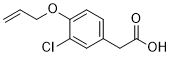
Alclofenac, a drug of the non-steroidal anti-inflammatory drug (NSAIDs) class, has been used to treat patients with chronic rheumatic diseases but was withdrawn from the UK market in 1979 due to concerns with its association with vasculitis and rash. Alclofenac has a pronounced effect upon the acute-phase protein response and the extent to which L-tryptophan is bound to plasma protein.
Physicochemical Properties
| Molecular Formula | C11H11O3CL |
| Molecular Weight | 226.65624 |
| Exact Mass | 226.04 |
| CAS # | 22131-79-9 |
| Related CAS # | 22131-79-9 (Alclofenac); 59960-34-8 (Alclofenac lysinate); 24049-18-1 (Alclofenac sodium) |
| PubChem CID | 30951 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.252g/cm3 |
| Boiling Point | 368.3ºC at 760mmHg |
| Flash Point | 176.6ºC |
| Vapour Pressure | 4.49E-06mmHg at 25°C |
| Index of Refraction | 1.553 |
| LogP | 2.531 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 15 |
| Complexity | 230 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | C=CCOC1=C(C=C(C=C1)CC(=O)O)Cl |
| InChi Key | ARHWPKZXBHOEEE-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H11ClO3/c1-2-5-15-10-4-3-8(6-9(10)12)7-11(13)14/h2-4,6H,1,5,7H2,(H,13,14) |
| Chemical Name | (4-(Allyloxy)-3-chlorophenyl)acetic acid |
| Synonyms | W 7320; W-7320; W7320; MY 101; MY-101; MY101; BRN 2116510; BRN-2116510; BRN2116510. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The absorption of alclofenac from the gastrointestinal tract is irregular. After oral or rectal administration maximum plasma concentrations are reached within 1-4 hours. Alclofenac is excreted in the urine mainly as glucuronide and as unchanged active substance. The volume of distribution is 0.1 L / kg For oral dose of 500mg: Renal clearance constant (av) 35ml/min Overall clearance constant (av) 37-69ml/min Metabolism / Metabolites the main metabolic product is alclofenac itself and alclofenac glucuronide Biological Half-Life The plasma half-life varies between 1.5 and 5.5 hours. |
| Toxicity/Toxicokinetics |
Protein Binding The binding to plasma proteins is 90-99%. |
| References | J Chromatogr. 1993 Nov 24;621(2):209-14. |
| Additional Infomation |
Alclofenac is an aromatic ether in which the ether oxygen links an allyl group to the 4-position of (3-chlorophenyl)acetic acid.A non-steroidal anti-inflammatory drug, it was withdrawn from the UK market in 1979 due to concerns with its association with vasculitis and rash. It has a role as a non-steroidal anti-inflammatory drug, a non-narcotic analgesic and a drug allergen. It is an aromatic ether, a monocarboxylic acid and a member of monochlorobenzenes. Alclofenac is a non-steroidal anti-inflammatory drug. It was withdrawn from the market in the United Kingdom in 1979. Drug Indication Alclofenac is indicated in rheumatology, in particular for the treatment of rheumatoid arthritis, ankylosing spondylitis and, as an analgesic, in painful arthritic pathologies. Mechanism of Action Alclofenac is an inhibitor of prostaglandin H2 synthase. The inhibition of the enzyme occurs through the reversible block of cyclooxygenase enzyme. Therefore, it prevents the production of inflammatory mediators (and pain) as prostacyclins and prostaglandins. Aclofenac has the ability to inhibit the biosynthesis of prostaglandins which may be an important factor in the action of these drugs, but in addition, the effect of these agents in displacing endogenous anti-inflammatory substances from plasma protein binding sites is thought to be an equally important effect in their mechanism of action |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.4119 mL | 22.0595 mL | 44.1189 mL | |
| 5 mM | 0.8824 mL | 4.4119 mL | 8.8238 mL | |
| 10 mM | 0.4412 mL | 2.2059 mL | 4.4119 mL |