Physicochemical Properties
| Molecular Formula | C17H11NO8 |
| Molecular Weight | 357.2711 |
| Exact Mass | 357.048 |
| CAS # | 17413-38-6 |
| PubChem CID | 161218 |
| Appearance | Yellow to orange solid powder |
| Density | 1.656 g/cm3 |
| Boiling Point | 689.5ºC at 760 mmHg |
| Melting Point | 262-263 °C |
| Flash Point | 370.8ºC |
| Vapour Pressure | 5.83E-20mmHg at 25°C |
| Index of Refraction | 1.776 |
| LogP | 3.565 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 26 |
| Complexity | 580 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | PADIFGYTAXNCRK-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H11NO8/c1-24-12-3-7(19)2-9-8(12)4-11(18(22)23)14-10(17(20)21)5-13-16(15(9)14)26-6-25-13/h2-5,19H,6H2,1H3,(H,20,21) |
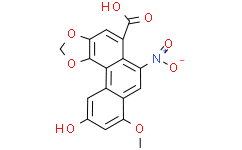
| Chemical Name | 10-hydroxy-8-methoxy-6-nitronaphtho[2,1-g][1,3]benzodioxole-5-carboxylic acid |
| Synonyms | Aristolochic acid D; Aristolochic acid d; 17413-38-6; Aristolochic acid-D; Aristolochic acid Iva; aristolochic D; Phenanthro[3,4-d]-1,3-dioxole-5-carboxylic acid, 10-hydroxy-8-methoxy-6-nitro-; 10-hydroxy-8-methoxy-6-nitrophenanthro[3,4-d][1,3]dioxole-5-carboxylic acid; CHEMBL604748; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural product |
| ln Vitro |
Aristolochic acid (AA) I was found to be most toxic followed by AA II, AA VIIIa, and AA Ia in decreasing levels of toxicity. The other compounds, nitrophenanthrene carboxylic acid analogues of AA I, aristolactams, and other derivatives did not exhibit considerable toxicity. The results showed significant relationships between cytotoxicity of AA compounds and the localization of functional groups in their structure. Analogues containing hydroxyl groups diminished cytotoxicity. The demethylated analogues of AA I are markedly less active. The negative impact on cytotoxicity was found on nitroreduction of AA I. AA induced caspase activation was also observed.
Conclusion: These cytotoxic data suggest that the nitro and methoxy groups are critical determinants of nephrotoxicologic potency of AA [2]. The nitrophenanthrene carboxylic acid derivative, Aristolochic acid D, was tested for cytotoxicity against the porcine renal proximal tubular cell line LLC-PK1. At concentrations up to 300 μmol/L, Aristolochic acid D did not exhibit considerable toxicity to the LLC-PK1 cells, as evidenced by the neutral red dye exclusion assay. Its IC50 was >300 μmol/L and it was categorized as non-toxic in this assay.[2] In a caspase 3/7 activity assay designed to investigate apoptotic mechanisms, Aristolochic acid D at a concentration of 300 μmol/L was not able to induce caspase 3/7 activity significantly in LLC-PK1 cells.[2] |
| Cell Assay |
A series of Aristolochic acid (AA) derivatives isolated from Aristolochia spp. were analyzed for their nephrotoxic potential using the neutral red dye exclusion assay in cultures of LLC-PK(1) cells. The structural relationships between AA I and its analogues were compared with their cytotoxic effects to predict structural determinants for AA toxicity. Further, caspase-3 assay was performed on toxic compounds to determine if caspases, the enzymes that play a critical role in apoptosis are involved in AA-induced cytotoxicity [2]. Cytotoxicity Assay (Neutral Red Uptake): LLC-PK1 cells were seeded in 96-well plates and allowed to attach for 24 hours. The cells were then treated with graded concentrations of Aristolochic acid D (or other test compounds) for 48 hours. After treatment, the medium was replaced with serum-free medium containing neutral red dye and incubated for 90 minutes to allow uptake by viable cells. The cells were then washed, and the incorporated dye was extracted using an acidified isopropanol solution. The absorbance of the extracted dye was measured at 540 nm, which correlates with cell viability. Percent viability was calculated relative to solvent-treated control wells.[2] Caspase 3/7 Activity Assay: LLC-PK1 cells were seeded in white-walled 96-well plates. After 24 hours, cells were treated with Aristolochic acid D (300 μmol/L) for 48 hours. A luminescent assay reagent containing a caspase-3/7 substrate (DEVD sequence) was added to lyse the cells and initiate the reaction. Caspase activity cleaves the substrate, generating a luminescent signal proportional to caspase activity, which was measured using a luminometer.[2] |
| Toxicity/Toxicokinetics |
Aristolochic acid D was found to be non-toxic to the renal epithelial cell line LLC-PK1, with an IC50 value greater than 300 μmol/L in the neutral red cytotoxicity assay.[2] It was mentioned in the discussion that Aristolochic acid D (referred alongside aristolic acid) had been reported as a less toxic compound against P388 cell lines and Salmonella strains, and it was also found to be non-toxic to LLC-PK1 cells in the present study.[2] |
| References |
[1]. The isolation and structural elucidation of novel derivatives of aristolochic acid from Aristolochia indica. J Org Chem. 1968 Oct;33(10):3735-8. [2]. Structure activity relationships of aristolochic acid analogues: toxicity in cultured renal epithelial cells. Kidney Int. 2005 May;67(5):1797-805. |
| Additional Infomation |
Aristolochic acid D is an aristolochic acid that is phenanthrene-1-carboxylic acid substituted by a methylenedioxy group at the 3,4 positions, by an hydroxy group at position 6, by a methoxy group at position 8, and by a nitro group at position 10. It has a role as a carcinogenic agent, a metabolite, a nephrotoxin and a toxin. It is a C-nitro compound, a member of aristolochic acids, an aromatic ether, a cyclic acetal, a monocarboxylic acid and an organic heterotetracyclic compound. Aristolochic acid d has been reported in Aristolochia mollissima, Aristolochia kaempferi, and Aristolochia heterophylla with data available. Aristolochic acid D is a novel derivative of aristolochic acid isolated from the roots of Aristolochia indica L. during a search for plant-derived tumor inhibitors. The plant extract showed tumor inhibitory activity against the adenocarcinoma 755 test system, with aristolochic acid identified as the principal active principle. [1] The compound was isolated as deep wine-red crystals with a melting point of 269-272 °C. Its molecular formula was determined to be C17H11NO8 based on elemental analysis. [1] The structure was elucidated as 3,4-methylenedioxy-6-hydroxy-8-methoxy-10-nitro-1-phenanthroic acid (4) through a series of chemical transformations and spectral analyses. Methylation with diazomethane yielded a dimethyl derivative (5). Hydrogenation of this derivative yielded aristolochic acid-D methyl ether lactam (6), which was identical to a naturally occurring compound. [1] Spectroscopic evidence, including UV, IR, and NMR, supported the proposed structure and helped differentiate the methoxy group position (C-8) from the alternative (C-6). [1] Aristolochic acid D is one of the minor nitrophenanthrene carboxylic acid derivatives present in Aristolochia plants.[2] Structurally, Aristolochic acid D is an analogue of Aristolochic acid I (AA I). The addition of a hydroxyl group at the R5 position of the AA I structure (forming AA-D) was reported to eliminate the toxic nature of the compound.[2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~69.98 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7990 mL | 13.9950 mL | 27.9900 mL | |
| 5 mM | 0.5598 mL | 2.7990 mL | 5.5980 mL | |
| 10 mM | 0.2799 mL | 1.3995 mL | 2.7990 mL |