent® and cDNA was synthesized using M-MuLV Reverse Transcriptase in the presence of random hexamers. The levels of CYP1A1 and glyceraldehyde-3-phosphate dehydrogenase [GAPDH] mRNAs were determined using the Light Cycler® 480 II apparatus, as described elsewhere. Measurements were carried out in triplicates. Gene expression was normalized to GAPDH as a housekeeping gene. The data were processed by the delta-delta method. Western Blotting [1] Total protein extracts were prepared by using ice-cold lysis buffer (150 mM NaCl; 50 mM HEPES; 5 mM EDTA; 1% (v/v) Triton X-100; anti-protease cocktail, anti-phosphatase cocktail). Protein concentration was determined using Bradford reagent. The amount of protein was adjusted to 25 µg per sample. Samples were separated at standard SDS-PAGE followed by western blotting. The following primary antibodies were used for the detection of target proteins—CYP1A1 (mouse-monoclonal, sc-393979, A-9, dilution 1:500) and β-actin (mouse-monoclonal, sc-47778, C4, dilution 1:2000). Chemiluminescent detection was performed using horseradish peroxidase-conjugated secondary antibodies and WesternSure® PREMIUM Chemiluminescent Substrate by C-DiGit® Blot Scanner. Experiments were performed in three consecutive cell passages. Nuclear Translocation of AhR–Immune Histochemistry [1] LS174T cells were seeded on chamber slides and cultured for two days. Then, cells were incubated for 90 min with vehicle (DMSO; 0.1% v/v), TCDD (10 nM), Avitriptan (100 μM) and Donitriptan (100 μM). After the treatment, cells were washed by PBS, fixed with 4% formaldehyde, permeabilized using 0.1% Triton X-100, blocked with 3% bovine serum albumin and incubated with Alexa Fluor 488 labelled primary antibody against AhR, as described previously [13]. Nuclei were stained by 4′,6-diamino-2-phenylindole (DAPI) and cells were enclosed by VectaShield® Antifade Mounting Medium. AhR translocation into the nucleus was visualized and evaluated using fluorescence microscope IX73. The whole staining protocol was performed in three independent experiments with all tested compounds in duplication. The AhR translocation was evaluated visually depending on the distinct signal intensity of AhR antibody in the nucleus and cytosol. |
| References | [1]. Antimigraine Drug Avitriptan Is a Ligand and Agonist of Human Aryl Hydrocarbon Receptor that Induces CYP1A1 in Hepatic and Intestinal Cells. Int J Mol Sci. 2020 Apr 17;21(8):2799. |
| Additional Infomation | We observed cell-specific induction of AhR target gene CYP1A1 in hepatic and intestinal cells. We may only speculate about the mechanisms underlying cell-specific induction, which may comprise differential cellular uptake/intake, metabolism of Avitriptan or distinct interactions with the AhR signaling pathway in cancer (LS180), immortalized (MIHA) and normal cells (primary human hepatocytes). Nevertheless, the lack of CYP1A1 induction in primary cultures of normal human hepatocytes may be considered favorable, in terms of no AhR systemic effects of orally Avitriptan intended for local intestinal treatments. Based on the data reported in the current study, we propose the possibility to repurpose (off-target use) formerly anti-migraine Avitriptan for local intestinal use as anti-IBD treatment through the AhR. This is supported by the facts that—(i) The AhR is emerging and suitable therapeutic target in IBD; (ii) Avitriptan is a ligand and agonist of the AhR; (iii) Avitriptan passed phase I and phase II of clinical studies, which may accelerate its introduction in clinical use; (iv) Orally Avitriptan has low bioavailability and it is not toxic to intestinal cells, which favors its local use in IBD treatment without having undesirable systemic effects. In conclusion, our data reporting activation of AhR by Avitriptan warrant potential off-label therapeutic application of Avitriptan as a AhR-agonist drug in the treatment of intestinal inflammatory pathologies. Ongoing studies should focus on in vitro and in vivo anti-inflammatory capability of Avitriptan.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |