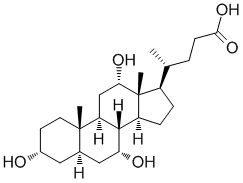
Allocholic acid (3-α,7-α,12-α-Trihydroxy-5-α-cholanoic Acid; 5α-Cholic Acid) is a fetal bile acid metabolized from animals and can be isolated from vertebrates and reappears during liver regeneration and carcinogenesis. It is a specific stimulant of the adult olfactory system and also a precursor to petromyzonol.
Physicochemical Properties
| Molecular Formula | C₂₄H₄₀O₅ |
| Molecular Weight | 408.57 |
| Exact Mass | 408.288 |
| CAS # | 2464-18-8 |
| PubChem CID | 160636 |
| Appearance | White to light yellow solid powder |
| Density | 1.184g/cm3 |
| Boiling Point | 583.9ºC at 760 mmHg |
| Melting Point | 250-251ºC |
| Flash Point | 321ºC |
| Vapour Pressure | 4.59E-16mmHg at 25°C |
| Index of Refraction | 1.558 |
| LogP | 3.448 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 29 |
| Complexity | 637 |
| Defined Atom Stereocenter Count | 11 |
| SMILES | C[C@H](CCC(=O)O)[C@H]1CC[C@@H]2[C@@]1([C@H](C[C@H]3[C@H]2[C@@H](C[C@@H]4[C@@]3(CC[C@H](C4)O)C)O)O)C |
| InChi Key | BHQCQFFYRZLCQQ-PGHAKIONSA-N |
| InChi Code | InChI=1S/C24H40O5/c1-13(4-7-21(28)29)16-5-6-17-22-18(12-20(27)24(16,17)3)23(2)9-8-15(25)10-14(23)11-19(22)26/h13-20,22,25-27H,4-12H2,1-3H3,(H,28,29)/t13-,14-,15-,16-,17+,18+,19-,20+,22+,23+,24-/m1/s1 |
| Chemical Name | (4R)-4-[(3R,5R,7R,8R,9S,10S,12S,13R,14S,17R)-3,7,12-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoic acid |
| Synonyms | Allocholic acid 3-α,7-α,12-α-Trihydroxy-5-α-cholanoic Acid 5α-Cholic Acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Allocholic acid promotes the conversion of [24-14C]cholic acid to [24-14C]isodeoxycholic acid and induces rapid culture in C. scindens and C. hylemonae cultures [4]. |
| ln Vivo | In gerbils, the primary acidic bile extraction product of cholestanol is allocholic acid, and vice versa [5]. |
| References |
[1]. Allocholic acid, the major component in bile from the river carpsucker, Carpiodes carpio (Rafinesque) (Catostomidae). Comp Biochem Physiol B. 1972 Jul 15;42(3):493-6. [2]. The olfactory system of migratory adult sea lamprey (Petromyzon marinus) is specifically and acutely sensitive to unique bile acids released by conspecific larvae. J Gen Physiol. 1995 May;105(5):569-87. [3]. Physiological characteristics of allo-cholic acid. J Lipid Res. 2003 Jan;44(1):84-92. [4]. Isolation and characterization of a bile acid inducible 7alpha-dehydroxylating operon in Clostridium hylemonae TN271. Anaerobe. 2010 Apr;16(2):137-46. [5]. Elliott WH, Hyde PM. Metabolic pathways of bile acid synthesis. Am J Med. 1971 Nov;51(5):568-79. |
| Additional Infomation |
Allocholic acid is an allo-bile acid that is 5alpha-cholan-24-oic acid bearing three alpha-hydroxy substituents at position 3, 7 and 12. It has a role as a marine metabolite, a rat metabolite and a human metabolite. It is a C24-steroid, a 3alpha-hydroxy steroid, a 7alpha-hydroxy steroid, a 12alpha-hydroxy steroid and an allo-bile acid. It is a conjugate acid of an allocholate. Allocholic acid has been reported in Petromyzon marinus with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~33.33 mg/mL (~81.58 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.09 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.09 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (5.09 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4476 mL | 12.2378 mL | 24.4756 mL | |
| 5 mM | 0.4895 mL | 2.4476 mL | 4.8951 mL | |
| 10 mM | 0.2448 mL | 1.2238 mL | 2.4476 mL |