Physicochemical Properties
| Molecular Formula | C32H52O6 |
| Molecular Weight | 532.7517 |
| Exact Mass | 532.376 |
| CAS # | 18674-16-3 |
| Related CAS # | Alisol A;19885-10-0 |
| PubChem CID | 76336194 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 641.1±55.0 °C at 760 mmHg |
| Melting Point | 194-196 ºC |
| Flash Point | 196.5±25.0 °C |
| Vapour Pressure | 0.0±4.3 mmHg at 25°C |
| Index of Refraction | 1.547 |
| LogP | 5.27 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 38 |
| Complexity | 1010 |
| Defined Atom Stereocenter Count | 9 |
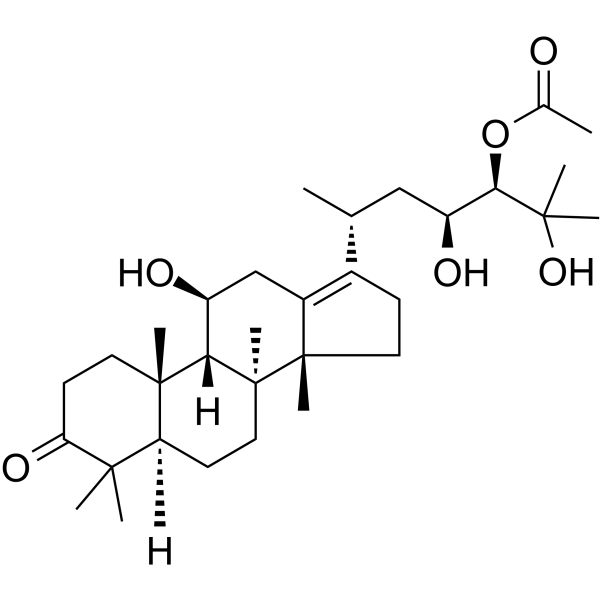
| SMILES | C[C@H](C[C@@H]([C@H](C(C)(C)O)OC(=O)C)O)C1=C2C[C@@H]([C@H]3[C@]4(CCC(=O)C([C@@H]4CC[C@@]3([C@]2(CC1)C)C)(C)C)C)O |
| InChi Key | WXHUQVMHWUQNTG-JSWHPQHOSA-N |
| InChi Code | InChI=1S/C32H52O6/c1-18(16-23(35)27(29(5,6)37)38-19(2)33)20-10-14-31(8)21(20)17-22(34)26-30(7)13-12-25(36)28(3,4)24(30)11-15-32(26,31)9/h18,22-24,26-27,34-35,37H,10-17H2,1-9H3/t18-,22+,23+,24+,26+,27-,30+,31+,32+/m1/s1 |
| Chemical Name | [(3R,4S,6R)-2,4-dihydroxy-6-[(5R,8S,9S,10S,11S,14R)-11-hydroxy-4,4,8,10,14-pentamethyl-3-oxo-1,2,5,6,7,9,11,12,15,16-decahydrocyclopenta[a]phenanthren-17-yl]-2-methylheptan-3-yl] acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Animal Protocol |
A pharmacokinetic study was conducted in rats. Male Wistar rats were fasted for 12 hours with free access to water prior to the experiment. Each rat was orally administered a Rhizoma Alismatis extract at a dose of 18 g/kg (calculated by crude drug weight). The extract was prepared by refluxing the crude drug with 95% ethanol and then dissolving the residue in an aqueous solution containing 0.5% sodium carboxymethylcellulose (CMC-Na). The final extract concentration was 1.5 g/mL (equivalent to crude drug), containing 0.5 mg/g Alisol A and 0.4 mg/g Alisol A 24-acetate. Blood samples (0.3 mL) were collected from the oculi chorioideae vein at predetermined time points (0, 0.08, 0.17, 0.33, 0.75, 1, 1.5, 2, 4, 6, 7, 8, 9, 10, 12, 16, and 24 hours) post-dose. The samples were placed in heparinized tubes and centrifuged to separate plasma. The plasma samples were stored at -20 °C until analysis. [1] |
| ADME/Pharmacokinetics |
A sensitive LC-MS method was developed and validated for the simultaneous determination of Alisol A and Alisol A 24-acetate in rat plasma. The lower limit of quantification (LLOQ) for both analytes was 10 ng/mL. The mean extraction recovery of Alisol A 24-acetate from rat plasma was above 72.4%. The analyte was stable in rat plasma through three freeze-thaw cycles, for 24 hours at room temperature, and for 30 days at -20 °C. Following oral administration of the Rhizoma Alismatis extract to rats, the concentration-time profile of Alisol A 24-acetate showed a significant double-peak phenomenon. The pharmacokinetic parameters (mean ± SD) for Alisol A 24-acetate were as follows: terminal half-life (T1/2) = 2.9 ± 0.9 hours, maximum plasma concentration (Cmax) = 233.1 ± 55.4 μg/L, time to reach Cmax (Tmax) = 9.2 ± 1.0 hours, area under the concentration-time curve from 0 to 24 hours (AUC0–24h) = 1820 ± 464 μg·h/L, and AUC extrapolated to infinity (AUC0–∞) = 1874 ± 450 μg·h/L. The double-peak phenomenon may be attributed to interconversion between related protostane triterpenes in the extract (e.g., alisol B 23-acetate or alisol B 11,23-diacetate potentially transforming into Alisol A 24-acetate), variability in gastrointestinal absorption, or enterohepatic recirculation. [1] |
| References |
[1]. A sensitive liquid chromatography-mass spectrometry method for simultaneous determination of alisol A and alisol A 24-acetate from Alisma orientale (Sam.) Juz. in rat plasma. Anal Bioanal Chem. 2011 Jan;399(3):1363-9. [2]. Stability and structure studies on alisol a 24-acetate. Chem Pharm Bull (Tokyo). 2008 Jan;56(1):41-5. |
| Additional Infomation |
alisol A 24-acetate has been reported in Alisma with data available. Alisol A 24-acetate is a major protostane-type triterpene isolated from the Traditional Chinese Medicine Rhizoma Alismatis (the dried rhizome of Alisma orientale). It is reported to possess diuretic, anti-inflammatory, and hypocholesterolemic effects. The developed LC-MS method represents the first report for the simultaneous determination of Alisol A and Alisol A 24-acetate in rat plasma following oral administration of a Rhizoma Alismatis extract. [1] Alisol A 24-acetate is a major protostane-type triterpenoid isolated from the Traditional Chinese Medicine Rhizoma Alismatis (Alisma orientalis). The compound is unstable in various solvents. In protic solvents (e.g., methanol, ethanol), Alisol A 24-acetate undergoes rapid acetyl group migration, transforming into its isomer, alisol A 23-acetate. The transformation rate is faster in protic solvents than in aprotic solvents (e.g., acetone, acetonitrile, ethyl acetate, chloroform). When stored in methanol for an extended period (e.g., over 14 days), Alisol A 24-acetate and alisol A 23-acetate can further deacetylate to yield alisol A. The single crystal X-ray structure of Alisol A 24-acetate was determined, confirming its configuration. Rings A and B are in a twist boat conformation, ring C in a chair conformation, and ring D in an envelope conformation. The A/B and B/C ring junctions are trans-linked. The study provides the first report on the structural interconversion between alisol A 24-acetate, alisol A 23-acetate, and alisol A, as well as the first single crystal X-ray structure of alisol A 24-acetate. [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~93.85 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3 mg/mL (5.63 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 30.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 3 mg/mL (5.63 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 30.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8771 mL | 9.3853 mL | 18.7705 mL | |
| 5 mM | 0.3754 mL | 1.8771 mL | 3.7541 mL | |
| 10 mM | 0.1877 mL | 0.9385 mL | 1.8771 mL |