Physicochemical Properties
| Molecular Formula | C21H27N5O7S |
| Molecular Weight | 493.53338 |
| Exact Mass | 493.163 |
| Elemental Analysis | C, 51.11; H, 5.51; N, 14.19; O, 22.69; S, 6.50 |
| CAS # | 63358-49-6 |
| Related CAS # | 63358-49-6; |
| PubChem CID | 71961 |
| Appearance | Solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 985.1±65.0 °C at 760 mmHg |
| Melting Point | 195-198° (dec) |
| Flash Point | 549.5±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.672 |
| LogP | -0.53 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 34 |
| Complexity | 861 |
| Defined Atom Stereocenter Count | 5 |
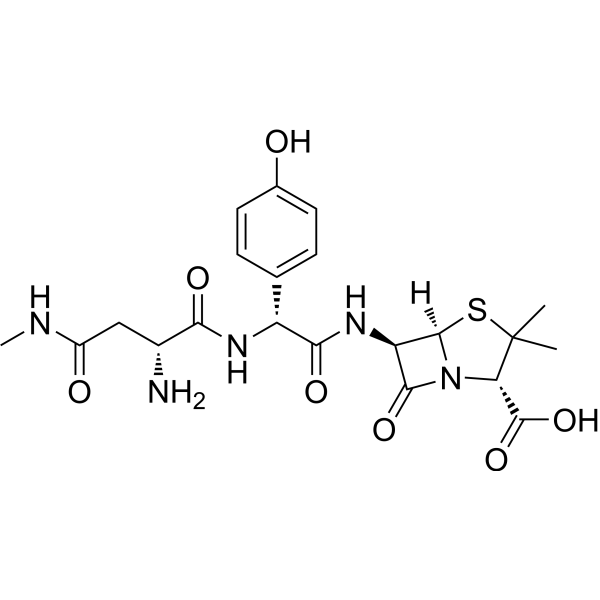
| SMILES | CC1([C@@H](N2C([C@@H](N(C([C@H](NC)CC(N)=O)=O)[C@@H](C(N)=O)C3=CC=C(O)C=C3)[C@H]2S1)=O)C(O)=O)C |
| InChi Key | BHELIUBJHYAEDK-OAIUPTLZSA-N |
| InChi Code | InChI=1S/C21H27N5O7S/c1-21(2)15(20(32)33)26-18(31)14(19(26)34-21)25-17(30)13(9-4-6-10(27)7-5-9)24-16(29)11(22)8-12(28)23-3/h4-7,11,13-15,19,27H,8,22H2,1-3H3,(H,23,28)(H,24,29)(H,25,30)(H,32,33)/t11-,13-,14-,15+,19-/m1/s1 |
| Chemical Name | (2S,5R,6R)-6-[[(2R)-2-[[(2R)-2-amino-4-(methylamino)-4-oxobutanoyl]amino]-2-(4-hydroxyphenyl)acetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| Synonyms | Aspoxicillin; TA 058; TA058; TA-058 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Aspoxicillin is a semi-synthetic derivative of penicillin [2]. In vitro, Aspoxicillin has a 1.7-hour post-antibiotic effect (PAE) on Staphylococcus aureus Smith. |
| ln Vivo | For 5.2 hours, apocillin produced PAE against S. aureus Smith in vivo in a thigh infection model in neutropenic mice [2]. After intravenously injecting hydroxychloroquine (5 mg/kg) into BALB/c mice, the amount of chlorotoquine (desethylhydroxychloroquine) in the mice's blood and tissues was measured. Tissue-to-blood concentration ratio (Kp) ≥1 signifies tissue accumulation of chlorotoquine. From high to low, the following tissues had the highest chlorotoquine Kp ratios: liver (114.3), kidney (24.4), spleen (19.3), lung (16.5), heart (5.5) [3]. |
| References |
[1]. Yoshimura H, et al. Comparative in vitro activity of 16 antimicrobial agents against Actinobacillus pleuropneumoniae. Vet Res Commun. 2002 Jan;26(1):11-9. [2]. Oshida T, et al. Activity of sub-minimal inhibitory concentrations of aspoxicillin in prolonging the postantibiotic effect against Staphylococcus aureus. J Antimicrob Chemother. 1990 Jul;26(1):29-38. |
| Additional Infomation |
Aspoxicillin is a peptide. Aspoxicillin is a broad-spectrum, semisynthetic penicillin derivative with antibacterial activity. Aspoxicillin binds to and inactivates penicillin-binding proteins (PBPs) located on the inner membrane of the bacterial cell wall. Inactivation of PBPs interferes with the cross-linkage of peptidoglycan chains necessary for bacterial cell wall strength and rigidity. This interrupts bacterial cell wall synthesis and results in the weakening of the bacterial cell wall, eventually causing cell lysis. |
Solubility Data
| Solubility (In Vitro) | H2O : ~25 mg/mL (~50.66 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 12.5 mg/mL (25.33 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0262 mL | 10.1311 mL | 20.2622 mL | |
| 5 mM | 0.4052 mL | 2.0262 mL | 4.0524 mL | |
| 10 mM | 0.2026 mL | 1.0131 mL | 2.0262 mL |