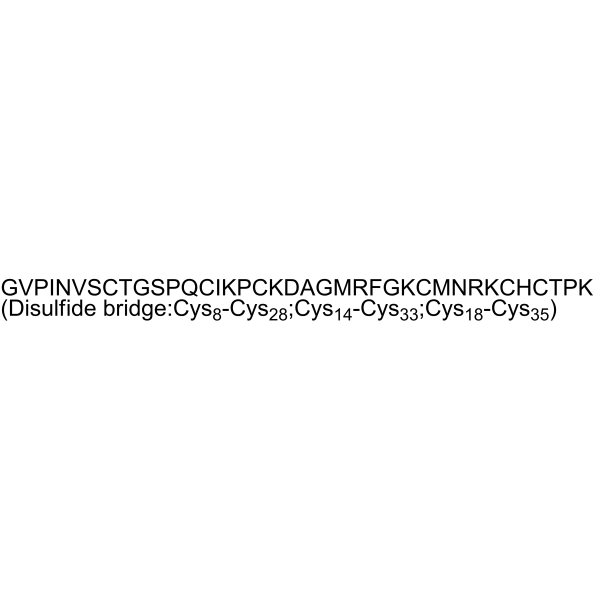Physicochemical Properties
| Molecular Formula | C169H278N54O48S8 |
| Molecular Weight | 4090.86864519119 |
| Exact Mass | 4088.877 |
| CAS # | 168147-41-9 |
| Related CAS # | Agitoxin-2 TFA |
| PubChem CID | 90489022 |
| Sequence | H-Gly-Val-Pro-Ile-Asn-Val-Ser-Cys(1)-Thr-Gly-Ser-Pro-Gln-Cys(2)-Ile-Lys-Pro-Cys(3)-Lys-Asp-Ala-Gly-Met-Arg-Phe-Gly-Lys-Cys(1)-Met-Asn-Arg-Lys-Cys(2)-His-Cys(3)-Thr-Pro-Lys-OH; glycyl-L-valyl-L-prolyl-L-isoleucyl-L-asparagyl-L-valyl-L-seryl-L-cysteinyl-L-threonyl-glycyl-L-seryl-L-prolyl-L-glutaminyl-L-cysteinyl-L-isoleucyl-L-lysyl-L-prolyl-L-cysteinyl-L-lysyl-L-alpha-aspartyl-L-alanyl-glycyl-L-methionyl-L-arginyl-L-phenylalanyl-glycyl-L-lysyl-L-cysteinyl-L-methionyl-L-asparagyl-L-arginyl-L-lysyl-L-cysteinyl-L-histidyl-L-cysteinyl-L-threonyl-L-prolyl-L-lysine (8->28),(14->33),(18->35)-tris(disulfide); H-GVPINVSC(1)TGSPQC(2)IKPC(3)KDAGMRFGKC(1)MNRKC(2)HC(3)TPK-OH; Gly-Val-Pro-Ile-Asn-Val-Ser-Cys-Thr-Gly-Ser-Pro-Gln-Cys-Ile-Lys-Pro-Cys-Lys-Asp-Ala-Gly-Met-Arg-Phe-Gly-Lys-Cys-Met-Asn-Arg-Lys-Cys-His-Cys-Thr-Pro-Lys (Disulfide bridge:Cys8-Cys28;Cys14-Cys33;Cys18-Cys35) |
| SequenceShortening | GVPINVSCTGSPQCIKPCKDAGMRFGKCMNRKCHCTPK (Disulfide bridge:Cys8-Cys28;Cys14-Cys33;Cys18-Cys35); GVPINVSCTGSPQCIKPCKDAGMRFGKCMNRKCHCTPK |
| Appearance | Typically exists as solid at room temperature |
| LogP | -18.2 |
| Hydrogen Bond Donor Count | 55 |
| Hydrogen Bond Acceptor Count | 65 |
| Rotatable Bond Count | 78 |
| Heavy Atom Count | 279 |
| Complexity | 9550 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCC(C)C1C(=O)NC(C(=O)N2CCCC2C(=O)NC3CSSCC(NC(=O)C(NC(=O)C4CSSCC(C(=O)N1)NC(=O)C(NC(=O)C5CCCN5C(=O)C(NC(=O)CNC(=O)C(NC(=O)C(CSSCC(C(=O)NC(C(=O)NC(C(=O)NC(C(=O)NC(C(=O)N4)CCCCN)CCCNC(=N)N)CC(=O)N)CCSC)NC(=O)C(NC(=O)CNC(=O)C(NC(=O)C(NC(=O)C(NC(=O)CNC(=O)C(NC(=O)C(NC(=O)C(NC3=O)CCCCN)CC(=O)O)C)CCSC)CCCNC(=N)N)CC6=CC=CC=C6)CCCCN)NC(=O)C(CO)NC(=O)C(C(C)C)NC(=O)C(CC(=O)N)NC(=O)C(C(C)CC)NC(=O)C7CCCN7C(=O)C(C(C)C)NC(=O)CN)C(C)O)CO)CCC(=O)N)CC8=CNC=N8)C(=O)NC(C(C)O)C(=O)N9CCCC9C(=O)NC(CCCCN)C(=O)O)CCCCN |
| InChi Key | MNSSWZUIQUJZTG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C169H278N54O48S8/c1-14-86(7)130-161(264)200-101(40-22-27-55-173)163(266)220-59-31-45-117(220)157(260)213-112-79-276-279-82-115(154(257)219-133(90(11)227)166(269)223-62-33-46-118(223)156(259)201-102(167(270)271)41-23-28-56-174)211-145(248)104(66-92-71-183-83-189-92)203-151(254)111-78-275-277-80-113(152(255)216-130)210-141(244)98(48-49-120(176)228)199-155(258)116-44-32-60-221(116)164(267)109(76-225)193-126(234)74-188-159(262)132(89(10)226)218-153(256)114(212-148(251)108(75-224)207-160(263)128(84(3)4)215-147(250)106(68-122(178)230)206-162(265)131(87(8)15-2)217-158(261)119-47-34-61-222(119)165(268)129(85(5)6)214-123(231)70-175)81-278-274-77-110(149(252)198-100(51-64-273-13)143(246)204-105(67-121(177)229)146(249)196-96(42-29-57-184-168(179)180)137(240)194-95(140(243)209-111)39-21-26-54-172)208-136(239)93(37-19-24-52-170)191-125(233)73-187-135(238)103(65-91-35-17-16-18-36-91)202-139(242)97(43-30-58-185-169(181)182)195-142(245)99(50-63-272-12)192-124(232)72-186-134(237)88(9)190-144(247)107(69-127(235)236)205-138(241)94(197-150(112)253)38-20-25-53-171/h16-18,35-36,71,83-90,93-119,128-133,224-227H,14-15,19-34,37-70,72-82,170-175H2,1-13H3,(H2,176,228)(H2,177,229)(H2,178,230)(H,183,189)(H,186,237)(H,187,238)(H,188,262)(H,190,247)(H,191,233)(H,192,232)(H,193,234)(H,194,240)(H,195,245)(H,196,249)(H,197,253)(H,198,252)(H,199,258)(H,200,264)(H,201,259)(H,202,242)(H,203,254)(H,204,246)(H,205,241)(H,206,265)(H,207,263)(H,208,239)(H,209,243)(H,210,244)(H,211,248)(H,212,251)(H,213,260)(H,214,231)(H,215,250)(H,216,255)(H,217,261)(H,218,256)(H,219,257)(H,235,236)(H,270,271)(H4,179,180,184)(H4,181,182,185) |
| Chemical Name | 6-amino-2-[[1-[2-[[47-[[2-[[2-[[4-amino-2-[[2-[[1-[2-[(2-aminoacetyl)amino]-3-methylbutanoyl]pyrrolidine-2-carbonyl]amino]-3-methylpentanoyl]amino]-4-oxobutanoyl]amino]-3-methylbutanoyl]amino]-3-hydroxypropanoyl]amino]-15,39,75,92-tetrakis(4-aminobutyl)-81-(2-amino-2-oxoethyl)-65-(3-amino-3-oxopropyl)-33-benzyl-89-butan-2-yl-30,78-bis(3-carbamimidamidopropyl)-18-(carboxymethyl)-50-(1-hydroxyethyl)-56-(hydroxymethyl)-4-(1H-imidazol-4-ylmethyl)-21-methyl-27,84-bis(2-methylsulfanylethyl)-2,5,13,16,19,22,25,28,31,34,37,40,48,51,54,57,63,66,74,77,80,83,86,87,90,93,99-heptacosaoxo-9,10,44,45,70,71-hexathia-a,3,6,14,17,20,23,26,29,32,35,38,41,49,52,55,58,64,67,73,76,79,82,85,88,91,94-heptacosazapentacyclo[40.30.14.1412,68.058,62.094,98]hectane-7-carbonyl]amino]-3-hydroxybutanoyl]pyrrolidine-2-carbonyl]amino]hexanoic acid |
| Synonyms | Agitoxin 2; Agitoxin-2; 168147-41-9; Agtx 2 (scorpion); Agitoxin 2 (reduced); 155646-22-3; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Voltage-dependent K⁺ channel (KV1.3) (IC50: 1.2 nM in cell-free assays) [1] |
| ln Vitro |
- KV1.3 channel inhibition: Agitoxin-2 potently blocks KV1.3 channels in cell-free assays with an IC50 of 1.2 nM. It shows high selectivity for KV1.3 over other K⁺ channel subtypes (e.g., KV1.1, KV1.2) [1]. - Recombinant expression and functional validation: In Pichia pastoris, Agitoxin-2 was recombinantly expressed and purified. Electrophysiological assays confirmed its ability to inhibit KV1.3 channels with similar potency to native toxin [2]. |
| Enzyme Assay |
KV1.3 channel activity assay: Xenopus oocytes expressing KV1.3 channels were voltage-clamped using the two-electrode voltage-clamp technique. Agitoxin-2 (0.1–100 nM) was applied to the bath solution, and K⁺ currents were recorded. IC50 was determined by fitting dose-response curves [1]. |
| Cell Assay |
KV1.3-mediated current inhibition: HEK293 cells transfected with KV1.3 cDNA were patch-clamped in the whole-cell configuration. Agitoxin-2 (0.1–100 nM) was perfused, and K⁺ currents were measured. Inhibition kinetics and potency were analyzed [1]. |
| References |
[1]. Purification and characterization of three inhibitors of voltage-dependent K+ channels from Leiurus quinquestriatus var. hebraeus venom. Biochemistry. 1994 Jun 7;33(22):6834-9. [2]. Recombinant expression of margatoxin and agitoxin-2 in Pichia pastoris: an efficient method for production of KV1.3 channel blockers. PLoS One. 2012;7(12):e52965. |
| Additional Infomation |
- Mechanism of action: Agitoxin-2 acts as a pore-blocking inhibitor of KV1.3 channels, binding to the outer vestibule and preventing K⁺ ion efflux [1]. - Production method: Recombinant Agitoxin-2 expressed in Pichia pastoris offers a scalable and cost-effective alternative to venom extraction [2]. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.2444 mL | 1.2222 mL | 2.4445 mL | |
| 5 mM | 0.0489 mL | 0.2444 mL | 0.4889 mL | |
| 10 mM | 0.0244 mL | 0.1222 mL | 0.2444 mL |