Physicochemical Properties
| Molecular Formula | C17H12O7 |
| Molecular Weight | 328.27 |
| Exact Mass | 328.058 |
| CAS # | 52819-96-2 |
| PubChem CID | 104757 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.767 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 24 |
| Complexity | 680 |
| Defined Atom Stereocenter Count | 3 |
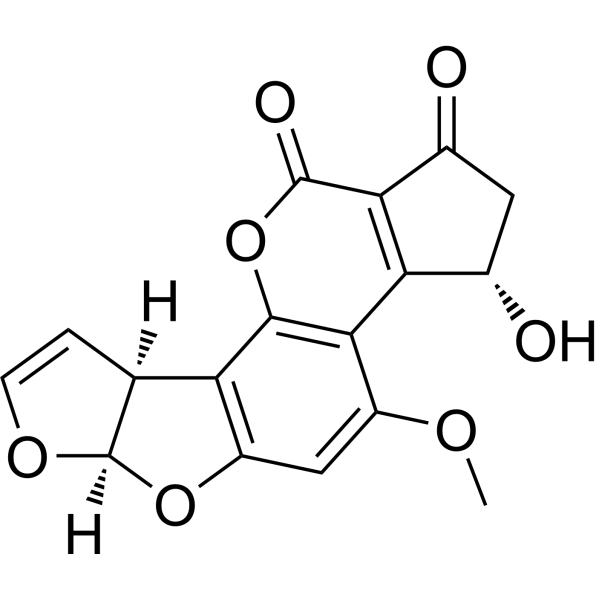
| SMILES | COc1cc2c(c3c1c4c(c(=O)o3)C(=O)C[C@@H]4O)[C@@H]5C=CO[C@@H]5O2 |
| InChi Key | GYNOTJLCULOEIM-XKRJZGAWSA-N |
| InChi Code | InChI=1S/C17H12O7/c1-21-9-5-10-11(6-2-3-22-17(6)23-10)15-14(9)12-7(18)4-8(19)13(12)16(20)24-15/h2-3,5-7,17-18H,4H2,1H3/t6-,7-,17+/m0/s1 |
| Chemical Name | (3S,7R,14S)-14-hydroxy-11-methoxy-6,8,19-trioxapentacyclo[10.7.0.02,9.03,7.013,17]nonadeca-1,4,9,11,13(17)-pentaene-16,18-dione |
| Synonyms | Aflatoxin Q1; 52819-96-2; (3S,7R,14S)-14-hydroxy-11-methoxy-6,8,19-trioxapentacyclo[10.7.0.02,9.03,7.013,17]nonadeca-1,4,9,11,13(17)-pentaene-16,18-dione; CCRIS 2010; (3S,6aR,9aS)-2,3,6a,9a-Tetrahydro-3-hydroxy-4-methoxycyclopenta[c]furo[3',2':4,5]furo[2,3-h][1]benzopyran-1,11-dione; [3S-(3a,6aa,9aa)]-2,3,6a,9a-Tetrahydro-3-hydroxy-4-methoxycyclopenta[c]furo[3',2':4,5]furo[2,3-h][1]benzopyran-1,11-dione; AFQ1; CHEBI:78582; DTXSID60967286; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Mycotoxin found in Aspergillus flavus |
| ln Vitro |
Aflatoxin Q1 8,9-oxide was synthesized and found to yield lower levels of N7-guanyl adducts than obtained from aflatoxin B1 8,9-oxide when mixed with calf thymus DNA or Salmonella typhimurium TA 98 cells. However, when S. typhimurium TA 98 was treated with the (analogous) epoxides of aflatoxin B1, aflatoxin G1, aflatoxin Q1, or sterigmatocystin, the ratios of revertants to N7-guanyl DNA adducts were similar. Aflatoxin Q1 and aflatoxin B1 8,9-oxide (trapped here as the glutathione conjugate) are the major oxidative products formed from aflatoxin B1 at all substrate concentrations in human liver microsomes, and cytochrome P-450 (P-450) 3A4 appears to be the dominant enzyme involved in both oxidations, as judged by studies involving correlation of activities in different liver samples, chemical inhibition, immunoinhibition, and reconstitution with purified hepatic and yeast recombinant P-450 3A4. Aflatoxin Q1 is not appreciably oxidized in human liver microsomes and is not very genotoxic. The postulated formation of both aflatoxin Q1 and aflatoxin 8,9-oxide from aflatoxin B1 can be rationalized by a model in which P-450 3A4 binds the substrate in either of two different configurations. This is further demonstrated by the dichotomous effect of 7,8-benzoflavone--this flavone stimulates 8,9-epoxidation while inhibiting the 3 alpha-hydroxylation reaction to form aflatoxin Q1. Thus, the 3 alpha-hydroxylation of aflatoxin B1 to aflatoxin Q1 is viewed as a potentially significant detoxication pathway [1]. Aflatoxin B1 (AFB1) is a mycotoxin produced by Aspergillus flavus (A. flavus). AFB1 is reported to have high thermal stability and is not decomposed by heat treatment during food processing. Therefore, in this study, knowing that AFB1 is metabolized by cytochrome P450 (CYP), our aim was to develop a method to detoxify A. flavus-contaminated maize, under normal temperature and pressure, using Escherichia coli expressing human CYP3A4. First, the metabolic activity of AFB1 by recombinant human CYP3A4 was evaluated. As a result, we confirmed that recombinant human CYP3A4 metabolizes 98% of AFB1. Next, we found that aflatoxin Q1, a metabolite of AFB1 was no longer mutagenic. Furthermore, we revealed that about 50% of the AFB1 metabolic activity can be maintained for 3 months when E. coli expressing human CYP3A4 is freeze-dried in the presence of trehalose. Finally, we found that 80% of AFB1 in A. flavus-contaminated maize was metabolized by E. coli expressing human CYP3A4 in the presence of surfactant triton X-405 at a final concentration of 10% (v/v). From these results, we conclude that AFB1 in A. flavus-contaminated maize can be detoxified under normal temperature and pressure by using E. coli expressing human CYP3A4 [2]. |
| Enzyme Assay |
Identification of Human CYP3A4 AFB1 Metabolites [2] In order to identify the resulting metabolites following human CYP3A4 metabolism of AFB1, the HPLC peak having a retention time of 12 to 13 min which corresponded to the metabolite was collected and analyzed by LC- MS (Fig. 3B). The [M+H]+ ions were detected at m/z 329 (Fig. 3C) and used as parent ions for Aflatoxin Q1 (AFQ1). We found that the main metabolite identified in this E. coli CYP3A4 expression system was AFQ1. AFB1 exo-8,9- epoxide easily intercalates into DNA and protein. Johnson et al. reported that AFB1 exo-8,9-epoxide reacts spontaneously with H2O at a pseudo-first order rate of 0.6 s-1 at 25°C. Therefore, it is considered that AFB1 exo-8,9-epoxide was not observed as a metabolite. Furthermore, it was thought that AFBO was not detected due to the low amount of AFB1 exo-8,9-epoxide produced by CYP3A4. This is consistent with previous studies which have shown that AFB1 is mainly metabolized to AFQ1 by CYP3A4. |
| Toxicity/Toxicokinetics |
Evaluation of Mutagenicity of Human CYP3A4 AFB1 Metabolites [2] Researchers evaluated the mutagenicity of human CYP3A4 AFB1 metabolites using the umu-test. They found that mutagenicity occurred with 16 μM AFB1 but not with the metabolite, Aflatoxin Q1 (AFQ1) (Table 1). Our results show that AFQ1 produced by metabolism of AFB1 by E. coli expressing human CYP3A4 is not mutagenic, even without undergoing additional metabolic activation. Therefore, E. coli expressing human CYP3A4 has the detoxifying effect of AFB1. |
| References |
[1]. Raney KD, et al. Oxidation of aflatoxins and sterigmatocystin by human liver microsomes: significance of aflatoxin Q1 as a detoxication product of aflatoxin B1. Chem Res Toxicol. 1992 Mar-Apr;5(2):202-10. [2]. Yamada M, et al. Detoxification of Aflatoxin B1 Contaminated Maize Using Human CYP3A4. J Microbiol Biotechnol. 2020 Aug 28;30(8):1207-1213. |
| Additional Infomation |
Aflatoxin Q1 is a member of the class of aflatoxins that is aflatoxin B1 in which the hydrogen at the pro-3S position is replaced by a hydroxy group. It has a role as a human xenobiotic metabolite and a carcinogenic agent. It is an aflatoxin, an aromatic ether and an aromatic ketone. It is functionally related to an aflatoxin B1. Aflatoxin Q1 has been reported in Aspergillus flavus with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0463 mL | 15.2314 mL | 30.4627 mL | |
| 5 mM | 0.6093 mL | 3.0463 mL | 6.0925 mL | |
| 10 mM | 0.3046 mL | 1.5231 mL | 3.0463 mL |