Physicochemical Properties
| Molecular Formula | C53H72N2O12+2 |
| Molecular Weight | 929.144980000001 |
| Exact Mass | 928.507 |
| CAS # | 64228-79-1 |
| Related CAS # | Atracurium besylate;64228-81-5 |
| PubChem CID | 47319 |
| Appearance | Typically exists as solid at room temperature |
| Melting Point | 185-194ºC |
| LogP | 1.04 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 26 |
| Heavy Atom Count | 67 |
| Complexity | 1390 |
| Defined Atom Stereocenter Count | 0 |
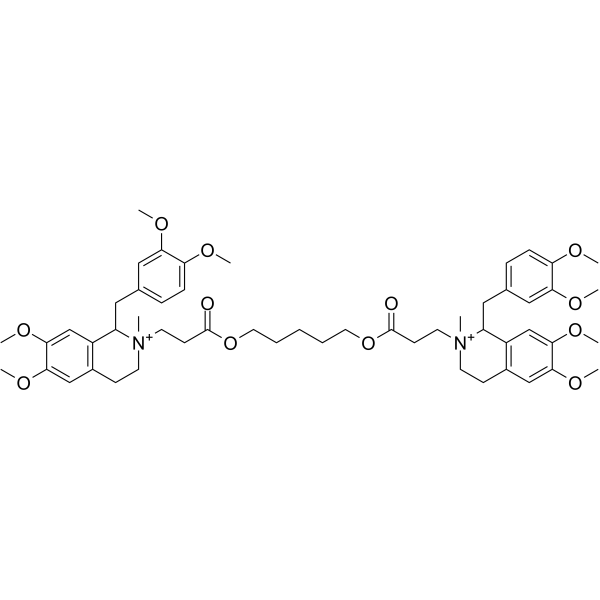
| SMILES | C[N+]1(CCC2=CC(=C(C=C2C1CC3=CC(=C(C=C3)OC)OC)OC)OC)CCC(=O)OCCCCCOC(=O)CC[N+]4(C)CCC5=CC(=C(C=C5C4CC6=CC(=C(C=C6)OC)OC)OC)OC |
| InChi Key | YXSLJKQTIDHPOT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C53H72N2O12/c1-54(22-18-38-32-48(62-7)50(64-9)34-40(38)42(54)28-36-14-16-44(58-3)46(30-36)60-5)24-20-52(56)66-26-12-11-13-27-67-53(57)21-25-55(2)23-19-39-33-49(63-8)51(65-10)35-41(39)43(55)29-37-15-17-45(59-4)47(31-37)61-6/h14-17,30-35,42-43H,11-13,18-29H2,1-10H3/q+2 |
| Chemical Name | 5-[3-[1-[(3,4-dimethoxyphenyl)methyl]-6,7-dimethoxy-2-methyl-3,4-dihydro-1H-isoquinolin-2-ium-2-yl]propanoyloxy]pentyl 3-[1-[(3,4-dimethoxyphenyl)methyl]-6,7-dimethoxy-2-methyl-3,4-dihydro-1H-isoquinolin-2-ium-2-yl]propanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In HSR040622 and HSR040821 cells, atracurium (10 µM; 72 hours) stimulates astrocyte differentiation but not neuronal differentiation [4]. In vitro GSC xenograft-treated mice, atracurium (10 µM; 48 hours) decreases tumor engraftment and increases survival [4]. Rat tetanic contractions completely disappear when exposed to atracurium (2.4 µM; 120 min); rat extensor digitorum longus muscle cells twitch very minimally [5]. |
| ln Vivo | In DBA/2 and SJL mice, atracurium (1, 5, 10, 20, 50 mg/kg; intravenously) causes bronchoconstriction [2]. Rats that receive an intravenous dose of atracurium (4.8 mg/kg) develop neuromuscular blockade [3]. |
| Cell Assay |
Cell Proliferation Assay[4] Cell Types: Glioblastoma Stem Cells (GSC) Tested Concentrations: 3, 10, 20 µM Incubation Duration: 72 hrs (hours) Experimental Results: Percentage of GFP-positive cells increased in a dose-dependent manner from 5.3% in DMSO to 15.4%, 81.1% and 86.8% in 3 μM, 10 μM and 20 μM respectively. |
| Animal Protocol |
Animal/Disease Models: 5-12 weeks, 15-20 g male mice [2] Doses: 1, 5, 10, 20, 50 mg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results: Induced bronchoconstriction and Atracurium-induced airway hyperresponsiveness was abolished in a dose-dependent manner by atropine or pancuronium pretreatment. Animal/Disease Models: 290 ± 30 g male Sprague ± Dawley rats (60 mg/kg heat-killed Corynebacterium parvum intravenously (iv) (iv)(iv)) [3] Doses: 4.8 mg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results: In large mice injected with Corynebacterium parvum Induction of neuromuscular blockade in rats. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the use of atracurium during breastfeeding. Because it is short acting, highly polar and poorly absorbed orally, it is not likely to reach the breastmilk in high concentration or to reach the bloodstream of the infant. When a combination of anesthetic agents is used for a procedure, follow the recommendations for the most problematic medication used during the procedure. Consider using an atracurium product that has no benzyl alcohol preservative. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References |
[1]. Clinical pharmacology of atracurium besylate (BW 33A): a new non-depolarizing muscle relaxant. Anesth Analg. 1982 Sep;61(9):723-9. [2]. Genetic susceptibility to atracurium-induced bronchoconstriction. Am J Respir Crit Care Med. 1995 May;151(5):1537-42. [3]. Inflammatory liver disease shortens atracurium-induced neuromuscular blockade in rats. Eur J Anaesthesiol. 2001 Sep;18(9):599-604. [4]. Atracurium Besylate and other neuromuscular blocking agents promote astroglial differentiation and deplete glioblastoma stem cells. Oncotarget. 2016 Jan 5;7(1):459-72. [5]. Cellular mechanisms of atracurium-induced tetanic fade in the isolated rat muscle. Basic Clin Pharmacol Toxicol. 2004 Jul;95(1):9-14. |
| Additional Infomation |
Atracurium is a diester compound consisting of pentane-1,5-diol with both hydroxyls bearing 3-[1-(3,4-dimethoxybenzyl)-6,7-dimethoxy-2-methyl-3,4-dihydroisoquinolinium-2(1H)-yl]propanoyl groups. It has a role as a muscle relaxant and a nicotinic antagonist. It is a quaternary ammonium ion and a diester. A non-depolarizing neuromuscular blocking agent with short duration of action. Its lack of significant cardiovascular effects and its lack of dependence on good kidney function for elimination provide clinical advantage over alternate non-depolarizing neuromuscular blocking agents. See also: Atracurium Besylate (has salt form). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.0763 mL | 5.3813 mL | 10.7625 mL | |
| 5 mM | 0.2153 mL | 1.0763 mL | 2.1525 mL | |
| 10 mM | 0.1076 mL | 0.5381 mL | 1.0763 mL |