Physicochemical Properties
| Molecular Formula | C8H15NO6 |
| Molecular Weight | 221.209 |
| Exact Mass | 221.089 |
| CAS # | 7512-17-6 |
| Related CAS # | D-N-Acetylgalactosamine;1811-31-0;N-Acetyl-D-glucosamine-13C;253679-94-6;N-Acetyl-D-glucosamine-13C2,15N;478529-44-1;N-Acetyl-D-glucosamine-13C,15N-1;N-Acetyl-D-glucosamine-13C6;1194446-34-8;N-Acetyl-D-glucosamine-13C-1;478518-87-5;N-Acetyl-D-glucosamine-13C-2;478518-89-7;N-Acetyl-D-glucosamine-18O;N-Acetyl-D-glucosamine-13C-3;478529-39-4;N-Acetyl-D-glucosamine-15N;478518-85-3;N-Acetyl-D-glucosamine-13C,15N;478529-40-7;N-Acetyl-D-glucosamine-13C3,15N;478529-43-0 |
| PubChem CID | 1738118 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 595.4±50.0 °C at 760 mmHg |
| Melting Point | 201-204ºC |
| Flash Point | 313.9±30.1 °C |
| Vapour Pressure | 0.0±3.8 mmHg at 25°C |
| Index of Refraction | 1.576 |
| LogP | -2.48 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 15 |
| Complexity | 221 |
| Defined Atom Stereocenter Count | 4 |
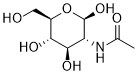
| SMILES | CC(=O)N[C@@H](C=O)[C@H]([C@@H]([C@@H](CO)O)O)O |
| InChi Key | OVRNDRQMDRJTHS-FMDGEEDCSA-N |
| InChi Code | InChI=1S/C8H15NO6/c1-3(11)9-5-7(13)6(12)4(2-10)15-8(5)14/h4-8,10,12-14H,2H2,1H3,(H,9,11)/t4-,5-,6-,7-,8-/m1/s1 |
| Chemical Name | N-Acetyl-beta-D-glucosamine |
| Synonyms | Acetylglucosamine GlcNAc Bio-NAG N-Acetylglucosamine N-Acetyl-D-glucosamineNSC 400525 NSC 524344GreenNAG Marine Sweet |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Approximately 90% of orally administered glucosamine (salt form) gets absorbed from the small intestine. Metabolism / Metabolites A significant fraction of ingested glucosamine is catabolized by first-pass metabolism in the liver. |
| References |
[1]. O-GlcNAc cycling: how a single sugar post-translational modification is changing the way we think about signaling networks. J Cell Biochem. 2006 Jan 1;97(1):71-83. |
| Additional Infomation |
Aldehydo-N-acetyl-D-glucosamine is the open-chain form of N-acetyl-D-glucosamine. It has a role as a human metabolite. The N-acetyl derivative of glucosamine. N-Acetylglucosamine is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). D-Glucose, 2-(acetylamino)-2-deoxy- has been reported in Arabidopsis thaliana, Homo sapiens, and other organisms with data available. The N-acetyl derivative of glucosamine. See also: Poliglusam (monomer of); Adomiparin (monomer of); Adomiparin Sodium (monomer of) ... View More ... Drug Indication For the treatment and prevention of osteoarthritis, by itself or in combination with chondroitin sulfate. Mechanism of Action The mechanism of action in relieving arthritic pain and in repair of cartilage is a matter of speculation. Biochemically, glucosamine is involved in glycoprotein metabolism. Glycoproteins, known as proteoglycans, form the ground substance in the extra-cellular matrix of connective tissue. Proteoglycans are polyanionic substances of high-molecular weight and contain many different types of heteropolysaccharide side-chains covalently linked to a polypeptide-chain backbone. These polysaccharides make up to 95% of the proteoglycan structure. In fact, chemically, proteoglycans resemble polysaccharides more than they do proteins. The polysaccharide groups in proteoglycans are called glycosaminoglycans (GAGs). GAGs include hyaluronic acid, chondroitin sulfate, dermatan sulfate, keratan sulfate, heparin and heparan sulfate. All of the GAGs contain derivatives of glucosamine or galactosamine. Glucosamine derivatives are found in hyaluronic acid, keratan sulfate and heparan sulfate. Chondroitin sulfate contains derivatives of galactosamine. The glucosamine-containing glycosaminoglycan hyaluronic acid is vital for the function of articular cartilage. GAG chains are fundamental components of aggrecan found in articular cartilage. Aggrecan confers upon articular cartilage shock-absorbing properties. It does this by providing cartilage with a swelling pressure that is restrained by the tensile forces of collagen fibers. This balance confers upon articular cartilage the deformable resilience vital to its function. In the early stages of degenerative joint disease, aggrecan biosynthesis is increased. However, in later stages, aggrecan synthesis is decreased, leading eventually to the loss of cartilage resiliency and to most of the symptoms that accompany osteoarthritis. During the progression of osteoarthritis, exogenous glucosamine may have a beneficial role. It is known that, in vitro, chondrocytes do synthesize more aggregan when the culture medium is supplemented with glucosamine. N-acetylglucosamine is found to be less effective in these in vitro studies. Glucosamine has also been found to have antioxidant activity and to be beneficial in animal models of experimental arthritis. The counter anion of the glucosamine salt (i.e. chloride or sulfate) is unlikely to play any role in the action or pharmacokinetics of glucosamine. Further, the sulfate in glucosamine sulfate supplements should not be confused with the glucosamine sulfate found in such GAGs as keratan sulfate and heparan sulfate. In the case of the supplement, sulfate is the anion of the salt. In the case of the above GAGs, sulfate is present as an ester. Also, there is no glucosamine sulfate in chondroitin sulfate (source: PDRhealth). |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~125 mg/mL (~565.07 mM) H2O : ~100 mg/mL (~452.06 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3.25 mg/mL (14.69 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 3.25 mg/mL (14.69 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 3.25 mg/mL (14.69 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 50 mg/mL (226.03 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.5206 mL | 22.6030 mL | 45.2059 mL | |
| 5 mM | 0.9041 mL | 4.5206 mL | 9.0412 mL | |
| 10 mM | 0.4521 mL | 2.2603 mL | 4.5206 mL |