Physicochemical Properties
| Molecular Formula | C14H7CLF3NO5 |
| Molecular Weight | 361.66 |
| Exact Mass | 360.996 |
| CAS # | 50594-66-6 |
| Related CAS # | 62476-59-9 (hydrochloride salt) |
| PubChem CID | 44073 |
| Appearance | White to off-white solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 422.4±45.0 °C at 760 mmHg |
| Melting Point | 161ºC |
| Flash Point | 209.2±28.7 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.576 |
| LogP | 4.35 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 24 |
| Complexity | 484 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | NUFNQYOELLVIPL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H7ClF3NO5/c15-10-5-7(14(16,17)18)1-4-12(10)24-8-2-3-11(19(22)23)9(6-8)13(20)21/h1-6H,(H,20,21) |
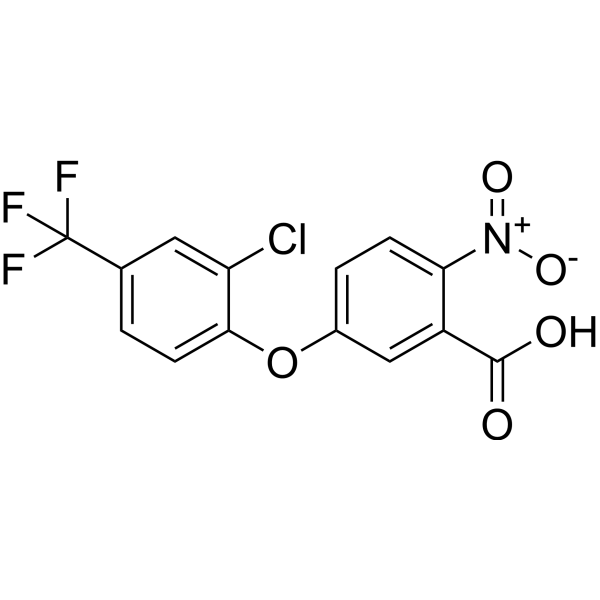
| Chemical Name | 5-[2-chloro-4-(trifluoromethyl)phenoxy]-2-nitrobenzoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Glutathione and ascorbate levels in cucumber (Cucumis sativus L.) cotyledon discs are examined in response to Acifluorfen in order to evaluate the connection between herbicide activity and endogenous antioxidants. Glutathione and ascorbate levels in discs exposed to less than 1.5 hours of white light (450 microeinsteins per square meter per second) are reduced by more than 50% when acifluorfen is used. Acifluorfen also causes far-red light-grown, photosynthetically inept plants to rapidly deplete their ascorbate stores[2]. |
| ln Vivo | Models of liver damage in animals can be created using acifluorfen in animal modeling. |
| References |
[1]. Involvement of Mouse Constitutive Androstane Receptor in Acifluorfen-Induced Liver Injury and Subsequent Tumor Development. Toxicol Sci. 2016;151(2):271-285. [2]. Effects of Acifluorfen on Endogenous Antioxidants and Protective Enzymes in Cucumber (Cucumis sativus L.) Cotyledons. Plant Physiol. 1985;79(3):862-866. |
| Additional Infomation |
Acifluorfen is a member of the class of benzoic acids that is 2-nitrobenzoic acid in which the hydrogen at position 5 is replaced by a 2-chloro-4-(trifluoromethyl)phenoxy group. It is a herbicide used for the post-emergence control of a variety of annual broadleaf weeds. It has a role as a herbicide, an agrochemical and an EC 1.3.3.4 (protoporphyrinogen oxidase) inhibitor. It is a member of benzoic acids, an organochlorine compound, an organofluorine compound, an aromatic ether, a monocarboxylic acid and a C-nitro compound. Acifluorfen is a protoporphyrinogen oxidase inhibitor. Acifluorfen is an herbicide. It is effective against broadleaf weeds and grasses and is used agriculturally on fields growing soybeans, peanuts, peas, and rice. Also registered as a spot treatment for residential use along driveways, sidewalks, and patios. acifluorfen has low acute toxicity via the oral, dermal, and inhalation routes of exposure, but causes severe eye irritation and moderate skin irritation. acifluorfen is currently classified as a B2 chemical carcinogen (probable human carcinogen). |
Solubility Data
| Solubility (In Vitro) | DMSO: 25 mg/mL (69.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (6.91 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.91 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7650 mL | 13.8251 mL | 27.6503 mL | |
| 5 mM | 0.5530 mL | 2.7650 mL | 5.5301 mL | |
| 10 mM | 0.2765 mL | 1.3825 mL | 2.7650 mL |