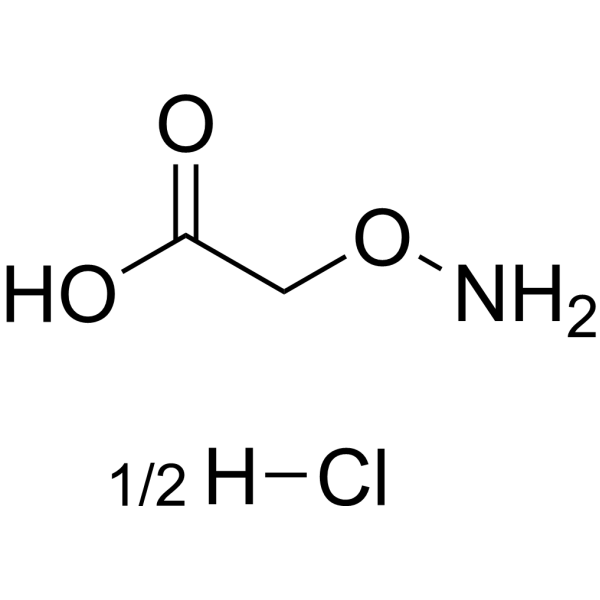
Physicochemical Properties
| Molecular Formula | C4H11CLN2O6 |
| Molecular Weight | 218.5929 |
| Exact Mass | 218.03 |
| CAS # | 2921-14-4 |
| Related CAS # | Aminoxyacetic acid;645-88-5 |
| PubChem CID | 2723609 |
| Appearance | White to off-white solid powder |
| Boiling Point | 326.7ºC at 760 mmHg |
| Melting Point | 156 °C (dec.)(lit.) |
| Flash Point | 151.4ºC |
| Vapour Pressure | 4.24E-05mmHg at 25°C |
| LogP | 0.125 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 13 |
| Complexity | 52.8 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | KBXIJIPYZKPDRU-UHFFFAOYSA-N |
| InChi Code | InChI=1S/2C2H5NO3.ClH/c2*3-6-1-2(4)5;/h2*1,3H2,(H,4,5);1H |
| Chemical Name | 2-aminooxyacetic acid;hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Malate-aspartate shuttle [1][3] - Glycolytic enzymes [1] - Glutamine metabolism-related targets [3] - GABA metabolism-related targets [2] |
| ln Vitro |
Aminoxyacetic acid hemihydrochloride (AOAA) dose-dependently decreases the stroma of C6 neuroastrocytoma cells. Treatment with aminooxyacetic acid hemihydrochloride greatly raised the proportion of cells in the G0/G1 phase of the cervix, and dramatically lowered the proportion of cells in the purified AS phase and G2/M phase. Treatment with hemihydrochloride resulted in a considerable reduction in the number of cells in the cell division phase. Aminoacetic acid hemihydrochloride considerably raised the percentage of early cells and surviving cells. Treatment of cells with 1 mM or 5 mM aminooxyacetic acid hemihydrochloride lowers cell senescence [1]. Glutamine-dependent cell lines revealed a stronger inhibitory effect on cell development by ammonia oxygen hemihydrochloride (AOA) than glutamine-dependent cells [3]. - In C6 glioma cells, Aminooxyacetic acid hemihydrochloride (as a malate-aspartate shuttle inhibitor) inhibited glycolysis, leading to a dose-dependent decrease in intracellular ATP levels. At concentrations of 0.5, 1, and 2 mM, ATP levels were reduced by 28%, 45%, and 63%, respectively, compared to the control group [1] - The compound altered the cell cycle of C6 glioma cells, increasing the proportion of cells in G0/G1 phase (from 42% to 68% at 2 mM) and decreasing S phase cells (from 38% to 17% at 2 mM) [1] - In breast cancer cell lines (MCF-7, MDA-MB-231, BT-474), Aminooxyacetic acid hemihydrochloride targeted glutamine metabolism, inhibiting cell proliferation with IC50 values ranging from 0.35 to 0.89 mM after 72 h of incubation [3] - It reduced the levels of glutamate and α-ketoglutarate (key metabolites of glutamine metabolism) in breast cancer cells by 35–52% at 0.5 mM [3] |
| ln Vivo |
GABA accumulates in the cerebellum and throughout the brain fairly quickly at first; five minutes after ethylene oxide hemihydrochloride (AOAA) injection, there is a noticeable increase in GABA levels. After 2 to 6 hours of ethylene oxide hemihydrochloride, the rapid final accumulation gradually slows down and achieves maximum levels (400 to 600% of control levels). Within twenty-four hours, GABA levels rose by about 250%. The amount of GABA increases by about 250% after 2–6 hours. the seizures were stopped. The 24-hour withdrawal of ammonia hemihydrochloride is identical to that which is required for expectant mothers [2]. - In rats, intraperitoneal administration of Aminooxyacetic acid hemihydrochloride (50 and 100 mg/kg) induced a dose-dependent accumulation of GABA in the brain. The GABA concentration in the cerebral cortex was increased by 62% (50 mg/kg) and 118% (100 mg/kg) compared to the control group [2] - The compound interacted with GABA receptors in the rat brain, enhancing GABA-mediated inhibitory neurotransmission [2] - In nude mice bearing MDA-MB-231 breast cancer xenografts, oral administration of Aminooxyacetic acid hemihydrochloride (100 mg/kg daily for 21 days) significantly inhibited tumor growth, reducing tumor volume by 48% and tumor weight by 52% compared to the control group [3] - It decreased the expression of glutamine metabolism-related enzymes (glutaminase 1, GLS1) in tumor tissues of xenograft mice [3] |
| Enzyme Assay |
- Glycolytic enzyme activity assay: C6 glioma cells were treated with Aminooxyacetic acid hemihydrochloride (0.5–2 mM) for 24 h. Cells were lysed, and the supernatant was collected. The activity of key glycolytic enzymes (hexokinase, pyruvate kinase) was measured by colorimetric assays based on substrate conversion and product formation [1] - Glutamine metabolism enzyme assay: Breast cancer cells were incubated with Aminooxyacetic acid hemihydrochloride (0.2–1 mM) for 48 h. Cell lysates were prepared, and the activity of glutaminase (GLS) was determined by detecting the release of ammonia from glutamine, with absorbance measured at a specific wavelength [3] |
| Cell Assay |
- C6 glioma cell assay: Cells were seeded in 96-well plates (5×10³ cells/well) and incubated overnight. Aminooxyacetic acid hemihydrochloride was added at concentrations of 0.1–2 mM, and incubation continued for 24–72 h. Intracellular ATP levels were measured using a luciferase-based assay kit. Cell cycle distribution was analyzed by propidium iodide staining and flow cytometry [1] - Breast cancer cell proliferation assay: Breast cancer cells were plated in 96-well plates (4×10³ cells/well) and allowed to adhere for 24 h. The compound was added at 0.1–1 mM, and cells were cultured for 72 h. Cell viability was assessed by a colorimetric assay, and IC50 values were calculated. Glutamate and α-ketoglutarate levels were quantified by HPLC after metabolite extraction [3] |
| Animal Protocol |
- Rat GABA accumulation assay: Male Wistar rats (200–250 g) were randomly divided into control group (saline i.p.) and Aminooxyacetic acid hemihydrochloride treatment groups (50, 100 mg/kg i.p.). The compound was dissolved in saline and administered once. Rats were sacrificed 2 h post-administration, and brain tissues (cerebral cortex, hippocampus) were collected. GABA concentration was determined by HPLC after extraction [2] - Breast cancer xenograft model: Nude mice (18–22 g) were implanted with MDA-MB-231 cells subcutaneously. When tumors reached 100–150 mm³, mice were divided into control group (vehicle oral gavage) and treatment group (100 mg/kg Aminooxyacetic acid hemihydrochloride oral gavage). The compound was dissolved in 0.5% carboxymethylcellulose sodium, administered once daily for 21 days. Tumor volume was measured every 3 days, and tumor weight was recorded at sacrifice. Tumor tissues were collected for enzyme expression analysis [3] |
| References |
[1]. Malate-aspartate shuttle inhibitor aminooxyacetic acid leads to decreased intracellular ATP levels and altered cell cycle of C6 glioma cells by inhibiting glycolysis. Cancer Lett. 2016 Aug 1;378(1):1-7. [2]. Aminooxyacetic acid induced accumulation of GABA in the rat brain. Interaction with GABA receptors and distribution in compartments. Naunyn Schmiedebergs Arch Pharmacol. 1983 Apr;322(3):210-5. [3]. Targeting Glutamine Metabolism in Breast Cancer with Aminooxyacetate. Clin Cancer Res. 2015 Jul 15;21(14):3263-73. |
| Additional Infomation |
(aminooxy)acetic acid hemihydrochloride is the hemihydrochloride salt of (aminooxy)acetic acid. It is a malate-aspartate shuttle (MAS) inhibitor which also inhibits the GABA degradating enzyme 4-aminobutyrate aminotransferase and cystathionine beta-synthetase. It contains an (aminooxy)acetate. - Aminooxyacetic acid hemihydrochloride is a potent inhibitor of the malate-aspartate shuttle, which plays a key role in intracellular redox balance and energy metabolism [1][3] - Its anticancer mechanism involves inhibiting glycolysis and glutamine metabolism, leading to energy depletion and cell cycle arrest in cancer cells [1][3] - The compound can cross the blood-brain barrier and modulate GABAergic neurotransmission by increasing brain GABA levels [2] - It shows potential as a therapeutic agent for glioma and breast cancer by targeting cancer cell metabolism [1][3] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~125 mg/mL (~1143.64 mM) H2O : ~100 mg/mL (~914.91 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (19.03 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (19.03 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (19.03 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 130 mg/mL (1189.39 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.5748 mL | 22.8739 mL | 45.7477 mL | |
| 5 mM | 0.9150 mL | 4.5748 mL | 9.1495 mL | |
| 10 mM | 0.4575 mL | 2.2874 mL | 4.5748 mL |