Physicochemical Properties
| Molecular Formula | C25H27NO7 |
| Molecular Weight | 453.48 |
| Exact Mass | 453.178 |
| CAS # | 2293951-01-4 |
| PubChem CID | 155521683 |
| Appearance | White to yellow solid powder |
| LogP | 3.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 33 |
| Complexity | 722 |
| Defined Atom Stereocenter Count | 0 |
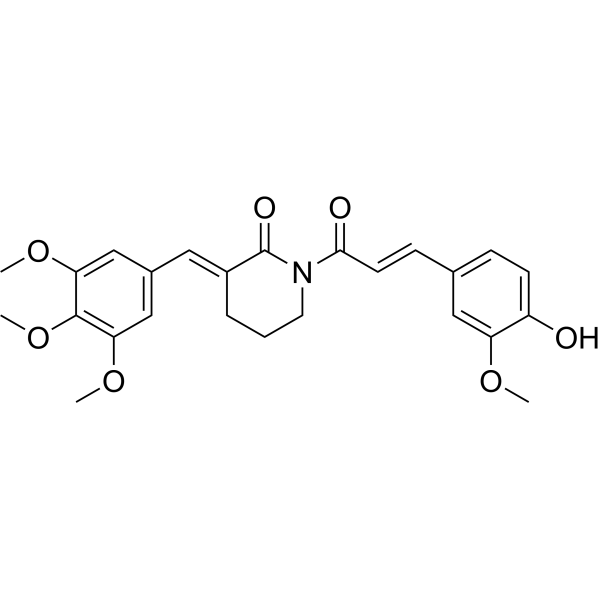
| SMILES | COC1=CC(=CC(=C1OC)OC)/C=C/2\CCCN(C2=O)C(=O)/C=C/C3=CC(=C(C=C3)O)OC |
| InChi Key | XQUXWWVMQPNGGF-KWKAUDIHSA-N |
| InChi Code | InChI=1S/C25H27NO7/c1-30-20-13-16(7-9-19(20)27)8-10-23(28)26-11-5-6-18(25(26)29)12-17-14-21(31-2)24(33-4)22(15-17)32-3/h7-10,12-15,27H,5-6,11H2,1-4H3/b10-8+,18-12+ |
| Chemical Name | (3E)-1-[(E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoyl]-3-[(3,4,5-trimethoxyphenyl)methylidene]piperidin-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Anti-inflammatory agent 36 (Compound 5a28) has an IC50 of 3.69 μM for TNF-α and 3.68 μM for IL-6, which inhibits the release of both cytokines from activated RAW 264.7 murine macrophages when exposed to LPS[1]. In RAW 264.7 murine macrophages, LPS-induced P-P38 and P-ERK is inhibited by anti-inflammatory drug 36 (10 μM, 0.5 hours)[1]. Anti-inflammatory drug 36 (10 μM, 0.5 h) suppresses TNF-α, IL-6, IL-1β, ICAM-1, and VCAM-1 transcription that is triggered by LPS [1]. |
| ln Vivo | In a mouse model of acute lung injury, anti-inflammatory drug 36 (compound 5a28) (10 mg/kg, intraperitoneal injection) reduces inflammation[1]. |
| Cell Assay |
Western Blot Analysis[1] Cell Types: RAW 264.7 mouse macrophages Tested Concentrations: 10 μM Incubation Duration: 0.5 h Experimental Results: Markedly inhibited P-P38 and P-ERK, indicating the suppression of MAPK signaling. |
| Animal Protocol |
Animal/Disease Models: Acute lung injury mouse model[1] Doses: 10 mg/kg Route of Administration: ip Experimental Results: decreased wet/dry weight ratio of the mice lungs. decreased biomarkers of lymphocytes and macrophages. Suppresses TNF-α, IL-6, IL -1β, 7 VACM-1 and ICAM-1 levels. |
| References | [1]. Qian J, et al. Design and synthesis novel di-carbonyl analogs of curcumin (DACs) act as potent anti-inflammatory agents against LPS-induced acute lung injury (ALI). Eur J Med Chem. 2019 Apr 1;167:414-425. |
Solubility Data
| Solubility (In Vitro) | DMSO: 25 mg/mL (55.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (2.76 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2052 mL | 11.0258 mL | 22.0517 mL | |
| 5 mM | 0.4410 mL | 2.2052 mL | 4.4103 mL | |
| 10 mM | 0.2205 mL | 1.1026 mL | 2.2052 mL |