Physicochemical Properties
| Molecular Formula | C23H24N3NA2O7P |
| Molecular Weight | 531.41 |
| Exact Mass | 529.099 |
| CAS # | 1518800-29-7 |
| Related CAS # | Afabicin;1518800-35-5; Afabicin disodium;1518800-29-7; 1518800-40-2 (potassium); 1518800-53-7 (calcium); 1518800-36-6 (diolamine) |
| PubChem CID | 72704026 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 36 |
| Complexity | 822 |
| Defined Atom Stereocenter Count | 0 |
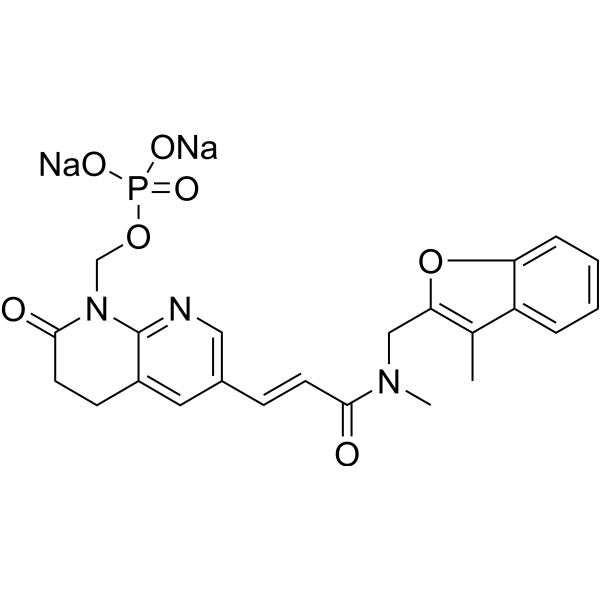
| SMILES | CC1=C(OC2=CC=CC=C12)CN(C)C(=O)/C=C/C3=CC4=C(N=C3)N(C(=O)CC4)COP(=O)([O-])[O-].[Na+].[Na+] |
| InChi Key | ZAWLQRSRUJJWHT-OJYIHNBOSA-L |
| InChi Code | InChI=1S/C23H24N3O7P.2Na/c1-15-18-5-3-4-6-19(18)33-20(15)13-25(2)21(27)9-7-16-11-17-8-10-22(28)26(23(17)24-12-16)14-32-34(29,30)31;;/h3-7,9,11-12H,8,10,13-14H2,1-2H3,(H2,29,30,31);;/q;2*+1/p-2/b9-7+;; |
| Chemical Name | disodium;[6-[(E)-3-[methyl-[(3-methyl-1-benzofuran-2-yl)methyl]amino]-3-oxoprop-1-enyl]-2-oxo-3,4-dihydro-1,8-naphthyridin-1-yl]methyl phosphate |
| Synonyms | Afabicin disodium; 1518800-29-7; disodium;[6-[(E)-3-[methyl-[(3-methyl-1-benzofuran-2-yl)methyl]amino]-3-oxoprop-1-enyl]-2-oxo-3,4-dihydro-1,8-naphthyridin-1-yl]methyl phosphate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Enoyl-acyl carrier protein reductase (FabI) (IC50: 0.002 μM in enzyme inhibition assays) [1] |
| ln Vitro |
- Antibacterial Activity: Afabicin (Debio 1450) demonstrated potent activity against Staphylococcus aureus and coagulase-negative Staphylococcus spp., including multidrug-resistant (MDR) strains. The MIC90 values were 0.06–0.12 μg/mL for methicillin-resistant S. aureus (MRSA) and 0.03–0.06 μg/mL for vancomycin-intermediate S. aureus (VISA) [1] - Mechanism of Action: By inhibiting FabI, Debio 1450 disrupts bacterial fatty acid biosynthesis, leading to cell membrane dysfunction and growth arrest [1] - Synergy with β-Lactams: Combinations with oxacillin showed synergistic effects against MRSA, reducing the MIC by 8- to 16-fold [1] As a prodrug of Debio1452, abafacin (Debio1450) primarily acts against Staphylococcus aureus and has negligible efficacy against other Gram-positive or Gram-negative bacterial species. An enzyme essential for staphylococcal fatty acid production, FabI, is inhibited by debio1452. The anti-Staphylococcus isolates, Debio1452, exhibits high activity with MIC50 and MIC90 values of 0.015 μg/mL and 0.12 μg/mL, respectively. Staphylococcus aureus is likewise inhibited by Debio1452, with a MIC ≤0.12 μg/mL and corresponding MIC50 and MIC90 values of 0.004 μg/mL and 0.008 μg/mL [1]. |
| ln Vivo |
- Mouse Infection Model: Oral administration of Debio 1450 (25 mg/kg twice daily) significantly reduced bacterial burden in a murine model of S. aureus sepsis, with a 3-log reduction in colony-forming units (CFU) in blood and kidneys compared to vehicle control [1] - Pharmacodynamic Response: Treated mice exhibited dose-dependent suppression of FabI activity in infected tissues, correlating with reduced bacterial replication [1] |
| Enzyme Assay |
- FabI Kinetic Assay: Recombinant FabI from S. aureus was incubated with NADH (0.2 mM), crotonyl-ACP (50 μM), and Debio 1450 (0.001–1 μM) in Tris-HCl buffer (pH 7.5). The decrease in absorbance at 340 nm was monitored to measure NADH oxidation, yielding an IC50 of 0.002 μM [1] - Binding Affinity: Isothermal titration calorimetry (ITC) confirmed specific binding of Debio 1450 to FabI with a KD of 0.001 μM, stabilizing the enzyme-substrate complex [1] |
| Cell Assay |
- Bacterial Growth Inhibition: S. aureus ATCC 29213 was cultured in Mueller-Hinton broth with Debio 1450 (0.01–1 μg/mL). Optical density at 600 nm was measured hourly, showing 90% growth inhibition at 0.1 μg/mL after 6 hours [1] - Membrane Integrity Assay: Propidium iodide uptake increased 5-fold in Debio 1450-treated S. aureus cells, indicating loss of membrane integrity [1] |
| Animal Protocol |
- Model Setup: Female BALB/c mice (20–25 g) were intravenously infected with 1×10^7 CFU of S. aureus USA300. Debio 1450 (25 mg/kg) or vehicle (10% DMSO in PBS) was administered orally every 12 hours for 3 days [1] - Sample Collection: At 24 and 72 hours post-infection, blood, kidneys, and spleen were harvested for CFU enumeration and histopathological analysis [1] |
| ADME/Pharmacokinetics |
- Oral Bioavailability: In rats, Debio 1450 showed an oral bioavailability of 85%, with a peak plasma concentration (Cmax) of 2.8 μg/mL at 1 hour post-dose [1] - Half-life: The terminal elimination half-life was 6.5 hours in mice, determined by non-compartmental analysis of plasma concentration-time data [1] - Tissue Distribution: Highest drug concentrations were detected in kidneys (4.2-fold higher than plasma) and lungs (3.5-fold), consistent with urinary and respiratory tract infection indications [1] - Metabolism: Debio 1450 was primarily metabolized by hepatic CYP3A4, with no active metabolites identified [1] |
| Toxicity/Toxicokinetics |
- Acute Toxicity: The LD50 of Debio 1450 in mice exceeded 2000 mg/kg after oral administration, indicating low acute toxicity [1] - Subchronic Safety: A 4-week repeated-dose study in rats showed no significant changes in hematology, serum biochemistry, or histopathology at doses up to 100 mg/kg/day [1] - Plasma Protein Binding: >99% of Debio 1450 was bound to plasma proteins in human serum, primarily albumin [1] |
| References |
[1]. Activity of Debio1452, a FabI inhibitor with potent activity against Staphylococcus aureus and coagulase-negative Staphylococcus spp., including multidrug-resistant strains. Antimicrob Agents Chemother. 2015 May;59(5):2583-7. |
| Additional Infomation |
- Clinical Development: Afabicin (Debio 1450) is currently in phase II clinical trials for the treatment of MDR S. aureus infections, including ventilator-associated pneumonia and complicated skin and soft tissue infections [1] - Resistance Profile: No cross-resistance was observed with existing antibiotics (e.g., vancomycin, linezolid) in preclinical studies [1] - Mechanism of Resistance: Mutations in the fabI gene (e.g., A112V substitution) reduced Debio 1450 susceptibility by 4-fold, highlighting the need for combination therapies [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8818 mL | 9.4089 mL | 18.8179 mL | |
| 5 mM | 0.3764 mL | 1.8818 mL | 3.7636 mL | |
| 10 mM | 0.1882 mL | 0.9409 mL | 1.8818 mL |