Physicochemical Properties
| Molecular Formula | C23H22O7 |
| Molecular Weight | 410.41658 |
| Exact Mass | 410.137 |
| Elemental Analysis | C, 67.31; H, 5.40; O, 27.29 |
| CAS # | 4208-09-7 |
| PubChem CID | 92207 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.675 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 30 |
| Complexity | 681 |
| Defined Atom Stereocenter Count | 3 |
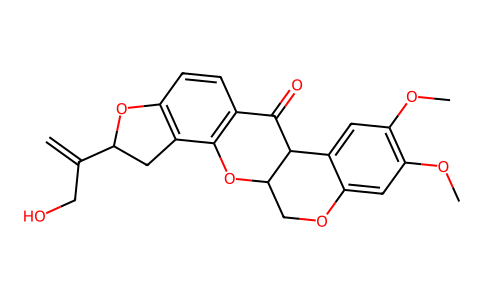
| SMILES | COC1=C(C=C2C(=C1)C3C(CO2)OC4=C(C3=O)C=CC5=C4CC(O5)C(=C)CO)OC |
| InChi Key | ZJMLELXRQUXRIU-HBGVWJBISA-N |
| InChi Code | InChI=1S/C23H22O7/c1-11(9-24)16-7-14-15(29-16)5-4-12-22(25)21-13-6-18(26-2)19(27-3)8-17(13)28-10-20(21)30-23(12)14/h4-6,8,16,20-21,24H,1,7,9-10H2,2-3H3/t16-,20-,21+/m1/s1 |
| Chemical Name | (1S,6R,13S)-6-(3-hydroxyprop-1-en-2-yl)-16,17-dimethoxy-2,7,20-trioxapentacyclo[11.8.0.03,11.04,8.014,19]henicosa-3(11),4(8),9,14,16,18-hexaen-12-one |
| Synonyms | Amorphigenin; 8'-Hydroxyrotenone; 4208-09-7; 3'-Hydroxyrotenone; Rotenone, 8'-hydroxy-; (2r,6as,12as)-2-(3-hydroxyprop-1-en-2-yl)-8,9-dimethoxy-1,2,12,12a-tetrahydrochromeno[3,4-b]furo[2,3-h]chromen-6(6ah)-one; (1S,6R,13S)-6-(3-hydroxyprop-1-en-2-yl)-16,17-dimethoxy-2,7,20-trioxapentacyclo[11.8.0.03,11.04,8.014,19]henicosa-3(11),4(8),9,14,16,18-hexaen-12-one; (1S,6R,13S)-6-(3-hydroxyprop-1-en-2-yl)-16,17-dimethoxy-2,7,20-trioxapentacyclo(11.8.0.03,11.04,8.014,19)henicosa-3(11),4(8),9,14,16,18-hexaen-12-one; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Larvicidal |
| ln Vitro |
Amorphigenin is a rotenoid compound isolated from the seeds of amorphigenin. Studies have shown that Amorphigenin can inhibit the proliferation of various tumor cells. This study aims to investigate the anti-tumor effects of amorphigenin on human lung adenocarcinoma cisplatin-resistant cell line A549/DDP and its possible molecular mechanism.[1] Amorphigenin could inhibit proliferation of A549/DDP cells for 48 h[half maximal inhibitory concentration (IC50)] as (2.19±0.92)μmol/L, inhibit clonal formation and induce cell apoptosis. In addition, combined Amorphigenin and cisplatin can synergistically inhibit A549/DDP cell growth and promote apoptosis. The expression of drug-resistant protein LRP was decreased.[1] |
| Cell Assay |
Cell culture [1] Human lung adenocarcinoma cisplatin-resistant cell lines A549/DDP and A549 cells were stored in our laboratory and cultured in RPMIS-1640 medium containing 10% fetal bovine serum, penicillin (100 U/mL) and streptomycin (100 U/mL) in an incubator at 37 ℃, 5%CO2 and saturated humidity. The cells were routinely passed every 2-3 days, and cells of logarithmic growth stage were used in all experiments. Cell viability was determined by CCK-8 [1] The cells of logarithmic growth stage were digested by conventional pancreatic enzymes into single cell suspension, which was planted in 96-well plates at a density of 5.0×103 cells/holes and incubated overnight in an incubator. The final concentration is 0.5 μmol/L, 1 μmol/L, 2 μmol/L, 4 μmol/L, 8 μmol/L and 16 μmol/L. amorphigeninor the final concentration is 2.5 μg/mL, 5 μg/mL, 10 μg/ L, respectively μg/mL, 20 μg/mL, 40 μg/mL cisplatin. After cultured in the incubator for 24 h and 48 h, the original culture medium was discarded, and then 100 μL CCK-8 reagent newly prepared according to the instructions was added to each well. After incubating in the incubator for 2 h, the absorbance value was measured at the wavelength of 450 nm with an enzyme label. The survival rate of the drug on cells was calculated according to the following formula: survival rate %= light absorption value of the experimental group/light absorption value of the control group ×100, and half maximal inhibitory concentration (IC50) for 48 h was calculated. Clone formation experiment [1] The cells of logarithmic growth stage were digested into a single cell suspension, and 500 cells/pores were inoculated in a 6cm petri dish. When the cell growth was stable, The final concentration was 0.062, 5 μmol/L, 0.125 μmol/L, 0.25 μmol/L amorphigenin, and the same amount of medium was added to blank control group. Cultured in an incubator for 15 days, the culture was terminated when visible cell clones were formed, washed twice with PBS, then fixed with methanol, then stained with crystal violet, washed and dried, photographed and counted under a microscope. Clone formation rate %=(number of clones in experimental group/number of clones in control group)×100%. Combination index calculation [1] We used the Chou-Talalay method to investigate the possibility of synergistic drug combinations. The cells of logarithmic growth stage were digested into single cell suspension by conventional pancreatic enzymes, which were planted in 96-well plates at a density of 5.0×103 cells/holes, incubated in an incubator overnight, and the original medium was removed. Human lung adenocarcinoma cisplatin-resistant cell line A549/DDP was treated with 0.5 μmol/L and 1 μmol/L amorphigeninfor 24 h. They were then treated with 2.5 μg/mL, 5 μg/mL, 10 μg/mL, 20 μg/mL, 40 μg/mL cisplatin for 24 h. The original culture medium was discarded, and then 100 μL CCK8 reagent newly prepared according to the instructions was added to each well. After incubating in the incubator for 2 h, the absorbance value was measured at the wavelength of 450 nm with an enzymoleter. The survival rate of cells was calculated according to the following formula: survival rate %= light absorption value of experimental group/light absorption value of control group ×100. CompuSyn software was used to automatically calculate the joint index of the two drugs and draw the equivalent diagram of the two drugs, where (D1/Dx1) is the horizontal coordinate, (D2/Dx2) is the vertical coordinate, D1 and D2 are the required concentrations of the two drugs when the combination of the two drugs produces x effect. Dx1 and Dx2 are the respective concentrations of the two drugs when x effect occurs when the two drugs are used alone. According to the Chou-Talalay theorem, if CI < 1, the combination of the two drugs is synergistic, if CI=1, it is additive, and if CI > 1, it is antagonistic. Apoptosis assay [1] Cells of logarithmic growth stage were digested into a single cell suspension (8×103 cells/well), inoculated on a 6-well plate and incubated overnight in an incubator. The final concentration is 0.5 μmol/L, 1 μmol/L, 2 μmol/L, 4 μmol/L, 8 μmol/L and 16 μmol/L. amorphigenin In the combined group, the concentration of amorphigenin was 0.5 μmol/L and cisplatin was 10 μg/mL. After treatment for 48 h, cells were digested and collected, stained with PI and FITC Annexin V. The apoptosis rate was detected by flow cytometry. Western Blotting [1] Cells of logarithmic growth stage were inoculated in a 6cm petri dish at a density of 2×105 cells/well. After incubation overnight, drugs were added as follows: After 48 h treatment inamorphigeningroup (0.5 μmol/L), cisplatin group (10 μg/mL), amorphigenin combined with cisplatin group, total cell protein can be extracted. The protein concentration is determined by BCA method. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was then transferred to PVDF membrane, 5% skim milk powder was enclosed at room temperature for 2 h, the primary antibody was added and incubated at 4 ℃ overnight, and the secondary antibody labeled with horseradish peroxidase was incubated at room temperature for 1h, and the membrane was exposed by horseradish peroxidase HRP-ECL spectrophotometry after washing. |
| References | [1]. Zhongguo Fei Ai Za Zhi. 2016 Dec 20;19(12):805–812. |
| Additional Infomation |
Amorphigenin is a member of rotenones. Amorphigenin has been reported in Berchemia discolor, Dalbergia monetaria, and Amorpha fruticosa with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4365 mL | 12.1826 mL | 24.3653 mL | |
| 5 mM | 0.4873 mL | 2.4365 mL | 4.8731 mL | |
| 10 mM | 0.2437 mL | 1.2183 mL | 2.4365 mL |