Physicochemical Properties
| Molecular Formula | C16H17N3O4 |
| Molecular Weight | 315.32388 |
| Exact Mass | 315.122 |
| CAS # | 4803-27-4 |
| PubChem CID | 12306674 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.505 g/cm3 |
| Boiling Point | 679.872ºC at 760mmHg |
| Melting Point | 188-194ºC |
| Flash Point | 364.974ºC |
| Index of Refraction | 1.723 |
| LogP | 1.36 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 23 |
| Complexity | 574 |
| Defined Atom Stereocenter Count | 2 |
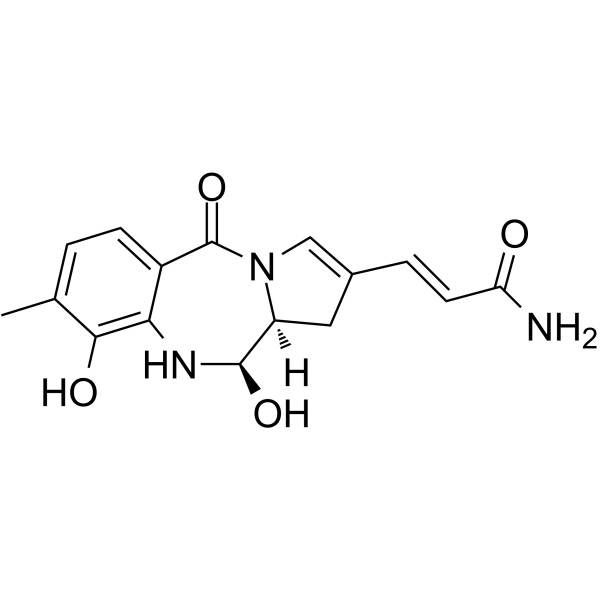
| SMILES | Cc1ccc2c(NC(O)C3CC(\C=C/C(N)=O)=CN3C2=O)c1O |c:16| |
| InChi Key | VGQOVCHZGQWAOI-YQRHFANHSA-N |
| InChi Code | InChI=1S/C16H17N3O4/c1-8-2-4-10-13(14(8)21)18-15(22)11-6-9(3-5-12(17)20)7-19(11)16(10)23/h2-5,7,11,15,18,21-22H,6H2,1H3,(H2,17,20)/b5-3+/t11-,15+/m0/s1 |
| Chemical Name | (E)-3-[(6R,6aS)-4,6-dihydroxy-3-methyl-11-oxo-5,6,6a,7-tetrahydropyrrolo[2,1-c][1,4]benzodiazepin-8-yl]prop-2-enamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Propylene glycol (PG), transcutol P (TC), and propylene glycol monolaurate (PGML) are delivered through the skin by ANT, which also actively "tracks" the penetration of PG and TC into the skin [1]. The ATPase activity of cardiac mitochondria is unaffected by anthramycin doses ranging from 10 to 1000 μM [3]. |
| ln Vivo | Anthramycin (0-0.5 mg/kg, IP, once) possesses significant anti-CCK (cholecystokinin) activity and analgesic effects on the central nervous system of mice [2]. Anthracycline (0.1-0.5 mg/kg, subcutaneous injection, once day for 8 days) has no effect on mitochondrial metabolism in rat hearts [3]. |
| Animal Protocol |
Animal/Disease Models: Male ddY mice (20 ± 2 g, 12-14 per group) [2] Doses: 0, 0.3 and 0.5 mg/kg Route of Administration: IP, once, before intracisternal (ic) injection of CCK 10 minutes: Significant inhibition of CCK-induced increase in pain threshold in a dose-dependent manner. High doses (0.5 mg/kg) almost completely inhibited the antinociceptive effects of CCK. Animal/Disease Models: Female CFN Gif Rat (140-180 g) [3] Doses: 0.1 mg/kg, 0.25 mg/kg and 0.5 mg/kg Route of Administration: subcutaneous injection one time/day for 8 days Experimental Results: Recorded P/O ratio, respiratory rate, and cardiac mitochondrial ATPase activity in anthramycin- and DMSO-treated rats. |
| References |
[1]. Topical delivery of anthramycin II. Influence of binary and ternary solvent systems. Eur J Pharm Sci. 2018 Aug 30;121:59-64. [2]. Cholecystokinin antagonism by anthramycin, a benzodiazepine antibiotic, in the central nervous system in mice. Brain Res. 1989 Apr 17;485(1):62-6. [3]. Effects of daunomycin and anthramycin on electrocardiogram and mitochondrial metabolism of the rat heart. J Natl Cancer Inst. 1974 Aug;53(2):481-6. |
| Additional Infomation |
Anthramycin is a member of the class of pyrrolobenzodiazepines that is (11aS)-5,10,11,11a-tetrahydro-1H-pyrrolo[2,1-c][1,4]benzodiazepine substituted at positions 2, 5, 8, 9 and 11R by a (1E)-3-amino-3-oxoprop-1-en-1-yl, oxo, methyl, hydroxy and hydroxy groups, respectively. It is a natural product isolated from Streptomyces refuineus subsp. thermotolerans. It has a role as a bacterial metabolite, an alkylating agent, a toxin, an antineoplastic agent and an antibacterial agent. It is a pyrrolobenzodiazepine, a member of phenols, a primary amide and an enamide. Anthramycin is a pyrrolo(1,4)benzodiazepine antineoplastic antibiotic isolated from the bacterium Streptomyces refuineus var. thermotolerans. Anthramycin binds covalently to guanine in the minor groove of DNA, thereby inhibiting DNA replication and RNA and protein synthesis. (NCI04) A broad-spectrum spectrum antineoplastic antibiotic isolated from Streptomyces refuineus var. thermotolerans. It has low toxicity, some activity against Trichomonas and Endamoeba, and inhibits RNA and DNA synthesis. It binds irreversibly to DNA. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1714 mL | 15.8569 mL | 31.7138 mL | |
| 5 mM | 0.6343 mL | 3.1714 mL | 6.3428 mL | |
| 10 mM | 0.3171 mL | 1.5857 mL | 3.1714 mL |