Physicochemical Properties
| Molecular Formula | C48H75NO17 |
| Molecular Weight | 938.105600000001 |
| Exact Mass | 937.503 |
| Elemental Analysis | C, 61.46; H, 8.06; N, 1.49; O, 28.99 |
| CAS # | 36148-89-7 |
| Related CAS # | Amphotericin B methyl ester hydrochloride;35375-29-2 |
| PubChem CID | 11968030 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 1102.6±65.0 °C at 760 mmHg |
| Flash Point | 620.6±34.3 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.602 |
| LogP | 1.63 |
| Hydrogen Bond Donor Count | 11 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 66 |
| Complexity | 1680 |
| Defined Atom Stereocenter Count | 19 |
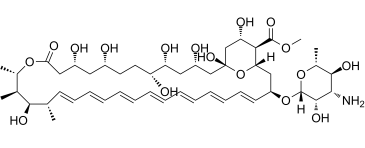
| SMILES | C1=CC=CC=CC=C[C@H](O[C@@H]2O[C@H](C)[C@@H](O)[C@H](N)[C@@H]2O)C[C@H]2[C@@H]([C@H](C[C@@](O2)(O)C[C@@H](O)C[C@@H](O)[C@H](O)CC[C@@H](O)C[C@@H](O)CC(=O)O[C@@H](C)[C@H](C)[C@H](O)[C@@H](C)C=CC=CC=C1)O)C(OC)=O |c:61,t:0,2,4,6,57,59,&1:8,10,12,14,16,18,21,22,23,25,29,32,34,38,41,47,49,51,53| |
| InChi Key | UAZIZEMIKKIBCA-TYVGYKFWSA-N |
| InChi Code | InChI=1S/C48H75NO17/c1-28-18-16-14-12-10-8-6-7-9-11-13-15-17-19-35(65-47-45(59)42(49)44(58)31(4)64-47)25-39-41(46(60)62-5)38(55)27-48(61,66-39)26-34(52)23-37(54)36(53)21-20-32(50)22-33(51)24-40(56)63-30(3)29(2)43(28)57/h6-19,28-39,41-45,47,50-55,57-59,61H,20-27,49H2,1-5H3/b7-6+,10-8+,11-9+,14-12+,15-13+,18-16+,19-17+/t28-,29-,30-,31+,32+,33+,34-,35-,36+,37+,38-,39-,41+,42-,43+,44+,45-,47-,48+/m0/s1 |
| Chemical Name | methyl (1R,3S,5R,6R,9R,11R,15S,16R,17R,18S,19E,21E,23E,25E,27E,29E,31E,33R,35S,36R,37S)-33-(((2R,3S,4S,5S,6R)-4-amino-3,5-dihydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-1,3,5,6,9,11,17,37-octahydroxy-15,16,18-trimethyl-13-oxo-14,39-dioxabicyclo[33.3.1]nonatriaconta-19,21,23,25,27,29,31-heptaene-36-carboxylate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Amphotericin B methyl ester prevents the formation of HIV-1 particles while having no discernible impact on the plasma membrane binding, lipid raft association, or multimerization of Gag[1]. |
| Toxicity/Toxicokinetics |
Hepatotoxicity Mild and transient elevations in liver enzymes occur in up to 20% of patients receiving amphotericin. Clinically apparent hepatotoxicity is rare, but several convincing cases have been published. The liver injury arises as early as 4 to 14 days after starting therapy, typically with a hepatocellular or mixed pattern of enzyme elevation. Most patients have no symptoms or jaundice. Recovery occurs promptly upon stopping therapy. In addition, isolated but dramatic instances of hyperbilirubinemia arising within days of starting amphotericin have been reported with elevations largely in the direct (conjugated) bilirubin fraction. These patients become visually jaundiced but have no constitutional symptoms, minimal if any elevations in serum ALT or alkaline phosphatase levels, and no evidence of frank hepatic injury. Finally, rare instances of acute cholestatic hepatitis with jaundice have been reported in patients receiving amphotericin, but these patients have generally been critically ill and exposed to multiple potentially hepatotoxic agents, so that the attribution to amphotericin has been weak. Likelihood score: C (probable cause of clinically apparent liver injury). |
| References |
[1]. Inhibition of human immunodeficiency virus type 1 assembly and release by the cholesterol-binding compound amphotericin B methyl ester: evidence for Vpu dependence. J Virol. 2008 Oct;82(19):9776-81. [2]. Comparative toxicological studies of amphotericin B methyl ester and amphotericin B in mice, rats, and dogs. ntimicrob Agents Chemother. 1976 Oct;10(4):687-90. |
| Additional Infomation |
Amphotericin B methyl ester is the methyl ester of amphotericin B. It has a role as an antifungal agent, an antiinfective agent and a metabolite. It is a macrolide, a monosaccharide derivative and a methyl ester. It is functionally related to an amphotericin B. Amphotericin B is an antifungal agent with a broad spectrum of activity against many fungal species. Amphotericin B commonly causes mild to moderate serum aminotransferase elevations and can cause hyperbilirubinemia, but acute, clinically apparent drug induced liver injury from amphotericin B therapy is exceedingly rare. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~53.30 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.0660 mL | 5.3299 mL | 10.6597 mL | |
| 5 mM | 0.2132 mL | 1.0660 mL | 2.1319 mL | |
| 10 mM | 0.1066 mL | 0.5330 mL | 1.0660 mL |