Physicochemical Properties
| Molecular Formula | C22H36O2 |
| Molecular Weight | 332.52004 |
| Exact Mass | 332.271 |
| CAS # | 28874-58-0 |
| PubChem CID | 5497181 |
| Appearance | Colorless to light yellow liquid |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 439.5±24.0 °C at 760 mmHg |
| Flash Point | 336.2±18.0 °C |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.498 |
| LogP | 7.95 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 24 |
| Complexity | 389 |
| Defined Atom Stereocenter Count | 0 |
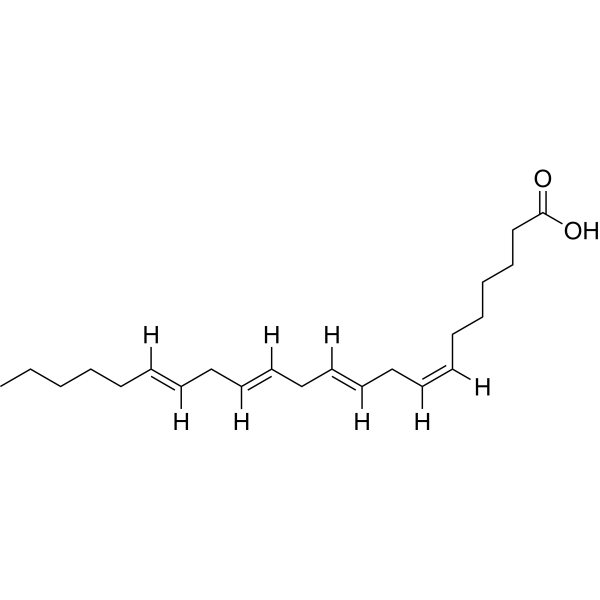
| SMILES | CCCCC/C=C/C/C=C/C/C=C/C/C=C\CCCCCC(O)=O |
| InChi Key | TWSWSIQAPQLDBP-DOFZRALJSA-N |
| InChi Code | InChI=1S/C22H36O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22(23)24/h6-7,9-10,12-13,15-16H,2-5,8,11,14,17-21H2,1H3,(H,23,24)/b7-6-,10-9-,13-12-,16-15- |
| Chemical Name | (7Z,10Z,13Z,16Z)-docosa-7,10,13,16-tetraenoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Nuclear factor-κB (NF-κB) signaling pathway [1] - Vascular smooth muscle cell potassium channels (for hyperpolarization) [2] - Pro-inflammatory cytokine (TNF-α, IL-6, IL-1β) expression regulatory targets [1] - Endothelial and zona glomerulosa hyperpolarization-related targets [2] |
| ln Vitro |
Arachidonic acid is elongated by ELOVL2 and ELOVL5, producing adenic acid. Interestingly, compared to db/db animals fed SD, db/db mice fed CDAHFD had hepatic free Adrenic Acid levels (22:4n6) that were considerably greater. The variation in the amounts of Adrenic Acid in plasma was found to be statistically significant (p<0.001). Additionally, it was discovered that there was a considerable rise in the concentration of phospholipid species that contained adrenic acid, such as PC or PE (40:4) (which contain side chains 18:0/22:4) and LPC 22:4 (sn-1/sn-2). (sn-1/sn-2). Adrenaline, an omega 6 polyunsaturated fatty acid (PUFA), was found to differ more in the liver and plasma of db/db mice fed CDAHFD than in db/db mice fed SD (8.6-fold vs. 1.6 times). - 2.2 times higher in plasma and 5.3 times higher in liver tissue than in arachidonic acid, a well-known pro-inflammatory polyunsaturated fatty acid [1]. - Adrenic Acid enhanced inflammation in human hepatoma HepG2 cells and mouse primary hepatocytes. At concentrations of 50, 100, and 200 μM, it dose-dependently increased the mRNA expression of TNF-α (1.8±0.2, 2.5±0.3, 3.7±0.4-fold), IL-6 (2.1±0.3, 3.2±0.4, 4.5±0.5-fold), and IL-1β (1.7±0.2, 2.3±0.3, 3.4±0.4-fold) compared to the control group [1] - It activated the NF-κB pathway in hepatocytes: 200 μM Adrenic Acid increased the phosphorylation of IκBα by 3.2±0.3-fold and nuclear translocation of NF-κB p65 by 2.8±0.3-fold [1] - In rat mesenteric artery endothelial cells (MAECs) and zona glomerulosa cells, Adrenic Acid (1–10 μM) induced concentration-dependent hyperpolarization. At 10 μM, it increased the hyperpolarization amplitude by 28±3 mV in MAECs and 25±2 mV in zona glomerulosa cells [2] - The compound enhanced large-conductance calcium-activated potassium (BKCa) channel currents in vascular smooth muscle cells, with a 2.4±0.2-fold increase at 10 μM [2] |
| ln Vivo |
- In high-fat diet (HFD)-induced non-alcoholic fatty liver disease (NAFLD) mice, intraperitoneal injection of Adrenic Acid (50 mg/kg twice weekly for 8 weeks) exacerbated liver inflammation. Hepatic TNF-α, IL-6, and IL-1β protein levels were increased by 68±7%, 75±8%, and 62±6% compared to HFD control mice [1] - It promoted hepatic steatosis in NAFLD mice: liver triglyceride content was increased by 45±5% at 50 mg/kg, and Oil Red O staining showed a significant increase in lipid droplet accumulation [1] - In normotensive rats, intravenous infusion of Adrenic Acid (1–10 μg/kg/min for 30 minutes) induced dose-dependent vasodilation of mesenteric arteries, with a maximum dilation rate of 42±4% at 10 μg/kg/min [2] - It reduced blood pressure in spontaneously hypertensive rats (SHRs): 10 μg/kg/min Adrenic Acid decreased systolic blood pressure by 22±3 mmHg [2] |
| Cell Assay |
- Hepatocyte inflammation assay: HepG2 cells and mouse primary hepatocytes were seeded in 6-well plates (2×10⁵ cells/well) and incubated overnight. Cells were treated with Adrenic Acid (50, 100, 200 μM) for 24 hours. Total RNA was extracted for RT-PCR to detect cytokine mRNA levels; cell lysates and nuclear fractions were prepared for Western blot analysis of NF-κB pathway proteins [1] - Vascular cell hyperpolarization assay: Rat MAECs and zona glomerulosa cells were cultured on glass coverslips. Adrenic Acid (1–10 μM) was applied, and membrane potential was recorded using patch-clamp technique. BKCa channel currents in vascular smooth muscle cells were measured under whole-cell patch-clamp configuration [2] |
| Animal Protocol |
- NAFLD mouse model: C57BL/6 mice were fed a HFD for 8 weeks to induce NAFLD, then randomly divided into HFD control group and Adrenic Acid treatment group (50 mg/kg i.p. twice weekly for 8 weeks). Mice were sacrificed, and liver tissues were collected for cytokine detection, triglyceride measurement, and histological staining [1] - Vasodilation and blood pressure assay: Normotensive rats and SHRs were anesthetized. Adrenic Acid was infused intravenously at 1–10 μg/kg/min for 30 minutes. Mesenteric artery vasodilation was measured using a myograph system, and systolic blood pressure was recorded via tail-cuff plethysmography [2] |
| References |
[1]. Adrenic acid as an inflammation enhancer in non-alcoholic fatty liver disease. Arch Biochem Biophys. 2017 Jun 1;623-624:64-75. [2]. Adrenic acid metabolites as endogenous endothelium-derived and zona glomerulosa-derived hyperpolarizing factors. Hypertension. 2010 Feb;55(2):547-54. |
| Additional Infomation |
All-cis-docosa-7,10,13,16-tetraenoic acid is the all-cis-isomer of a C22 polyunsaturated fatty acid having four double bonds in the 7-, 10-, 13- and 16-positions. One of the most abundant fatty acids in the early human brain. It has a role as a human metabolite and an algal metabolite. It is a docosatetraenoic acid and an omega-6 fatty acid. It is a conjugate acid of an all-cis-docosa-7,10,13,16-tetraenoate. Adrenic acid has been reported in Homo sapiens, Euglena gracilis, and Caenorhabditis elegans with data available. - Adrenic Acid (all-cis-7,10,13,16-docosatetraenoic acid) is a long-chain polyunsaturated fatty acid (LC-PUFA) [1][2] - Its pro-inflammatory mechanism in NAFLD involves activating the NF-κB pathway to upregulate pro-inflammatory cytokine expression, thereby exacerbating hepatic inflammation and steatosis [1] - It exerts vasodilatory and hypotensive effects by acting as a precursor of endothelium-derived and zona glomerulosa-derived hyperpolarizing factors, which enhance BKCa channel activity and induce vascular smooth muscle cell hyperpolarization [2] - Adrenic Acid is implicated in the pathogenesis of NAFLD as an inflammation enhancer and plays a regulatory role in vascular tone and blood pressure [1][2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0073 mL | 15.0367 mL | 30.0734 mL | |
| 5 mM | 0.6015 mL | 3.0073 mL | 6.0147 mL | |
| 10 mM | 0.3007 mL | 1.5037 mL | 3.0073 mL |