Physicochemical Properties
| Molecular Formula | C13H13BRFN5O4S2 |
| Molecular Weight | 466.30600 |
| Exact Mass | 464.957 |
| CAS # | 348635-87-0 |
| PubChem CID | 10238657 |
| Appearance | White to off-white solid powder |
| Density | 1.9±0.1 g/cm3 |
| Boiling Point | 651.5±65.0 °C at 760 mmHg |
| Melting Point | 128.6-130.0 °C |
| Flash Point | 347.8±34.3 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.718 |
| LogP | 1.98 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 26 |
| Complexity | 741 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | BREATYVWRHIPIY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C13H13BrFN5O4S2/c1-8-12(14)10-5-4-9(15)6-11(10)20(8)25(21,22)13-16-7-19(17-13)26(23,24)18(2)3/h4-7H,1-3H3 |
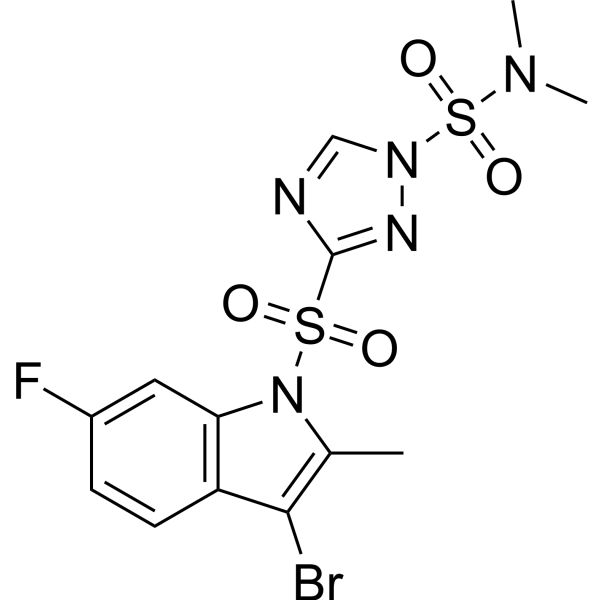
| Chemical Name | 3-(3-bromo-6-fluoro-2-methylindol-1-yl)sulfonyl-N,N-dimethyl-1,2,4-triazole-1-sulfonamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion In a rat metabolism study, Han Wistar rats were treated with a single gavage dose of (indole- (14)C) or (triazole- (14)C) amisulbrom (radiochemical purity > or = 95.2%) at 10 and 1000 mg/kg in aqueous 0.5% methy1cellulose. A series of experiments were then performed (4/sex/dose group) evaluating the excretion and tissue distribution of radioactivity at each dose level at up to 120 hours post-dose, and plasma and whole blood kinetics were determined. An additional experiment was conducted examining the excretion of radioactivity from bile duct-cannulated rats following a single oral dose of (indole-(14)C) NC-224 at 10 or 1000 mg/kg. To examine the distribution of radioactivity in tissues over time, rats were also dosed with (indole- (14) C) amisulbrom at 10 or 1000 mg/kg and 3 rats/sex/time point were then sacrificed at 2 and 24 hours post-dose (10 mg/kg) or 12 and 72 hours post-dose (1000 mg/kg). Excreta and tissue/organ samples from these experiments were radioassayed, and selected pooled samples of feces, urine, bile, liver, and plasma were analyzed to determine the metabolic profile. Radioactivity was not detectable in expired air in a preliminary study. The overall recovery of the radioactivity was 90-108% of the administered dose for both the 10 and 1000 mg/kg doses, and the pattern of excretion was similar between sexes for the two (14)C-labels. Fecal excretion was the major route of elimination. For the 10 mg/kg dose groups, fecal excretion accounted for 80-98% of the administered dose, while urine accounted for 10-14%. Radioactivity remaining in the carcass and tissues at 120 hours post-dose accounted for <0.5%. For the 1000 mg/kg dose groups, fecal excretion accounted for 89-100% of the administered dose, urine accounted for 1-3% of the administered dose, and radioactivity remaining in the carcass at 120 hours post-dose accounted for 0.05%. At both dose levels, the majority of excretion occurred from 0-48 hours post-dose. The overall total recovery from bile duct cannulated rats was 92-97% of the administered dose, and the patterns of absorption and excretion were similar between sexes. Data from the bile duct cannulated rats indicate that 49-50% of the administered dose was absorbed from the gastro-intestinal tract at 10 mg/kg; however, in contrast, only 5% of the administered dose was absorbed at the 1000 mg/kg dose level, indicating that gastro-intestinal absorption was saturated at the high dose. In the 10 mg/kg dose group, biliary excretion accounted for 40-41 % of the administered dose and 44% of the administered dose was unabsorbed test material eliminated in the feces. Only 9% of the administered dose was in the urine. At the 1000 mg/kg dose, feces accounted for 85-86% of the administered dose, and bile and urine each accounted for 1-3% of the administered dose. The time to maximum concentration in plasma (T max) occurred at 2-6 hours post -dose for the 10 mg/kg groups and 6-12 post-hours in the 1000 mg/kg group, and maximum plasma concentrations (Cmax) were 2-6 Ilg equiv./g for the 10 mg/kg group and 12-30 Ilg equiv./g in the 1000 mg/kg group. The elimination half-life was 18-35 hours at 10 mg/kg and 8-13 hours at 1000 mg/kg. For the triazole labeled material the T max was delayed especially at the 1000 mg/kg dose group. At all the time points, the concentrations of (14)C-residues in tissues (excluding the GI tract) were typically highest in liver followed by kidneys, and the distribution of (14)C-residues among tissues was generally similar between sexes. Residue levels increased with dose, but the increases were not proportional to the increase in the dose level. By 120 hours post-dose, only minor amounts of radioactivity "0.5% administered dose) were detectable in a few organs, such as kidneys and liver. Metabolism / Metabolites In a rat metabolism study, Han Wistar rats were treated with a single gavage dose of (indole- (14) C) or (triazole- (14)C) amisulbrom (radiochemical purity > or = 95.2%) at 10 and 1000 mg/kg in aqueous 0.5% methy1cellulose. A series of experiments were then performed (4/sex/dose group) evaluating the excretion and tissue distribution of radioactivity at each dose level at up to 120 hours post-dose, and plasma and whole blood kinetics were determined. An additional experiment was conducted examining the excretion of radioactivity from bile duct-cannulated rats following a single oral dose of (indole-(14)C) NC-224 at 10 or 1000 mg/kg. To examine the distribution of radioactivity in tissues over time, rats were also dosed with (indole- (14) C) amisulbrom at 10 or 1000 mg/kg and 3 rats/sex/time point were then sacrificed at 2 and 24 hours post-dose (10 mg/kg) or 12 and 72 hours post-dose (1000 mg/kg). Excreta and tissue/organ samples from these experiments were radioassayed, and selected pooled samples of feces, urine, bile, liver, and plasma were analyzed to determine the metabolic profile. ... Parent compound was the major (14)C-residue identified in feces of both sexes and (14)C-labels, accounting for 40-52% of the administered dose at 10 mg/kg and 83-89% at 1000 mg/kg. Other components/metabolites isolated from feces, urine and bile each accounted for <5% of the administered dose, with the exception of one component isolated from the bile of the low-dose rats that accounted for 5.3% of the administered dose. This component was tentatively identified as an unspecified conjugate of Metabolite IT-2. In addition to parent, nine primary metabolites and a glucuronide conjugate of one metabolite were identified in urine, feces and/or bile. Two of these metabolites (IT-4 and IT-5) were also identified as being the major (14)C-residues in liver (each 12-20% of the total radioactive residues; TRR in liver) and plasma (each 14-56% of the TRR in blood) at Tmax. Based on the metabolic profile, the metabolism of amisulbrom in the rat involves a series of reactions including cleavage of the sulfonyl amino side chain on the triazole ring, debromination, oxidation/hydroxylation, limited cleavage of the sulfonyl bridge between the indole and triazole moieties, indole ring opening, and conjugation of primary metabolites. The major route of metabolism of amisulbrom in rats appears to involve the initial cleavage of the sulfonylamino side to form IT-4 and it subsequent hydroxylation to IT-5, followed by further degradation or conjugation of these primary metabolites. Biological Half-Life In a rat metabolism study, Han Wi star rats were treated with a single gavage dose of (indole- (14) C) or (triazole- (14)C) amisulbrom (radiochemical purity > or = 95.2%) at 10 and 1000 mg/kg in aqueous 0.5% methy1cellulose. ... The elimination half-life was 18-35 hours at 10 mg/kg and 8-13 hours at 1000 mg/kg. |
| References |
[1]. Amisulbrom causes cardiovascular toxicity in zebrafish (Danio rerio). Chemosphere. 2021 Nov;283:131236. |
| Additional Infomation |
Amisulbrom is a member of the class of bromoindoles that is 3-bromo-6-fluoro-2-methylindole substituted at position 1 by a 1-(dimethylsulfamyl)-1,2,4-triazole-3-sulfonyl group. A fungicide for use on potatoes to control late blight (Phytophthora infestans) and downy mildew [Plasmopara viticola). It has a low mammalian toxicity but it is considered to be a reproduction toxicant, is moderately toxic to birds and honey bees but poses a greater risk to aquatic species and earthworms. It has a role as a mitochondrial cytochrome-bc1 complex inhibitor and an antifungal agrochemical. It is a member of sulfamides, a sulfonamide, a member of triazoles, an organofluorine compound, a bromoindole, a sulfonamide fungicide and a triazole fungicide. Mechanism of Action Amisulbrom ... is an oomycete-specific fungicide which acts by inhibiting the mitochondrial respiration within the fungus target species. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1445 mL | 10.7225 mL | 21.4450 mL | |
| 5 mM | 0.4289 mL | 2.1445 mL | 4.2890 mL | |
| 10 mM | 0.2144 mL | 1.0722 mL | 2.1445 mL |