Physicochemical Properties
| Molecular Formula | C27H29NO8 |
| Molecular Weight | 495.52 |
| Exact Mass | 495.189 |
| CAS # | 2293951-00-3 |
| PubChem CID | 155549737 |
| Appearance | White to off-white solid powder |
| LogP | 3.7 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 36 |
| Complexity | 826 |
| Defined Atom Stereocenter Count | 0 |
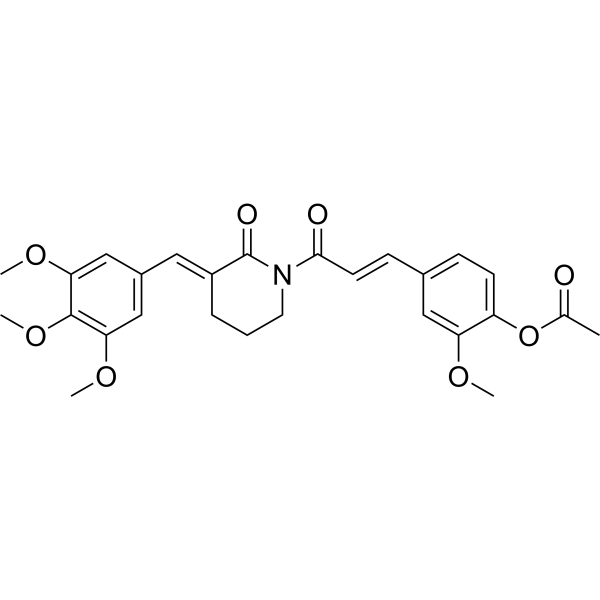
| SMILES | N1(C(=O)/C=C/C2=CC=C(OC(C)=O)C(OC)=C2)CCC/C(=C\C2=CC(OC)=C(OC)C(OC)=C2)/C1=O |
| InChi Key | QLGWATTXCFBRGY-ZEQSIELKSA-N |
| InChi Code | InChI=1S/C27H29NO8/c1-17(29)36-21-10-8-18(14-22(21)32-2)9-11-25(30)28-12-6-7-20(27(28)31)13-19-15-23(33-3)26(35-5)24(16-19)34-4/h8-11,13-16H,6-7,12H2,1-5H3/b11-9+,20-13+ |
| Chemical Name | [2-methoxy-4-[(E)-3-oxo-3-[(3E)-2-oxo-3-[(3,4,5-trimethoxyphenyl)methylidene]piperidin-1-yl]prop-1-enyl]phenyl] acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | p38 MAPK ERK I-kappaBalpha IL-6 p65 NF-κB |
| ln Vitro | In mice, the development of pro-inflammatory cytokines (IL-6, TNF-α) by LPS (0.5 μg/mL; 24 hours) is inhibited by anti-inflammatory drug 35 (compound 5a27) (10 μM; 30 minutes). It is not cytotoxic to produce macrophages. With an IC50 of 2.40 μM and 2.23 μM, respectively, anti-inflammatory drug 35 suppresses the synthesis of TNF-α and IL-6 [1]. RAW 264.7 murine macrophages' activation caused by LPS was greatly reduced by anti-inflammatory drug 35 (10 μM; 30 min). Furthermore, it suppresses IκB levels, p-p38, and p-ERK substantially, which suggests that MAPK and NF-κB signaling are inhibited [1]. |
| ln Vivo | Curcumin (HY-N0005) had a lower bioavailability than anti-inflammatory drug 35 (Compound 5a27) (50 mg/kg; oral; single dose, measured 0–25 minutes) [1]. By reducing inflammation in mice models, anti-inflammatory drug 35 (10 mg/kg; intraperitoneal injection; once daily for 1 week) ameliorates LPS-induced ALI [1]. Rat pharmacokinetic parameters [1]: Route Dose (mg/kg) AUC(0-t) (μg/L·h) AUC(0-∞) (μg/L·h) MRT(0-) (h) t1/2 (h) Tmax (h) CLz/F (L/h/kg) Vz/F (L/kg) Cmax po 50 231.2 325.6 7.8 12.3 6.7 3.3 5062.6 827.1 113.3 iv 5 34.3 122.4 11.3 19.9 0.2 0.1 404.4 59.5 16.4 |
| Cell Assay |
Western Blot Analysis[1] Cell Types: RAW 264.7 mouse macrophages Tested Concentrations: 10 μM Incubation Duration: 30 min; followed by incubation with 0.5 μg/mL LPS for another 30 min Experimental Results: diminished the phosphorylation of p38 and ERK. And down-regulated IκB (inhibitor of NF-κB), inhibits the transcription of TNF-α, IL-6, IL-1β, ICAM-1. |
| Animal Protocol |
Animal/Disease Models: ALI mouse model (C57BL/6 mice)[1] Doses: 10 mg/kg Route of Administration: intraperitoneal (ip)injection, one time/day for 1 week; 30 min later every dose, followed by 5 mg/kg LPS, intratracheal injection Experimental Results: Dramatically normalized the wet/dry ratio of lungs. |
| References |
[1]. Design and synthesis novel di-carbonyl analogs of curcumin (DACs) act as potent anti-inflammatory agents against LPS-induced acute lung injury (ALI). Eur J Med Chem. 2019 Apr 1;167:414-425. |
Solubility Data
| Solubility (In Vitro) | DMSO : 16.67 mg/mL (33.64 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.67 mg/mL (3.37 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 16.7 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0181 mL | 10.0904 mL | 20.1808 mL | |
| 5 mM | 0.4036 mL | 2.0181 mL | 4.0362 mL | |
| 10 mM | 0.2018 mL | 1.0090 mL | 2.0181 mL |