Physicochemical Properties
| Molecular Formula | C17H14O6 |
| Molecular Weight | 314.28946 |
| Exact Mass | 314.079 |
| CAS # | 29611-03-8 |
| PubChem CID | 104744 |
| Appearance | White to off-white solid powder |
| Density | 1.56g/cm3 |
| Boiling Point | 552.1ºC at 760mmHg |
| Flash Point | 210.2ºC |
| Vapour Pressure | 5.02E-13mmHg at 25°C |
| Index of Refraction | 1.698 |
| LogP | 2.127 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 23 |
| Complexity | 610 |
| Defined Atom Stereocenter Count | 3 |
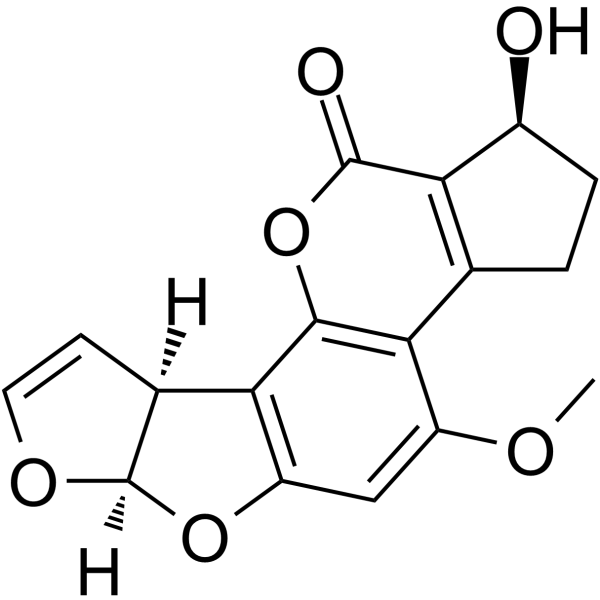
| SMILES | COC1C=C2O[C@H]3OC=C[C@H]3C2=C2OC(C3C(O)CCC=3C=12)=O |
| InChi Key | WYIWLDSPNDMZIT-IRWWLHRVSA-N |
| InChi Code | InChI=1S/C17H14O6/c1-20-10-6-11-14(8-4-5-21-17(8)22-11)15-13(10)7-2-3-9(18)12(7)16(19)23-15/h4-6,8-9,17-18H,2-3H2,1H3/t8-,9-,17+/m0/s1 |
| Chemical Name | (3S,7R,16S)-16-hydroxy-11-methoxy-6,8,19-trioxapentacyclo[10.7.0.02,9.03,7.013,17]nonadeca-1,4,9,11,13(17)-pentaen-18-one |
| Synonyms | AFLATOXICOL; 29611-03-8; Aflatoxin Ro; Aflatoxin R0; Aflatoxicol natural epimer; 45I1K5482V; (3S,7R,16S)-16-hydroxy-11-methoxy-6,8,19-trioxapentacyclo[10.7.0.02,9.03,7.013,17]nonadeca-1,4,9,11,13(17)-pentaen-18-one; Cyclopenta(c)furo(3',2':4,5)furo(2,3-h)(1)benzopyran-11(1H)-one, 2,3,6a,9a-tetrahydro-1-hydroxy-4-methoxy-, (1S,6aR,9aS)-; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Secondary metabolite |
| ln Vitro |
Four fungal strains, namely, Aspergillus niger, Eurotium herbariorum, a Rhizopus sp., and non-aflatoxin (AF)-producing Aspergillus flavus, which could convert AF-B1 to aflatoxicol (AFL), could also reconvert AFL to AF-B1. The interconversion of AF-B1 to AFL and of AFL to AF-B1 was ascertained to occur during proliferation of the fungi. These reactions were distinctly observed in cell-free systems obtained from disrupted mycelia of A. flavus and the Rhizopus sp., but they were not observed in culture filtrates from intact (nondisrupted) mycelia of the same strains. The interconversion activities of AF-B1 and AFL were not observed when the cell-free systems were preheated at 100 degrees C. These findings strongly suggest that the interconversion of AF-B1 and AFL is mediated by intracellular enzymes of A. flavus and the Rhizopus sp. In addition, the isomerization of AFL-A to AFL-B observed in culture medium was also found to occur by the lowering of the culture pH. [1] Between 1996 and 1998, 580 litres of milk in Mexico were surveyed for aflatoxin B(1) (AFB(1)) and its metabolite aflatoxicol (AFL), which are mutagenic and carcinogenic mycotoxins that interconvert AFB(1)-AFL-AFB(1). The seven most consumed brands from different regions of Mexico included pasteurized and ultrapasteurized milk with four different fat levels: whole fat (28-33 g l(-1)), half-skimmed (10-20 g l(-1)), light (1-4 g l(-1)) and with vegetable oil (33 g l(-1)). Aflatoxins in each sample were concentrated with total aflatoxin immunoaffinity columns and quantitated by high-performance liquid chromatography. A milk sample was considered contaminated if it contained >/=0.05 microg l(-1) AFL. Pasteurization and ultrapasteurization of milk did not control contamination with AFL, which was present in 13% of samples at >/=0.05 microg l(-1) and in 8% at >/=0.5 microg l(-1), with a range of AFL from 0 to 12.4 microg l(-1). AFB(1) was present mainly in traces (0-0.4 microg l(-1)). The safest milk in relation to AFL contamination was imported milk powder with vegetable oil. There was a significant correlation between contamination of milk with AFL and the autumn (p<0.0002); the fat content was not significant[2]. |
| References |
[1]. Interconversion of aflatoxin B1 and aflatoxicol by several fungi. Appl Environ Microbiol. 1990 May;56(5):1465-70. [2]. Aflatoxin B1 and its interconverting metabolite aflatoxicol in milk: the situation in Mexico. Food Addit Contam. 2003 Nov;20(11):1077-86. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1818 mL | 15.9089 mL | 31.8177 mL | |
| 5 mM | 0.6364 mL | 3.1818 mL | 6.3635 mL | |
| 10 mM | 0.3182 mL | 1.5909 mL | 3.1818 mL |