Physicochemical Properties
| Molecular Formula | C7H11NO2.HCL |
| Molecular Weight | 177.62868 |
| Exact Mass | 177.056 |
| CAS # | 6018-28-6 |
| Related CAS # | Arecaidine;499-04-7;Arecaidine hydrobromide;6013-57-6 |
| PubChem CID | 12305194 |
| Appearance | White to light yellow solid powder |
| Boiling Point | 266.7ºC at 760 mmHg |
| Melting Point | 260ºC |
| Flash Point | 115.1ºC |
| LogP | 1.072 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 11 |
| Complexity | 174 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | PIVDNPNYIBGXPL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C7H11NO2.ClH/c1-8-4-2-3-6(5-8)7(9)10;/h3H,2,4-5H2,1H3,(H,9,10);1H |
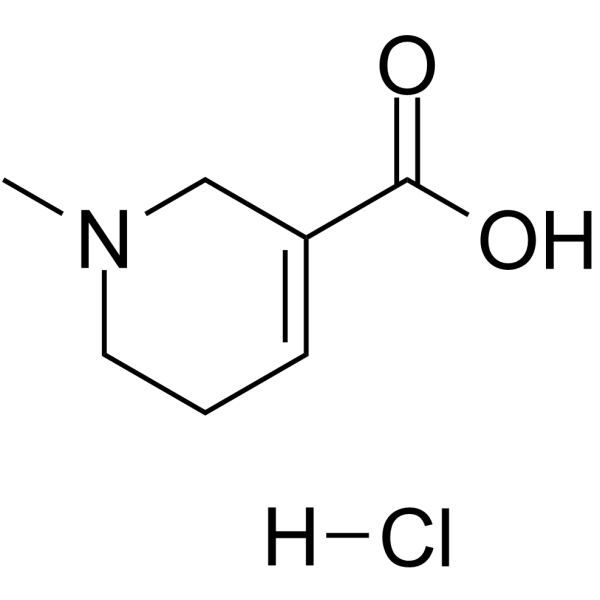
| Chemical Name | 1-methyl-3,6-dihydro-2H-pyridine-5-carboxylic acid;hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- γ-Aminobutyric acid (GABA) receptors or GABAergic system [1] - Human proton-coupled amino acid transporter 1 (hPAT1) (served as a substrate of hPAT1; pH-dependent transport activity was confirmed) [2] |
| ln Vitro |
- Experiments were performed on synaptosomal preparations or brain slices isolated from cats to investigate the effect of Arecaidine on GABA-mediated inhibitory responses in the central nervous system. When Arecaidine was applied together with GABA, it was observed to antagonize the inhibitory postsynaptic potentials (IPSPs) induced by GABA. Specifically, Arecaidine reduced both the amplitude and duration of GABA-evoked IPSPs in neurons from cat central nervous system regions (e.g., cerebral cortex and thalamus). This result suggested that Arecaidine could interfere with GABAergic inhibitory neurotransmission. [1] - In HEK293 cells transfected with the human hPAT1 gene (empty vector-transfected cells as control), the transport of Arecaidine was evaluated using radiolabeled tracers. The results showed that Arecaidine was actively transported by hPAT1, and this transport exhibited pH dependence—significantly higher uptake of Arecaidine was detected under acidic conditions (mimicking the environment of the intestinal lumen) compared to neutral conditions. Additionally, the uptake of Arecaidine by hPAT1-transfected cells was competitively inhibited by excess unlabeled hPAT1 substrates (such as GABA and L-proline), which confirmed that the transport of Arecaidine was mediated specifically by hPAT1. [2] |
| ln Vivo |
- Adult cats were used as experimental animals. After anesthesia, intracranial microelectrodes were implanted into specific brain regions (e.g., cerebral cortex and thalamus) of the cats to record extracellular electrical activities of neurons. Arecaidine was dissolved in artificial cerebrospinal fluid and administered via intracerebral microinjection near the recorded neurons. After administration of Arecaidine, the inhibitory effect of exogenously applied GABA on neuronal firing was weakened—manifested as an increase in the discharge frequency of neurons compared to the group treated with GABA alone. The antagonistic effect of Arecaidine lasted for approximately 15-30 minutes, and the neuronal electrical activity gradually recovered thereafter. [1] |
| Enzyme Assay |
- GABA receptor binding assay: Brain homogenates were prepared from the cerebral cortex of cats, serving as the source of GABA receptors. Radiolabeled [³H]-GABA (as a ligand) was mixed with the brain homogenates and different concentrations of Arecaidine in binding buffer. The mixture was incubated at 4°C for a certain period, then filtered through glass fiber filters to separate the bound [³H]-GABA from the free form. The radioactivity of the filters (representing bound [³H]-GABA) was measured using a liquid scintillation counter. The results indicated that Arecaidine could compete with [³H]-GABA for binding to GABA receptors, as it reduced the specific binding of [³H]-GABA in a concentration-dependent manner, confirming the interaction between Arecaidine and GABA receptors. [1] - hPAT1-mediated transport assay: HEK293 cells were transfected with hPAT1 expression plasmids (or empty vectors as controls) and cultured for 48 hours. The cells were harvested and resuspended in HBSS buffer adjusted to different pH values (acidic and neutral). Radiolabeled Arecaidine was added to the cell suspension, and the mixture was incubated at 37°C for different time intervals. The reaction was terminated by adding ice-cold HBSS buffer, followed by centrifugation to collect the cells. The cells were lysed, and the radioactivity in the cell lysate was measured. The protein concentration of the lysate was determined to normalize the uptake amount of Arecaidine. For competition experiments, excess unlabeled hPAT1 substrates (e.g., GABA, L-proline) were co-incubated with radiolabeled Arecaidine to verify the specificity of hPAT1-mediated transport. [2] |
| Cell Assay |
- Primary cat neuron culture assay: Neurons were isolated from the cerebral cortex of neonatal cats and cultured in appropriate neurobasal medium for 7-10 days. The effect of Arecaidine on GABA-induced calcium responses was detected using the calcium-sensitive dye Fura-2 AM. The neurons were loaded with Fura-2 AM for 30 minutes, then treated with GABA alone or GABA combined with Arecaidine. The fluorescence intensity of Fura-2 AM (excitation at 340 nm and 380 nm, emission at 510 nm) was recorded using a fluorescence microscope. It was found that Arecaidine decreased the GABA-induced calcium influx in neurons, which was consistent with its GABA receptor antagonistic effect. [1] - hPAT1-expressing HEK293 cell uptake assay: HEK293 cells transfected with hPAT1 were seeded into 24-well plates and cultured until reaching 80% confluence. Before the experiment, the cells were washed twice with HBSS buffer of different pH values. Radiolabeled Arecaidine in HBSS buffer was added to each well, and the cells were incubated at 37°C for a specific time. The incubation was stopped by aspirating the medium and washing the cells with ice-cold HBSS buffer. The cells were lysed with NaOH, and the radioactivity in the lysate was counted. The results showed that the uptake of radiolabeled Arecaidine by hPAT1-transfected cells was significantly higher than that by empty vector-transfected cells, and this uptake was markedly inhibited by co-incubation with unlabeled GABA (a known hPAT1 substrate), confirming the role of hPAT1 in Arecaidine transport. [2] |
| Animal Protocol |
- Adult cats (weight not specified) were anesthetized with pentobarbital sodium (induction via intraperitoneal injection, maintenance via continuous intravenous infusion). The anesthetized cats were fixed in a stereotaxic frame, and a craniotomy was performed to expose the target brain regions (cerebral cortex or thalamus). Glass microelectrodes filled with 3 M NaCl were inserted into the brain tissue to record extracellular potentials of single neurons. Arecaidine was dissolved in artificial cerebrospinal fluid (composition: 126 mM NaCl, 2.5 mM KCl, 1.2 mM MgCl₂, 1.2 mM CaCl₂, 1.0 mM NaH₂PO₄, 26 mM NaHCO₃, 10 mM glucose) to prepare solutions of different concentrations. Microinjections of Arecaidine (volume: 0.5-1 μL) were delivered to the area near the recorded neuron using a microsyringe. The electrical activity of the neuron was recorded continuously for 60 minutes (15 minutes before injection and 45 minutes after injection) to monitor the effect of Arecaidine on GABA-induced inhibition. After the experiment, the cats were euthanized with an overdose of pentobarbital sodium. [1] |
| ADME/Pharmacokinetics |
- Arecaidine was identified as a substrate of hPAT1, which is highly expressed in the apical membrane of intestinal epithelial cells. This finding suggests that Arecaidine may be absorbed from the gastrointestinal tract via hPAT1-mediated transport, especially in the acidic environment of the stomach and upper small intestine (where the proton gradient drives hPAT1 activity). However, no in vivo ADME parameters (e.g., absorption rate, bioavailability) of Arecaidine were determined. [2] |
| References |
[1]. Effects of the Areca nut constituents arecaidine and guvacine on the action of GABA in the cat central nervous system. Brain Res. 1977 Nov 18;136(3):513-22. [2]. Transport of the areca nut alkaloid arecaidine by the human proton-coupled amino acid transporter 1 (hPAT1). J Pharm Pharmacol. 2013 Apr;65(4):582-90. |
| Additional Infomation |
- Arecaidine is a naturally occurring pyridine alkaloid isolated from Areca nut (the fruit of Areca catechu L.), a common component of betel quids. This study focused on its effect on the central nervous system GABAergic system—GABA is the major inhibitory neurotransmitter in the mammalian central nervous system. The results demonstrated that Arecaidine acts as a GABA receptor antagonist, which may contribute to the stimulant effects (e.g., increased alertness, euphoria) associated with Areca nut consumption by reducing GABA-mediated central inhibition. [1] - Human hPAT1 (SLC36A1) belongs to the solute carrier 36 family, which mediates the uptake of small neutral amino acids, imino acids, and certain neurotransmitters (e.g., GABA) in a proton-coupled, electrogenic manner. In addition to intestinal absorption, hPAT1 is also expressed in renal proximal tubules, where it may participate in the reabsorption of filtered Arecaidine. The identification of Arecaidine as a hPAT1 substrate provides insights into the molecular mechanism of its absorption and disposition in humans, and also implies potential drug-drug interactions if Arecaidine is co-administered with other hPAT1 substrates (e.g., certain amino acid supplements, GABA analogs). [2] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~100 mg/mL (~562.97 mM) DMSO : ~25 mg/mL (~140.74 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (14.07 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (14.07 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (14.07 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.6297 mL | 28.1484 mL | 56.2968 mL | |
| 5 mM | 1.1259 mL | 5.6297 mL | 11.2594 mL | |
| 10 mM | 0.5630 mL | 2.8148 mL | 5.6297 mL |