Physicochemical Properties
| Molecular Formula | C18H28N2O4 |
| Molecular Weight | 336.43 |
| Exact Mass | 336.204 |
| CAS # | 37517-30-9 |
| Related CAS # | Acebutolol hydrochloride;34381-68-5;Acebutolol-d7;Acebutolol-d5;1189500-68-2 |
| PubChem CID | 1978 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 564.1±50.0 °C at 760 mmHg |
| Melting Point | 119-123ºC |
| Flash Point | 295.0±30.1 °C |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.543 |
| LogP | 1.95 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 24 |
| Complexity | 401 |
| Defined Atom Stereocenter Count | 0 |
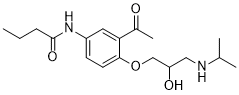
| SMILES | CCCC(NC1=CC(C(C)=O)=C(OCC(O)CNC(C)C)C=C1)=O |
| InChi Key | GOEMGAFJFRBGGG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H28N2O4/c1-5-6-18(23)20-14-7-8-17(16(9-14)13(4)21)24-11-15(22)10-19-12(2)3/h7-9,12,15,19,22H,5-6,10-11H2,1-4H3,(H,20,23) |
| Chemical Name | N-[3-acetyl-4-[2-hydroxy-3-(propan-2-ylamino)propoxy]phenyl]butanamide |
| Synonyms | Acetobutolol Neptal Acebutolol Sectral Dl-Acebutolol Prent |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Acebutolol is a beta-blocker that is used to treat cardiac arrhythmias and high blood pressure. In rats, acebutolol (10 mg/kg) had a plasma clearance rate of 61.9 mL/min/kg, a volume of distribution of 9.6 L/kg, and an elimination half-life of 1.8 hours. In rats, acebutolol (50 mg/kg) had a plasma clearance rate of 46.5 mL/min/kg, a volume of distribution of 9.5 L/kg, and an elimination half-life of 2.3 hours [1]. In Sprague-Dawley rats, acebutolol (30 mg/kg) reduced cardiac output by 65% and 31% after measurements at 1 and 10 minutes, respectively. Acebutolol (30 mg/kg) significantly reduced regional blood flow (RBF) in most organs measured at 1 or 10 minutes compared with baseline values in Sprague-Dawley rats. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Well absorbed from the Gl tract with an absolute bioavailability of approximately 40% for the parent compound. Elimination via renal excretion is approximately 30% to 40% and by non-renal mechanisms 50% to 60%, which includes excretion into the bile and direct passage through the intestinal wall. Metabolism / Metabolites Subject to extensive first-pass hepatic biotransformation (primarily to diacetolol). Biological Half-Life The plasma elimination half-life is approximately 3 to 4 hours. The half-life of its metabolite, diacetolol, is 8 to 13 hours. |
| Toxicity/Toxicokinetics |
Hepatotoxicity Acebutolol is associated with a low rate of mild-to-moderate elevations of serum aminotransferase levels during treatment. These enzyme elevations are usually asymptomatic and transient and resolve even with continuation of therapy. There have been few documented cases of clinically apparent, acute liver injury attributed to acebutolol. The time to onset of injury was typically between 1 and 6 weeks of starting. The pattern of liver enzyme elevations was usually hepatocellular with an acute hepatitis-like presentation, although some cases present with a mixed pattern of enzymes. Fever commonly accompanied the liver injury, but usually without rash and eosinophia. Acebutolol is known to induce autoantibodies such as antinuclear antibody in 10% to 30% of patients, some of whom develop a lupus-like syndrome with fatigue, skin rash and arthralgias. Serum enzyme elevations may accompany this syndrome, but jaundice and symptoms of liver injury are uncommon. The published cases of hepatotoxicity due to acebutolol were relatively mild, self-limited and recovery was rapid upon stopping. Rechallenge resulted in rapid recurrence of injury. Likelihood score: C (probable cause of clinically apparent liver injury). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Because of the relatively extensive excretion of acebutolol and its active metabolite diacetolol into breastmilk and some possible reports of adverse reactions in breastfed infants, other agents are preferred, especially while nursing a newborn or preterm infant. ◉ Effects in Breastfed Infants A study of mothers taking beta-blockers during nursing found a numerically, but not statistically significant increased number of adverse reactions in those taking any beta-blocker. Although the ages of infants were matched to control infants, the ages of the affected infants were not stated. One mother reported no adverse effects in her breastfed infant (age unstated) during acebutolol use. Hypotension, bradycardia, and transient tachypnea occurred in a newborn infant, probably because of acebutolol and diacetolol in breastmilk. The mother was taking 400 mg daily of acebutolol and had renal impairment. Two other neonates in this report who were breastfed had no adverse reactions noted. A mother with essential hypertension had been taking acebutolol for several years. She continued the drug during her first pregnancy and while breastfeeding. Her infant was generally healthy, but seemed to have decreased muscle tone. She stopped the drug, but the infant continued to have stridor and possible sleep apnea. She was also taking acebutolol during her second pregnancy, but her blood pressure was uncontrolled and a cesarean section was performed. The infant did well in the NICU, except for decreased tone including lying with extremities extended, an incomplete Moro response and marked head lag. The infant was not breastfed. It is possible that the late postpartum adverse effects in the first infant were caused by acebutolol and diacetolol in breastmilk, but no measurements of infant plasma drug levels were made. ◉ Effects on Lactation and Breastmilk Relevant published information on the effects of beta-blockade or acebutolol during normal lactation was not found as of the revision date. A study in 6 patients with hyperprolactinemia and galactorrhea found no changes in serum prolactin levels following beta-adrenergic blockade with propranolol. Protein Binding 26% |
| References |
[1]. Piquette-Miller, M. and F. Jamali, Pharmacokinetics and multiple peaking of acebutolol enantiomers in rats. Biopharm Drug Dispos, 1997. 18(6): p. 543-56. [2]. Bristow MR, et al. Treatment of chronic heart failure with β-adrenergic receptor antagonists: a convergence of receptor pharmacology and clinical cardiology. Circ Res. 2011 Oct 28;109(10):1176-94. [3]. Mostafavi, S., R. Lewanczuk, and R. Foster, Influence of acebutolol and metoprolol on cardiac output and regional blood flow in rats. Biopharm Drug Dispos, 2000. 21(4): p. 121-8. |
| Additional Infomation |
Acebutolol is an ether that is the 2-acetyl-4-(butanoylamino)phenyl ether of the primary hydroxy group of 3-(propan-2-ylamino)propane-1,2-diol. It has a role as a beta-adrenergic antagonist, an anti-arrhythmia drug, an antihypertensive agent and a sympathomimetic agent. It is a member of ethanolamines, a propanolamine, a secondary amino compound, an ether, a monocarboxylic acid amide and an aromatic amide. It is a conjugate base of an acebutolol(1+). A cardioselective beta-adrenergic antagonist with little effect on the bronchial receptors. The drug has stabilizing and quinidine-like effects on cardiac rhythm as well as weak inherent sympathomimetic action. Acebutolol is a beta-Adrenergic Blocker. The mechanism of action of acebutolol is as an Adrenergic beta-Antagonist. Acebutolol is a cardioselective beta-blocker used in the treatment of hypertension, angina pectoris and cardiac arrhythmias. Acebutolol has been linked to several instances of clinically apparent drug induced liver injury. Acebutolol is a synthetic butyranilide derivative with hypotensive and antiarrhythmic activity. Acebutolol acts as a cardioselective beta-adrenergic antagonist with little effect on bronchial receptors and has intrinsic sympathomimetic properties. Having stabilizing and quinidine-like effects on cardiac rhythm, Acebutolol is used in ventricular arrhythmias. Other indications include hypertension, alone or in combinations with other agents. A cardioselective beta-1 adrenergic antagonist with little effect on the bronchial receptors. The drug has stabilizing and quinidine-like effects on cardiac rhythm, as well as weak inherent sympathomimetic action. See also: Acebutolol Hydrochloride (has salt form); Diacetolol (has subclass); Secradex (is active moiety of). Drug Indication For the management of hypertension and ventricular premature beats in adults. Mechanism of Action Acebutolol is a selective β1-receptor antagonist. Activation of β1-receptors by epinephrine increases the heart rate and the blood pressure, and the heart consumes more oxygen. Acebutolol blocks these receptors, lowering the heart rate and blood pressure. This drug then has the reverse effect of epinephrine. In addition, beta blockers prevent the release of renin, which is a hormone produced by the kidneys which leads to constriction of blood vessels. Pharmacodynamics Acebutolol is a cardioselective, beta-adrenoreceptor blocking agent, which possesses mild intrinsic sympathomimetic activity (ISA) in its therapeutically effective dose range. In general, beta-blockers reduce the work the heart has to do and allow it to beat more regularly. Acebutolol has less antagonistic effects on peripheral vascular ß2-receptors at rest and after epinephrine stimulation than nonselective beta-antagonists. Low doses of acebutolol produce less evidence of bronchoconstriction than nonselective agents like propranolol but more than atenolol. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9724 mL | 14.8619 mL | 29.7239 mL | |
| 5 mM | 0.5945 mL | 2.9724 mL | 5.9448 mL | |
| 10 mM | 0.2972 mL | 1.4862 mL | 2.9724 mL |