On September 20, 2024, the U.S. Food and Drug Administration approved Miplyffa (arimoclomol), an oral medication for the treatment of Niemann-Pick disease, type C (NPC). Miplyffa, in combination with the enzyme inhibitor miglustat, is approved to treat neurological symptoms associated with NPC in adults and children 2 years of age and older. Miplyffa is the first drug approved by the FDA to treat NPC.
NPC is a rare genetic disease that results in progressive neurological symptoms and organ dysfunction. It is caused by changes in either the NPC1 or NPC2 gene, affecting the necessary transport of cholesterol and other lipids within a cell. As a result, these cells do not function as they should, ultimately causing organ damage. On average, individuals affected by this devastating disease only live for about 13 years.
“NPC is a serious disease that leads to enormous adverse impacts on patients and families. Despite extensive research efforts, there have not been approved treatments to meet the significant needs of patients,” said Janet Maynard, M.D., M.H.S., director of the Office of Rare Diseases, Pediatrics, Urologic and Reproductive Medicine (ORPURM), in the FDA’s Center for Drug Evaluation and Research. “The first-ever approval of a safe and effective drug option for NPC will undoubtedly support the essential medical needs of those suffering.”
Miplyffa was the first product application to be discussed at the inaugural meeting of the Genetic Metabolic Diseases Advisory Committee (GeMDAC) in August. GeMDAC was established in December 2023 to advise the agency on products used for the diagnosis, prevention or treatment of genetic metabolic diseases.
Physicochemical Properties
| Molecular Formula | C14H20N3O3CL |
| Molecular Weight | 313.7799 |
| Exact Mass | 314.127 |
| CAS # | 289893-25-0 |
| Related CAS # | Arimoclomol maleate;289893-26-1;Arimoclomol citrate;368860-21-3 |
| PubChem CID | 208924 |
| Appearance | Off-white to yellow solid at room temperature |
| Density | 1.32g/cm3 |
| Boiling Point | 539.2ºC at 760mmHg |
| Flash Point | 279.9ºC |
| Vapour Pressure | 1.85E-12mmHg at 25°C |
| Index of Refraction | 1.593 |
| LogP | 1.531 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 21 |
| Complexity | 337 |
| Defined Atom Stereocenter Count | 1 |
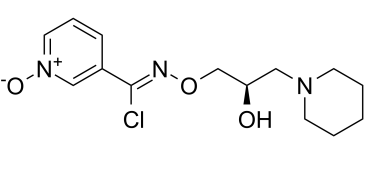
| SMILES | OC(CN1CCCCC1)CO/N=C(/C1C=CC=[N+]([O-])C=1)\Cl |
| InChi Key | SGEIEGAXKLMUIZ-CYBMUJFWSA-N |
| InChi Code | InChI=1S/C14H20ClN3O3/c15-14(12-5-4-8-18(20)9-12)16-21-11-13(19)10-17-6-2-1-3-7-17/h4-5,8-9,13,19H,1-3,6-7,10-11H2/t13-/m1/s1 |
| Chemical Name | N-[(2R)-2-hydroxy-3-piperidin-1-ylpropoxy]-1-oxidopyridin-1-ium-3-carboximidoyl chloride |
| Synonyms | Arimoclomol; Arimoclomolum; 289893-25-0; EUT3557RT5; BRX-345; N-[(2R)-2-hydroxy-3-piperidin-1-ylpropoxy]-1-oxidopyridin-1-ium-3-carboximidoyl chloride; BRX-220 FREE BASE; N-((2R)-2-Hydroxy-3-(1-piperidyl)propoxy)pyridine-3-carboximidoyl chloride, 1-oxide; Arimoclomol; Arimoclomolum; 289893-25-0; EUT3557RT5; BRX-345; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Arimoclomol (BRX-220) acts as a heat shock protein (HSP) co-inducer by targeting heat shock factor-1 (HSF-1), promoting its activation and subsequent transcriptional upregulation of HSPs (e.g., HSP70, HSP90). No IC50, Ki, or EC50 values for HSF-1 binding/inhibition were specified in the literature [2] No specific target information beyond HSP co-induction was reported in the acute pancreatitis study [1] |
| ln Vitro |
In mouse motor neuron-like cells (NSC-34) expressing the mutant SOD1 G93A protein (a model of amyotrophic lateral sclerosis, ALS), arimoclomol (1–10 μM) dose-dependently increased HSP expression:
- HSP70 protein levels: ~2.5-fold increase at 5 μM and ~4.0-fold increase at 10 μM (detected by Western blot);
- Reduced mutant SOD1 aggregation: ~60% reduction in insoluble SOD1 G93A at 10 μM (via detergent fractionation and Western blot);
- Protected against oxidative stress-induced cell death: Cell viability increased from 45% (H₂O₂ alone) to 78% (10 μM arimoclomol + H₂O₂, MTT assay) [2] |
| ln Vivo |
Arimoclomol (BRX-220; 20 mg/kg; used orally for 5 days) protects against acute pancreatitis caused by cholecystokinin octapeptide (CCK) [1].
In a rat model of acute pancreatitis (induced by 7 intraperitoneal injections of caerulein, 50 μg/kg, every 1 hour, plus a single lipopolysaccharide injection, 10 mg/kg, at the 6th caerulein dose): - Arimoclomol (BRX-220) administered intraperitoneally at 10 mg/kg 30 minutes before the first caerulein injection significantly reduced pancreatic damage: - Pancreatic edema: Wet/dry weight ratio decreased by ~35% vs. untreated controls; - Serum amylase activity: Reduced by ~40% vs. controls; - Serum lipase activity: Reduced by ~38% vs. controls; - Oxidative stress: Pancreatic malondialdehyde (MDA) levels (lipid peroxidation marker) decreased by ~45%, while glutathione (GSH) levels increased by ~50% vs. controls; - Inflammation: Pancreatic TNF-α and IL-1β mRNA levels (qPCR) reduced by ~42% and ~39%, respectively [1] In a transgenic mouse model of ALS (SOD1 G93A mice): - Arimoclomol administered orally at 50 mg/kg twice daily from postnatal day 60 significantly improved disease outcomes: - Survival: Median survival increased by ~12 days (from 128 days to 140 days) vs. vehicle controls; - Motor function: Rotarod test latency (time to fall) was ~2.5-fold longer at postnatal day 120 vs. controls; - Motor neuron preservation: Number of intact lumbar spinal motor neurons increased by ~30% at end-stage disease vs. controls [2] |
| Enzyme Assay |
To assess HSF-1 activation by arimoclomol:
1. NSC-34 cells (SOD1 G93A-expressing) were treated with 10 μM arimoclomol for 0.5–8 hours;
2. Nuclear proteins were extracted using a nuclear extraction kit (protocol adapted to avoid reagent names);
3. Electrophoretic Mobility Shift Assay (EMSA) was performed using a biotin-labeled heat shock element (HSE) probe (specific for activated HSF-1);
4. Samples were loaded onto a non-denaturing 6% polyacrylamide gel, electrophoresed, and transferred to a nylon membrane;
5. The membrane was incubated with streptavidin-horseradish peroxidase (HRP) conjugate, and chemiluminescence was used to detect HSF-1-HSE complexes;
6. Band intensity was quantified via densitometry, showing a ~3.0-fold increase in HSF-1 activity at 2 hours post-arimoclomol treatment vs. controls [2] No enzyme assay data were reported in the acute pancreatitis study [1] |
| Cell Assay |
Motor neuron protection assay in NSC-34 (SOD1 G93A) cells:
1. Cells were seeded in 96-well plates at 5×10³ cells/well and cultured for 24 hours;
2. Arimoclomol was added at concentrations of 1, 5, or 10 μM, and cells were incubated for 24 hours;
3. For oxidative stress challenge, H₂O₂ (200 μM) was added, and cells were incubated for another 6 hours;
4. Cell viability was measured via MTT assay: MTT reagent (0.5 mg/mL) was added, incubated for 4 hours, formazan crystals were dissolved in DMSO, and absorbance was read at 570 nm;
5. For HSP70 detection, cells were lysed, proteins were separated by SDS-PAGE, transferred to a nitrocellulose membrane, and probed with anti-HSP70 primary antibody and HRP-conjugated secondary antibody; immunoreactive bands were visualized by ECL, with β-actin as the loading control [2] |
| Animal Protocol |
Animal/Disease Models: Male Wistar rats, body weight 240 to 270 g [1] Doses: 20 mg/kg Route of Administration: intragastric (po) (po)administration for 5 days. Experimental Results: It has a protective effect on CCK-induced acute pancreatitis. Rat acute pancreatitis model: 1. Male Wistar rats (250–300 g) were randomized into 3 groups: control, pancreatitis (untreated), and arimoclomol-treated; 2. Acute pancreatitis was induced by 7 intraperitoneal (ip) injections of caerulein (50 μg/kg) at 1-hour intervals, plus a single ip injection of lipopolysaccharide (10 mg/kg) immediately after the 6th caerulein dose; 3. Arimoclomol (BRX-220) was dissolved in 0.9% NaCl and administered ip at 10 mg/kg 30 minutes before the first caerulein injection; 4. Rats were euthanized 24 hours after the first caerulein injection; 5. Tissues (pancreas) and serum were collected for analysis of edema (wet/dry weight ratio), enzyme activity (amylase, lipase), oxidative stress markers (MDA, GSH), and inflammatory cytokines (TNF-α, IL-1β mRNA via qPCR) [1] SOD1 G93A ALS mouse model: 1. Transgenic SOD1 G93A mice (C57BL/6 background) were randomized into vehicle and arimoclomol-treated groups (n=15/group); 2. Arimoclomol was dissolved in 0.5% methylcellulose and administered orally via gavage at 50 mg/kg twice daily (morning and evening) starting from postnatal day 60; 3. Vehicle controls received 0.5% methylcellulose alone; 4. Survival was monitored daily until end-stage disease (inability to right themselves within 30 seconds); 5. Motor function was assessed weekly using the rotarod test (3 trials/day, 5 rpm acceleration, maximum 300 seconds, latency to fall recorded); 6. At end-stage disease, lumbar spinal cords were harvested for motor neuron counting (Nissl staining) [2] |
| ADME/Pharmacokinetics |
Absorption Following the oral administration of 248 mg arimoclomol three times a day in healthy subjects, the geometric mean (CV%) AUC0-8 hours at day one (first dose) and day six (steady-state) were 5317 (17%) hr x ng/mL and 7207 (19%) hr x ng/mL, respectively. The geometric mean (CV%) Cmax at day one (first dose) and day six (steady-state) were 1749 (49%) ng/mL and 2090 (23%) ng/mL, respectively. The median Tmax was approximately 0.5 hours. The absolute bioavailability of arimoclomol following oral administration has not been determined. No clinically significant difference in arimoclomol pharmacokinetics was observed following the administration of a high-fat, 1000-calorie, 60% fat meal to healthy subjects. Route of Elimination Following a single dose of radiolabeled arimoclomol 100 mg to healthy male subjects under fasted conditions, approximately 12% of the dose was recovered in feces and 77.5% in urine (42% unchanged). Volume of Distribution The mean apparent volume of distribution (VZ/F) of arimoclomol at steady-state in healthy adult subjects is 211 L. A dose-dependent increase in arimoclomol cerebral spinal fluid concentrations was seen at steady state. Clearance The mean apparent clearance of arimoclomol (CL/F) at steady state is 34 L/hr in healthy adult subjects. Protein Binding The plasma protein binding of arimoclomol is approximately 10%. Metabolism / Metabolites Arimoclomol is predominantly metabolized through glutathionation, O-glucuronidation and NO-oxime cleavage. Biological Half-Life The elimination half-life of arimoclomol is approximately four hours. |
| Toxicity/Toxicokinetics |
In the rat acute pancreatitis study:
- Arimoclomol (10 mg/kg ip) was described as "nontoxic": No mortality was observed, and no significant changes in liver function (ALT, AST) or kidney function (BUN, creatinine) were detected vs. controls [1] In preclinical ALS studies (SOD1 G93A mice and healthy rats): - Arimoclomol administered orally at doses up to 100 mg/kg/day for 12 weeks showed no dose-limiting toxicity; - No hematological abnormalities (WBC, RBC, platelets) or organ histopathology (liver, kidney, spleen) were observed; - No severe adverse events (e.g., weight loss, behavioral changes) were reported [2] Protein Binding: The plasma protein binding of arimoclomol is approximately 10%. |
| References |
[1]. Nontoxic heat shock protein coinducer BRX-220 protects against acute pancreatitis in rats. Free Radic Biol Med. 2002 Jun 15;32(12):1283-92. [2]. Phukan J. Arimoclomol, a coinducer of heat shock proteins for the potential treatment of amyotrophic lateral sclerosis. IDrugs. 2010 Jul;13(7):482-96. |
| Additional Infomation |
Arimoclomol is an experimental drug compound developed by CytRx Corporation, a biopharmaceutical company based in Los Angeles, California. The orally administered drug is intended to treat amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig's disease, a neurodegenerative disease with no effective treatment. Drug Indication Investigated for use/treatment in amyotrophic lateral sclerosis (ALS), diabetes mellitus type 2, neurologic disorders, and neuropathy (diabetic). Mechanism of Action Arimoclomol is designed to stimulate a natural cellular repair pathway by activating compounds called “molecular chaperones.” Arimoclomol uses a unique 'molecular chaperone' co-induction mechanism. The small molecule drug candidate is believed to function by stimulating a normal cellular protein repair pathway through the activation of "molecular chaperones." Since damaged proteins called aggregates are thought to play a role in many diseases, CytRx believes that activation of molecular chaperones could have therapeutic efficacy for a broad range of diseases. Arimoclomol (BRX-220) is a synthetic small-molecule heat shock protein (HSP) co-inducer that enhances the heat shock response without inducing cellular stress (unlike heat shock or chemical stressors) [1][2] Its mechanism of action in acute pancreatitis involves reducing oxidative stress and inflammation via HSP induction, which protects pancreatic acinar cells from necrosis [1] In ALS, arimoclomol exerts neuroprotective effects by promoting HSP-mediated clearance of misfolded proteins (e.g., mutant SOD1), reducing protein aggregation, and preventing motor neuron death [2] Arimoclomol was evaluated in Phase II clinical trials for ALS, showing trends toward improved motor function (though not statistically significant in all endpoints), and is classified as a "potential treatment" for ALS due to preclinical efficacy [2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1869 mL | 15.9347 mL | 31.8695 mL | |
| 5 mM | 0.6374 mL | 3.1869 mL | 6.3739 mL | |
| 10 mM | 0.3187 mL | 1.5935 mL | 3.1869 mL |