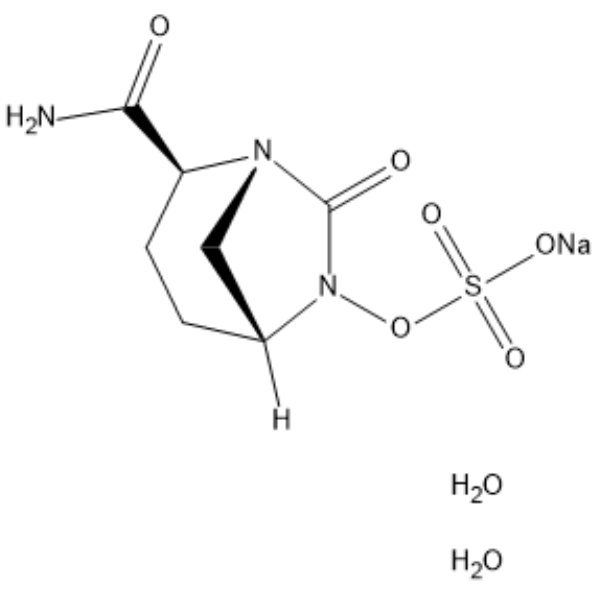
Avibactam sodium dihydrate (NXL-104) is a non-β-lactam and covalent β-lactamase inhibitor that inhibits β-lactamase TEM-1 and CTX-M-15 with IC50s of 8 nM and 5 nM, respectively.
Physicochemical Properties
| Molecular Formula | C7H14N3NAO8S |
| Molecular Weight | 323.26 |
| Exact Mass | 323.039929 |
| PubChem CID | 140885641 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 20 |
| Complexity | 462 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | C1C[C@H](N2C[C@@H]1N(C2=O)OS(=O)(=O)[O-])C(=O)N.O.O.[Na+] |
| InChi Key | ZIJOUXPPZNEISO-LEIZOONBSA-M |
| InChi Code | InChI=1S/C7H11N3O6S.Na.2H2O/c8-6(11)5-2-1-4-3-9(5)7(12)10(4)16-17(13,14)15;;;/h4-5H,1-3H2,(H2,8,11)(H,13,14,15);;2*1H2/q;+1;;/p-1/t4-,5+;;;/m1.../s1 |
| Chemical Name | sodium;[(2S,5R)-2-carbamoyl-7-oxo-1,6-diazabicyclo[3.2.1]octan-6-yl] sulfate;dihydrate |
| Synonyms | Avibactam (sodium dihydrate); Avibactam sodium dihydrate; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | β-lactamase |
| ln Vitro | Avibactam (NXL104) is a molecule with little antibacterial activity, that inhibits class A and C β-lactamases. Avibactam inactivates most important β-lactamases except metallo types and Acinetobacter OXA carbapenemases[2]. |
| ln Vivo | Avibactam sodium displays a slow return of activity with an off-rate of 0.045±0.022 min-1, which converts to a residence time half-life (tt1/2) of 16±8 min. The measured off-rate for Avibactam suggests that slow deacylation through hydrolysis or reversibility is occurring, and it is in contrast to previously reported extremely long t1/2 values of >1 or >7 d for Avibactam inhibition of TEM-1[1]. Avibactam is a new promising β-lactamase inhibitor, to overcome resistance caused by β-lactamases. Mice are infected with ca.106 CFU of Pseudomonas aeruginosa intramuscularly into the thigh or intranasally to cause pneumonia and are given 8 different (single) subcutaneous doses of Ceftazidime and Avibactam in various combined concentrations, ranging from 1 to 128 mg/kg of body weight in 2-fold increases. The mean estimated half-life in plasma of Ceftazidime in the terminal phase is 0.28 h (SD, 0.02 h), and that of Avibactam is 0.24 h (SD, 0.04 h). Volumes of distribution are 0.80 liters/kg (SD, 0.14 liters/kg) and 1.18 liters/kg (SD, 0.34 liters/kg), respectively[3]. |
| Enzyme Assay |
In a 200 μL reaction volume, 1 μM TEM-1 is incubated with and without 5 μM avibactam for 5 min at 37°C and subjected to two ultrafiltration cartridge (UFC) steps to remove excess inhibitor (Ultrafree-0.5 with Biomax membrane, 5-kDa cutoff). Centrifugation at 10,600× g for 8 min is performed at 4°C. After each ultrafiltration step, 20 μL retentate are diluted with 180 μL assay buffer to restore the original enzyme concentration. After two UFC treatments, the amount of free Avibactam is quantified by liquid chromotography/MS/MS and found to be <5% of the original concentration. Loss of protein during UFC is assessed by measuring TEM-1 activity (on 4,000-fold dilution) in the acyl-enzyme sample compare with non-UFC-treated enzyme, and loss is found to be <5%[1]. Acylation Kinetic Measurements. [1] Using a Cary 400 Bio UV visual spectrophotometer outfitted with a temperature controller, reactions were initiated in stirred 1-cm quartz cuvettetes by adding 20 μL 2.5 nM TEM-1 and 980 μL 204 μM nitrocefin solution in the presence or absence of avibactam. Enzyme activity was monitored using a continuous measurement of 460 nm absorbance in 0.1-s intervals between measurements. For data analysis, the offset between reaction initiation and the first absorbance read was 3 s. Experiments at higher avibactam concentrations were performed on a Bio-Logic SFM-4 Stopped-Flow/Quench-Flow instrument using a cuvettete with a 2-mm path length. A three-syringe method was used to give a constant final concentration of 2 nM TEM-1 and 200 μM nitrocefin. The total flow rate was adjusted to 3 mL/s. Absorbance was recorded continuously at 490 nm in 0.002-s intervals. For data analysis, the offset between reaction initiation and the first absorbance read was 250 ms. Deacylation koff Measurement. [1] Enzyme (1 μM) was incubated with inhibitor (5 μM avibactam, 20 μM tazobactam, or 100 μM clavulanic acid) for 5 min at 37 °C and diluted 4,000-fold in the assay buffer with or without inhibitor. The free enzyme control was diluted in the absence of the inhibitor. The background absorbance control omitted the enzyme. After dilution, TEM-1 activity was assayed in a 96-well microtiter plate by adding 20 μL to 180 μL 400 μM nitrocefin for a final TEM-1 concentration of 25 pM. Absorbance at 490 nm was monitored continuously in a Spectramax plate reader. Data were fit to Eq. 1 to obtain koff. In the off-rate experiment, V0 represents fully inhibited enzyme velocity, and it was estimated using a reaction with no TEM-1. VS represents the uninhibited enzyme velocity, and it was measured in a reaction with TEM-1 and no avibactam. The koff value is reported as ±2 SD from three separate determinations. Preparation of TEM-1 Acyl-Enzyme. [1] In a 200 μL reaction volume, 1 μM TEM-1 was incubated with and without 5 μM avibactam for 5 min at 37 °C and subjected to two ultrafiltration cartridge (UFC) steps to remove excess inhibitor (Ultrafree-0.5 with Biomax membrane, 5-kDa cutoff). Centrifugation at 10,600 × g for 8 min was performed at 4 °C. After each ultrafiltration step, 20 μL retentate were diluted with 180 μL assay buffer to restore the original enzyme concentration. After two UFC treatments, the amount of free avibactam was quantified by liquid chromotography/MS/MS and found to be <5% of the original concentration. Loss of protein during UFC was assessed by measuring TEM-1 activity (on 4,000-fold dilution) in the acyl-enzyme sample compared with non-UFC–treated enzyme, and loss was found to be <5%. |
| Cell Assay | Cells (~109 cfu) from overnight broth culture are spread on Mueller-Hinton agar supplemented with either (i) Ceftaroline plus Avibactam (1 or 4 mg/L) at 1-16× the MICs or (ii) Ceftaroline at 1 or 4 mg/L plus Avibactam at 1-8× the concentration needed to reduce the Ceftaroline MIC to 1 or 4 mg/L. Colonies are counted after overnight incubation and representatives are retained[2]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Avibactam and ceftazidime are excreted mainly by the kidneys. The steady state volumes of distribution of avibactam and ceftazidime is 22.2L and 17L respectively. Avibactam and ceftazidime has a clearance of ~12L/h and ~7L/h respectively. Metabolism / Metabolites No metabolism of avibactam was observed in human liver preparations. Unchanged avibactam is the major drug-related component in human plasma and urine. 80-90% of ceftazidime is eliminated as unchanged . Biological Half-Life Ceftazidime-avibactam has a half life of ~2.7-3.0 hours. |
| Toxicity/Toxicokinetics |
Protein Binding 5.7%-8.2% of avibactam is bound to plasma protein, and less than 10% of ceftazidime is protein bound. |
| References | [1]. Avibactam is a covalent, reversible, non-β-lactam β-lactamase inhibitor. Proc Natl Acad Sci U S A. 2012 Jul 17;109(29):11663-8. |
| Additional Infomation |
Avibactam sodium is an organic sodium salt that is the monosodium salt of avibactam. Used in combination with ceftazidime pentahydrate for the treatment of complicated urinary tract infections including pyelonephritis. It has a role as an EC 3.5.2.6 (beta-lactamase) inhibitor, an antibacterial drug and an antimicrobial agent. It contains an avibactam(1-).
AVIBACTAM SODIUM is a small molecule drug with a maximum clinical trial phase of IV (across all indications) that was first approved in 2015 and has 3 approved and 1 investigational indication. Avibactam is a β-lactamase inhibitor that is in clinical development, combined with β-lactam partners, for the treatment of bacterial infections comprising gram-negative organisms. Avibactam is a structural class of inhibitor that does not contain a β-lactam core but maintains the capacity to covalently acylate its β-lactamase targets. Using the TEM-1 enzyme, we characterized avibactam inhibition by measuring the on-rate for acylation and the off-rate for deacylation. The deacylation off-rate was 0.045 min(-1), which allowed investigation of the deacylation route from TEM-1. Using NMR and MS, we showed that deacylation proceeds through regeneration of intact avibactam and not hydrolysis. Other than TEM-1, four additional clinically relevant β-lactamases were shown to release intact avibactam after being acylated. We showed that avibactam is a covalent, slowly reversible inhibitor, which is a unique mechanism of inhibition among β-lactamase inhibitors.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0935 mL | 15.4674 mL | 30.9349 mL | |
| 5 mM | 0.6187 mL | 3.0935 mL | 6.1870 mL | |
| 10 mM | 0.3093 mL | 1.5467 mL | 3.0935 mL |