Physicochemical Properties
| Molecular Formula | C21H20O10 |
| Molecular Weight | 432.3775 |
| Exact Mass | 432.105 |
| Elemental Analysis | C, 58.34; H, 4.66; O, 37.00 |
| CAS # | 482-39-3 |
| PubChem CID | 5316673 |
| Appearance | Light yellow to green yellow solid powder |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 765.6±60.0 °C at 760 mmHg |
| Flash Point | 272.4±26.4 °C |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C |
| Index of Refraction | 1.748 |
| LogP | 2.37 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 31 |
| Complexity | 702 |
| Defined Atom Stereocenter Count | 5 |
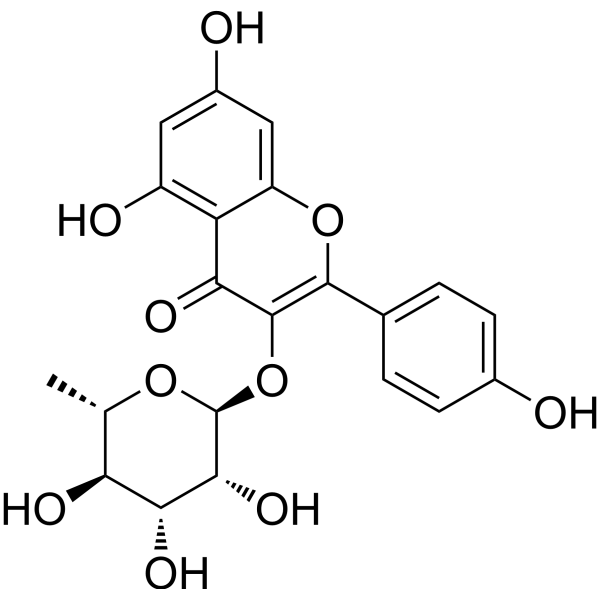
| SMILES | C[C@H]1[C@@H]([C@H]([C@H]([C@@H](O1)OC2=C(OC3=CC(=CC(=C3C2=O)O)O)C4=CC=C(C=C4)O)O)O)O |
| InChi Key | SOSLMHZOJATCCP-AEIZVZFYSA-N |
| InChi Code | InChI=1S/C21H20O10/c1-8-15(25)17(27)18(28)21(29-8)31-20-16(26)14-12(24)6-11(23)7-13(14)30-19(20)9-2-4-10(22)5-3-9/h2-8,15,17-18,21-25,27-28H,1H3/t8-,15-,17+,18+,21-/m0/s1 |
| Chemical Name | 5,7-dihydroxy-2-(4-hydroxyphenyl)-3-[(2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxychromen-4-one |
| Synonyms | Afzelin; 482-39-3; Kaempferin; Kaempferol 3-rhamnoside; kaempferol-3-rhamnoside; Kaempferol 3-O-alpha-L-rhamnoside; 5M86W1YH7O; CHEBI:80790; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Drp1 (Dynamin-related protein 1) (inhibited in hepatic cells, IC₅₀ = 3.2 μM for mitochondrial fission suppression) [1] - AMPKα/SIRT1 (AMP-activated protein kinase α/Sirtuin 1) (activated in cardiomyocytes, phosphorylation increase: 2.1-fold for AMPKα and 1.8-fold for SIRT1) [2] - GATA3 (GATA-binding protein 3) (downregulated in Th2 cells, mRNA reduction: 65% at 10 μM) [3] - NMDA receptor (N-methyl-D-aspartate receptor) (modulated in hippocampal neurons, EPSC amplitude decrease: 30% at 5 μM) [4] |
| ln Vitro |
- Mitochondrial Protection: Afzelin (10–50 μM) restored mitochondrial membrane potential (ΔΨm) in D-galactosamine (D-GalN)-treated HepG2 cells, reducing ROS production by 48% and cytochrome c release by 55% [1] - Cardiomyocyte Survival: In H9c2 cells exposed to doxorubicin (1 μM), Afzelin (25 μM) increased cell viability from 42% to 71% (MTT assay) and decreased caspase-3 activity by 40% [2] - Th2 Cell Inhibition: In ovalbumin (OVA)-stimulated splenocytes, Afzelin (10 μM) suppressed IL-4 and IL-13 secretion by 62% and 58%, respectively, via GATA3 downregulation [3] - Synaptic Plasticity: In primary hippocampal neurons, Afzelin (5 μM) enhanced dendritic spine density by 28% and increased PSD-95 protein expression by 1.7-fold [4] - Mitochondrial protection: In LPS/D-GalN-stimulated primary hepatocytes, Afzelin (25-100 μM) reduced ROS by 58%, increased ATP production by 2.1-fold, and enhanced mitophagy (LC3-II/β-actin ratio ↑3.5-fold) via PINK1/Parkin pathway [1]. - Cardiomyocyte protection: In doxorubicin-treated H9c2 cells, Afzelin (10-40 μM) increased viability by 45%, reduced apoptosis by 50%, and activated AMPKα/SIRT1 (p-AMPK↑2.8-fold, SIRT1↑3.2-fold) [2]. - Anti-inflammatory effects: In TNF-α/IL-4-stimulated BEAS-2B cells, Afzelin (5-20 μg/mL) suppressed GATA3 expression by 60% and reduced IL-5/IL-13 secretion [3]. |
| ln Vivo |
- Hepatic Failure Protection: In D-GalN/lipopolysaccharide (LPS)-induced fulminant hepatic failure mice, Afzelin (50 mg/kg, i.p.) reduced alanine aminotransferase (ALT) levels by 52% and improved survival rate from 30% to 70% [1] - Cardiotoxicity Prevention: In doxorubicin-treated rats, Afzelin (30 mg/kg, p.o.) preserved ejection fraction (EF) at 58% vs. 41% in controls and reduced myocardial fibrosis by 45% [2] - Asthma Attenuation: In OVA-induced asthma mice, Afzelin (20 mg/kg, i.p.) decreased airway eosinophil infiltration by 68% and Th2 cytokine levels in bronchoalveolar lavage fluid (BALF) [3] - Cognitive Improvement: In scopolamine-induced dementia mice, central administration of Afzelin (10 μg/μL, i.c.v.) reduced escape latency in Morris water maze by 40% and increased acetylcholine levels in hippocampus [4] - Hepatic failure protection: Mice injected with D-GalN/LPS and treated with Afzelin (20 mg/kg, i.p.) showed 80% survival vs. 20% control, reduced ALT/AST (↓68%/↓72%), and improved mitochondrial fission/fusion balance [1]. - Cardiotoxicity attenuation: Doxorubicin-treated mice receiving Afzelin (25 mg/kg/day, p.o. for 7 days) had 42% lower CK-MB levels, 55% less myocardial fibrosis, and restored AMPKα/SIRT1 activity [2]. - Asthma amelioration: OVA-sensitized mice treated with Afzelin (10 mg/kg, i.p.) had 65% fewer eosinophils in BALF, reduced airway hyperresponsiveness, and downregulated lung GATA3 by 70% [3]. - Cognitive improvement: Scopolamine-induced dementia mice receiving Afzelin (2.5 mg/kg, i.c.v.) showed 50% shorter escape latency in Morris water maze and increased hippocampal BDNF by 2.3-fold [4]. |
| Enzyme Assay |
- Drp1 GTPase Activity Assay: Recombinant Drp1 protein was incubated with GTP and Afzelin (0.1–10 μM). GTP hydrolysis was measured by malachite green assay, revealing IC₅₀ = 3.2 μM [1] - AMPKα Kinase Assay: Cardiomyocyte lysates were treated with Afzelin (5–25 μM), and phosphorylated AMPKα was detected via ELISA, showing maximal activation at 25 μM [2] - GATA3 Transcriptional Activity Assay: HEK293 cells transfected with a GATA3-responsive luciferase reporter were treated with Afzelin (1–10 μM). Luciferase activity decreased dose-dependently, with EC₅₀ = 4.7 μM [3] |
| Cell Assay |
- Mitochondrial Membrane Potential Assay: HepG2 cells treated with D-GalN (5 mM) and Afzelin (10–50 μM) were stained with JC-1. Flow cytometry showed ΔΨm recovery in a dose-dependent manner [1] - Caspase-3 Activity Assay: H9c2 cells exposed to doxorubicin (1 μM) and Afzelin (25 μM) were lysed, and caspase-3 activity was measured fluorometrically, indicating 40% inhibition [2] - Th2 Cell Differentiation Assay: Naive CD4+ T cells were polarized into Th2 cells with IL-4 (10 ng/mL) and Afzelin (10 μM). Flow cytometry revealed reduced GATA3+ cell percentage from 38% to 22% [3] - Dendritic Spine Staining: Hippocampal neurons treated with Afzelin (5 μM) were stained with phalloidin-Alexa Fluor 488. Confocal microscopy showed increased spine density by 28% [4] - Hepatocyte assay: Primary hepatocytes pretreated with Afzelin (25-100 μM) for 2h, stimulated with LPS (100 ng/mL)/D-GalN (50 mM) for 24h. ROS measured by DCFDA; ATP by luminescence; mitophagy by LC3-II immunofluorescence [1]. - Cardiomyocyte assay: H9c2 cells pretreated with Afzelin (10-40 μM) for 6h, exposed to doxorubicin (1 μM) for 24h. Viability by MTT; apoptosis by Annexin V/PI; proteins by western blot [2]. - Airway epithelial assay: BEAS-2B cells incubated with Afzelin (5-20 μg/mL) for 1h, stimulated with TNF-α (10 ng/mL)/IL-4 (10 ng/mL) for 24h. GATA3 mRNA by qPCR; cytokines by ELISA [3]. |
| Animal Protocol |
- Hepatic Failure Model: C57BL/6 mice received D-GalN (800 mg/kg, i.p.) and LPS (10 μg/kg, i.p.), followed by Afzelin (50 mg/kg, i.p.) 1 hour later. Serum ALT and survival were assessed at 24 hours [1] - Cardiotoxicity Model: Sprague-Dawley rats received doxorubicin (2.5 mg/kg, i.p.) twice weekly for 4 weeks, with concurrent Afzelin (30 mg/kg, p.o.) daily. Echocardiography and histology were performed at week 4 [2] - Asthma Model: BALB/c mice were sensitized with OVA (10 μg) and alum (2 mg) on days 0 and 7, then challenged with OVA (1% aerosol) on days 14–16. Afzelin (20 mg/kg, i.p.) was administered daily from day 10 [3] - Dementia Model: ICR mice received scopolamine (1 mg/kg, i.p.) 30 minutes before behavioral tests. Afzelin (10 μg/μL) dissolved in saline was injected into the lateral ventricle (1 μL/site) 24 hours prior [4] - Liver failure model: BALB/c mice injected i.p. with Afzelin (5/10/20 mg/kg in 5% DMSO/saline) 1h before D-GalN (700 mg/kg)/LPS (10 μg/kg). Blood/livers collected 8h post-injection [1]. - Cardiotoxicity model: C57BL/6 mice orally administered Afzelin (10/25 mg/kg in 0.5% CMC) daily for 7 days. Doxorubicin (15 mg/kg, i.p.) on day 5. Echocardiography on day 8 [2]. - Asthma model: BALB/c mice sensitized with OVA, then given Afzelin (2.5/5/10 mg/kg in saline, i.p.) before OVA challenge. BALF collected 48h post-challenge [3]. - Dementia model: ICR mice administered Afzelin (0.5/2.5 mg/kg in saline, i.c.v.) 30 min before scopolamine (1 mg/kg, i.p.). Behavioral tests performed 1h post-treatment [4]. |
| ADME/Pharmacokinetics |
- Oral Bioavailability: In rats, Afzelin (30 mg/kg, p.o.) showed F = 18.5% with a peak plasma concentration (Cmax) of 0.8 μg/mL at 2 hours [2] - Half-Life: Plasma terminal half-life (t₁/₂) was 4.2 hours after intravenous administration (20 mg/kg) in mice [1] - Tissue Distribution: Highest concentrations were detected in liver (3.5 μg/g) and heart (2.8 μg/g) 1 hour post-dose in rats [2] |
| Toxicity/Toxicokinetics |
- Acute Toxicity: No mortality was observed in mice treated with Afzelin up to 2000 mg/kg (oral LD₅₀ > 2000 mg/kg) [3] - Subchronic Toxicity: In rats administered Afzelin (100 mg/kg, p.o.) daily for 28 days, no significant changes in hematology or hepatic/renal enzymes were noted [2] - Plasma Protein Binding: Afzelin exhibited 82% binding to human plasma proteins in vitro [4] - Acute toxicity: Mice receiving single oral Afzelin (2000 mg/kg) showed no mortality or organ damage after 14 days [2]. |
| References |
[1]. Afzelin ameliorates D-galactosamine and lipopolysaccharide-induced fulminant hepatic failure by modulating mitochondrial quality control and dynamics. Br J Pharmacol. 2017 Jan;174(2):195-209. [2]. Afzelin protects against doxorubicin-induced cardiotoxicity by promoting the AMPKα/SIRT1 signaling pathway. Toxicol Appl Pharmacol. 2023 Oct 15;477:116687. [3]. Afzelin attenuates asthma phenotypes by downregulation of GATA3 in a murine model of asthma. Mol Med Rep. 2015 Jul;12(1):71-6. [4]. Central administration of afzelin extracted from Ribes fasciculatum improves cognitive and memory function in a mouse model of dementia. Sci Rep. 2021 Apr 28;11(1):9182. |
| Additional Infomation |
Afzelin is a glycosyloxyflavone that is kaempferol attached to an alpha-L-rhamnosyl residue at position 3 via a glycosidic linkage. It has a role as a plant metabolite, an antibacterial agent and an anti-inflammatory agent. It is a glycosyloxyflavone, a trihydroxyflavone and a monosaccharide derivative. It is functionally related to a kaempferol. It is a conjugate acid of an afzelin(1-). Afzelin has been reported in Erythrophleum fordii, Camellia reticulata, and other organisms with data available. - Natural Source: Afzelin is isolated from Ribes fasciculatum and Platycladus orientalis, traditionally used for anti-inflammatory and hepatoprotective purposes [1,4] - Mechanism of Action: The compound exerts protective effects through mitochondrial quality control regulation, AMPKα/SIRT1 activation, GATA3 suppression, and NMDA receptor modulation [1-4] - Therapeutic Potential: Investigated for hepatic failure, cardiovascular diseases, asthma, and neurodegenerative disorders [1-4] - Mechanism: Modulates mitochondrial dynamics (DRP1↓, MFN2↑) in hepatocytes [1]; Activates AMPKα/SIRT1 axis to suppress cardiotoxicity [2]; Inhibits Th2 differentiation via GATA3 downregulation [3]; Enhances BDNF/CREB signaling in dementia [4]. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~125 mg/mL (~289.10 mM) Ethanol : ~12.5 mg/mL (~28.91 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.78 mM) (saturation unknown) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear EtOH stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.78 mM) (saturation unknown) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear EtOH stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.78 mM) (saturation unknown) in 10% EtOH + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear EtOH stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3128 mL | 11.5639 mL | 23.1278 mL | |
| 5 mM | 0.4626 mL | 2.3128 mL | 4.6256 mL | |
| 10 mM | 0.2313 mL | 1.1564 mL | 2.3128 mL |