Physicochemical Properties
| Molecular Formula | C30H48O4 |
| Molecular Weight | 472.6997 |
| Exact Mass | 472.355 |
| CAS # | 18649-93-9 |
| Related CAS # | Alisol B 23-acetate;26575-95-1 |
| PubChem CID | 15558620 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 567.1±50.0 °C at 760 mmHg |
| Flash Point | 181.2±23.6 °C |
| Vapour Pressure | 0.0±3.5 mmHg at 25°C |
| Index of Refraction | 1.560 |
| LogP | 4.37 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 34 |
| Complexity | 917 |
| Defined Atom Stereocenter Count | 9 |
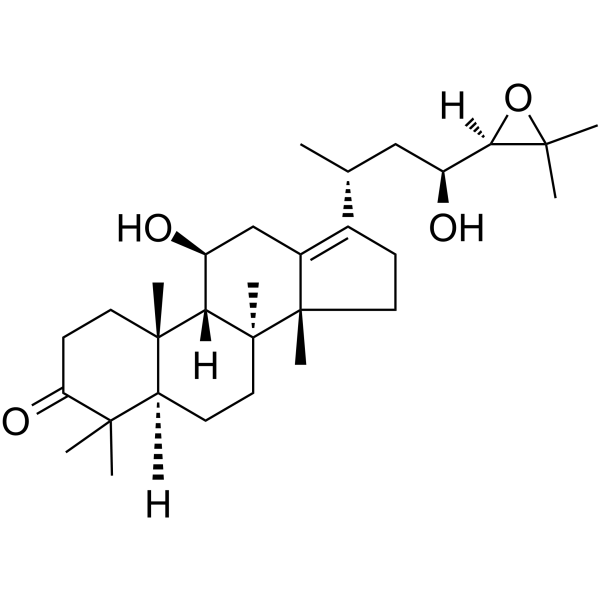
| SMILES | C[C@H](C[C@@H]([C@@H]1C(O1)(C)C)O)C2=C3C[C@@H]([C@H]4[C@]5(CCC(=O)C([C@@H]5CC[C@@]4([C@]3(CC2)C)C)(C)C)C)O |
| InChi Key | GBJKHDVRXAVITG-UNPOXIGHSA-N |
| InChi Code | InChI=1S/C30H48O4/c1-17(15-21(32)25-27(4,5)34-25)18-9-13-29(7)19(18)16-20(31)24-28(6)12-11-23(33)26(2,3)22(28)10-14-30(24,29)8/h17,20-22,24-25,31-32H,9-16H2,1-8H3/t17-,20+,21+,22+,24+,25-,28+,29+,30+/m1/s1 |
| Chemical Name | (5R,8S,9S,10S,11S,14R)-17-[(2R,4S)-4-[(2R)-3,3-dimethyloxiran-2-yl]-4-hydroxybutan-2-yl]-11-hydroxy-4,4,8,10,14-pentamethyl-1,2,5,6,7,9,11,12,15,16-decahydrocyclopenta[a]phenanthren-3-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Alisol B exhibited cytotoxic effects against a panel of cancer cell lines including Hep3B, HepG2, HeLa, SK-BR-3, MDA-MB-231, MCF-7, PC3, and C666-1, with IC₅₀ values ranging from approximately 20 to 49 μmol/L at 48 hours. It was more potent than alisol A and alisol A 24-acetate, and comparable to alisol B 23-acetate [3]. Treatment with Alisol B induced autophagy, as evidenced by increased GFP-LC3 puncta formation in multiple cell lines (e.g., MCF-7), the appearance of double-membraned autophagosomes observed via transmission electron microscopy, and enhanced LC3-I to LC3-II conversion in the presence of lysosomal protease inhibitors, indicating increased autophagic flux [3]. Alisol B treatment caused cell cycle arrest at the G₁ phase in MCF-7 cells in a time-dependent manner, accompanied by accumulation of the cell cycle inhibitor p27 [3]. Alisol B induced apoptosis, as demonstrated by increased Annexin V/7-AAD positive staining (early and late apoptosis) in MCF-7 cells after 48 hours of treatment and cleavage of poly ADP ribose polymerase (PARP) [3]. The induction of autophagy by Alisol B was mediated through calcium mobilization, leading to activation of the CaMKK-AMPK-mTOR pathway. Inhibition of CaMKK (with STO-609) or chelation of intracellular calcium (with BAPTA/AM) attenuated AMPK phosphorylation and GFP-LC3 puncta formation [3]. Alisol B triggered endoplasmic reticulum (ER) stress and the unfolded protein response (UPR), specifically activating the PERK-elF2α-ATF4-CHOP/GRP78 and ATF6 signaling pathways, but not the IRE1-XBP1 splicing pathway, as shown by Western blot, reporter assay, and RT-PCR analysis [3]. Genetic ablation of autophagy (using ATG7-deficient mouse embryonic fibroblasts) increased sensitivity to Alisol B-induced cell death, suggesting autophagy acts as a pro-survival response in this context [3]. |
| Enzyme Assay |
The inhibitory activity of Alisol B on the SERCA pump was assessed using purified Ca²⁺ ATPase from female rabbit hind leg skeletal muscle sarcoplasmic reticulum (SR) membranes (enriched in SERCA1A isoform). ATPase activity was determined using an enzyme-coupled assay system involving pyruvate kinase and lactate dehydrogenase, which links ATP hydrolysis to NADH oxidation, monitored spectrophotometrically. Inhibition curves were generated by incubating the enzyme with varying concentrations of Alisol B [3]. The effect of Alisol B on the SERCA2B isoform was evaluated using porcine brain microsomal membranes. Due to lower Ca²⁺ ATPase activity in these membranes, a more sensitive phosphate liberation assay was employed to measure the rate of Ca²⁺-dependent ATP hydrolysis. The microsomes were incubated with Alisol B at different concentrations, and the amount of inorganic phosphate released was quantified [3]. |
| Cell Assay |
A GFP-LC3 reporter assay was used to screen for autophagy inducers. MCF-7 cells were transiently transfected with a GFP-LC3 plasmid. After transfection, cells were treated with test compounds or controls (e.g., tamoxifen). Autophagy induction was quantified by counting the percentage of cells displaying punctate GFP-LC3 fluorescence under a fluorescence microscope [3]. Cytotoxicity was measured using the MTT assay. Cells were seeded in plates, treated with serial dilutions of Alisol B for a specified period (e.g., 48 hours), followed by incubation with MTT reagent. The formed formazan crystals were dissolved, and absorbance was measured to determine cell viability relative to untreated controls. IC₅₀ values were calculated from dose-response curves [3]. Transmission electron microscopy was performed to visualize autophagic structures. Cells treated with Alisol B were fixed with glutaraldehyde, post-fixed with osmium tetroxide, embedded in resin, and sectioned. Ultrathin sections were stained with uranyl acetate and lead citrate and examined under an electron microscope to identify double-membraned autophagosomes and autolysosomes [3]. Cell cycle distribution and apoptosis were analyzed by flow cytometry. For cell cycle analysis, cells were fixed, treated with RNase, stained with propidium iodide, and analyzed for DNA content. For apoptosis detection, cells were stained with Annexin V and 7-AAD and analyzed to distinguish viable, early apoptotic, late apoptotic, and necrotic populations [3]. Western blot analysis was used to examine protein expression and phosphorylation. Cells were lysed, proteins were separated by SDS-PAGE, transferred to membranes, and probed with specific primary antibodies (e.g., against LC3, p-AMPK, p-eIF2α, CHOP, PARP, β-actin). Signals were detected using appropriate secondary antibodies and chemiluminescence [3]. To assess the role of calcium, cells were pre-treated with the intracellular calcium chelator BAPTA/AM or the CaMKK inhibitor STO-609 prior to Alisol B treatment, followed by analysis of autophagy markers (GFP-LC3 puncta) or cell viability [3]. To investigate the role of autophagy pharmacologically and genetically, cells were pre-treated with the autophagy inhibitor 3-methyladenine (3-MA), or transfected with small interfering RNAs targeting beclin1, or compared wild-type versus ATG7-deficient fibroblasts, before exposure to Alisol B and subsequent viability or death assays [3]. UPR pathway activation was assessed via multiple methods: Western blot for PERK, eIF2α phosphorylation, and downstream proteins (ATF4, CHOP, GRP78); luciferase reporter assay for ATF6 transcriptional activity; and RT-PCR to analyze splicing of XBP1 mRNA as a readout for IRE1 activation [3]. |
| References |
[1]. [Alisol B inhibited complement 3a-induced human renal tubular epithelial to mesenchymal transition]. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2012 Oct;32(10):1407-12. [2]. Alisol-B, a novel phyto-steroid, suppresses the RANKL-induced osteoclast formation and prevents bone loss in mice. Biochem Pharmacol. 2010 Aug 1;80(3):352-61. [3]. Alisol B, a novel inhibitor of the sarcoplasmic/endoplasmic reticulum Ca(2+) ATPase pump, induces autophagy, endoplasmic reticulum stress, and apoptosis. Mol Cancer Ther. 2010 Mar;9(3):718-30. |
| Additional Infomation |
Alisol B is a triterpenoid. Alisol B has been reported in Alisma, Alisma lanceolatum, and other organisms with data available. Alisol B is a protostane-type triterpenoid isolated from the rhizome of Alisma orientale, a plant used in traditional Chinese medicine [3]. This study identifies Alisol B as a novel natural product inducer of autophagy. Its mechanism of action involves inhibition of the SERCA pump, leading to disruption of calcium homeostasis. This perturbation subsequently activates two parallel cellular responses: 1) A pro-survival autophagy pathway via CaMKK-AMPK-mTOR signaling, and 2) ER stress/UPR (specifically PERK and ATF6 branches) culminating in apoptotic cell death [3]. The study suggests that the unique property of Alisol B in selectively activating certain UPR pathways (PERK/ATF6 but not IRE1) might be advantageous for promoting apoptotic death over survival. It proposes Alisol B as a candidate for further development, potentially via prodrug strategies similar to those explored for other SERCA inhibitors like thapsigargin [3]. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~211.55 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (5.29 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.29 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.29 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1155 mL | 10.5775 mL | 21.1551 mL | |
| 5 mM | 0.4231 mL | 2.1155 mL | 4.2310 mL | |
| 10 mM | 0.2116 mL | 1.0578 mL | 2.1155 mL |