Physicochemical Properties
| Molecular Formula | C32H43N5O11 |
| Molecular Weight | 673.71072 |
| Exact Mass | 673.295 |
| CAS # | 219137-97-0 |
| PubChem CID | 25108630 |
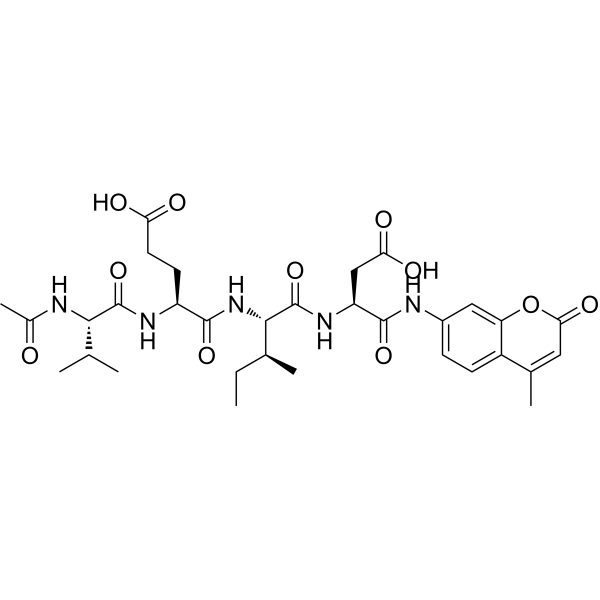
| Sequence | Ac-Val-Glu-Ile-Asp-AMC |
| SequenceShortening | VEID |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 1106.8±65.0 °C at 760 mmHg |
| Flash Point | 623.2±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.571 |
| LogP | 2.36 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 48 |
| Complexity | 1280 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | CC[C@@H]([C@H](NC([C@@H](NC([C@@H](NC(C)=O)C(C)C)=O)CCC(O)=O)=O)C(N[C@H](C(NC1=CC2=C(C(C)=CC(O2)=O)C=C1)=O)CC(O)=O)=O)C |
| InChi Key | QMXIJZDGCJEANV-LFZGUJIPSA-N |
| InChi Code | InChI=1S/C32H43N5O11/c1-7-16(4)28(37-29(44)21(10-11-24(39)40)35-31(46)27(15(2)3)33-18(6)38)32(47)36-22(14-25(41)42)30(45)34-19-8-9-20-17(5)12-26(43)48-23(20)13-19/h8-9,12-13,15-16,21-22,27-28H,7,10-11,14H2,1-6H3,(H,33,38)(H,34,45)(H,35,46)(H,36,47)(H,37,44)(H,39,40)(H,41,42)/t16-,21-,22-,27-,28-/m0/s1 |
| Chemical Name | (4S)-4-[[(2S)-2-acetamido-3-methylbutanoyl]amino]-5-[[(2S,3S)-1-[[(2S)-3-carboxy-1-[(4-methyl-2-oxochromen-7-yl)amino]-1-oxopropan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-5-oxopentanoic acid |
| Synonyms | Ac-VEID-AMC; Ac-Val-Glu-Ile-Asp-AMC; 219137-97-0; (4S)-4-[[(2S)-2-acetamido-3-methylbutanoyl]amino]-5-[[(2S,3S)-1-[[(2S)-3-carboxy-1-[(4-methyl-2-oxochromen-7-yl)amino]-1-oxopropan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-5-oxopentanoic acid; SCHEMBL3319962; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Caspase-6 |
| ln Vitro |
Guidelines (The following protocol is recommended as a general guideline and should be adjusted based on your specific experimental requirements). Enzyme Activity Assay (Example: Rabbit Osteoclasts)[1]: 1. Cell Treatment: o Culture purified, differentiated cells in medium for 48 hours (duration may vary depending on cell type). 2. Cell Lysis: o Transfer treated cells into 4.5 mL lysis buffer. 3. Substrate Incubation: o Divide each lysate into three equal aliquots. o Incubate with 40 μM Ac-VEID-AMC at 37°C for 1 hour. 4. Fluorescence Detection: o Measure the cleavage of fluorescent substrates using: Excitation wavelength (Ex) = 340–360 nm Emission wavelength (Em) = 440–460 nm Caspase-3 is the major effector caspase in osteoclasts following bisphosphonate treatment [1] Caspase-3-like, caspase-6, and caspase-7 activity was determined in lysates from rabbit osteoclast-like cells by the ability to cleave Ac-DEVD-AMC, Ac-VEID-AMC, and MCA-VDQVDGWK-(DNP)-NH2, respectively. The cleavage of Ac-DEVD-AMC was consistently greater in lysates from osteoclast-like cells that had been treated for 48 h with 100 μmol/L CLO or ALN than in lysates from control cultures. However, with the same lysates, there was no such increase in the ability to cleave caspase-6 or caspase-7 substrates (Ac-VEID-AMC or MCA-VDQVDGWK-(DNP)-NH2, respectively) (Figure 5). |
| Cell Assay |
Measurement of caspase activity in osteoclast lysates by proteolytic cleavage of fluorogenic substrates [1] Purified, differentiated rabbit osteoclasts remaining in the Petri dishes after pronase-EDTA digestion were treated with α-MEM/10% FCS containing 100 μmol/L ZOL, 100 μmol/L RIS, 100 μmol/L ALN, 100 μmol/L PAM, 100 μmol/L ETI, 100 μmol/L CLO, or medium alone for 48 h (four Petri dishes per treatment). The osteoclasts were harvested directly into 1.5 mL lysis buffer containing 50 mmol/L Tris (pH 7.4), 1 mmol/L EDTA, 10 mmol/L EGTA (ethylene-glycol-bis[β-aminoethylether]-N,N,N′,N′-tetraacetic acid), 0.5% (w/v) CHAPS (3-[(3-cholamidopropyl)-dimethylammonio]-1-propane sulfonate), and 5 mmol/L cysteine7 and then incubated with 40 μmol/L Ac-DEVD-AMC (a substrate for caspase-3-like proteases) at 37°C for 1 h. The release of aminomethylcoumarin (AMC) was then measured on a Perkin-Elmer fluorimeter using an excitation/emission wavelength of 380 nm/460 nm. The results were corrected for total protein content in the lysates using the bicinchoninic acid assay (Pierce, Rockford, IL), and expressed as the percentage of activity in lysates from control cultures. Larger numbers of osteoclasts were required for the simultaneous detection of caspase-3-like, caspase-6, and caspase-7 activity. Cultures of purified rabbit osteoclast-like cells were treated with 100 μmol/L ALN, 100 μmol/L CLO, or medium alone for 48 h (generally eight Petri dishes per treatment). Cells were then lysed into 4.5 mL of lysis buffer as described earlier, and then each lysate was divided into thirds and incubated with 40 μmol/L Ac-DEVD-AMC, Ac-VEID-AMC, or MCA-VDQVDGWK-(DNP)-NH2 at 37°C for 1 h. Cleavage of the fluorogenic substrates was then assayed as before, using an excitation/emission wavelength of 380 nm/460 nm for Ac-DEVD-AMC and Ac-VEID-AMC, or 325 nm/392 nm for MCA-VDQVDGWK-(DNP)-NH2. |
| References |
[1]. Visualization of bisphosphonate-induced caspase-3 activity in apoptotic osteoclasts in vitro. Bone. 2001 May;28(5):465-73. |
| Additional Infomation | Bisphosphonates inhibit osteoclast-mediated bone resorption by mechanisms that have only recently become clear. Whereas nitrogen-containing bisphosphonates affect osteoclast function by preventing protein prenylation (especially geranylgeranylation), non-nitrogen-containing bisphosphonates have a different molecular mechanism of action. In this study, we demonstrate that nitrogen-containing bisphosphonates (risedronate, alendronate, pamidronate, and zoledronic acid) and non-nitrogen-containing bisphosphonates (clodronate and etidronate) cause apoptosis of rabbit osteoclasts, human osteoclastoma-derived osteoclasts, and human osteoclast-like cells generated in cultures of bone marrow in vitro. Osteoclast apoptosis was shown to involve characteristic morphological changes, loss of mitochondrial membrane potential, and the activation of caspase-3-like proteases capable of cleaving peptide substrates with the sequence DEVD. Caspase-3-like activity could be visualized in unfixed, dying osteoclasts and osteoclast-like cells using a cell-permeable, fluorogenic substrate. Bisphosphonate-induced osteoclast apoptosis was dependent on caspase activation, because apoptosis resulting from alendronate, clodronate, or zoledronic acid treatment was suppressed by zVAD-fmk, a broad-range caspase inhibitor, or by SB-281277, a specific isatin sulfonamide inhibitor of caspase-3/-7. Furthermore, caspase-3 (but not caspase-6 or caspase-7) activity could be detected and quantitated in lysates from purified rabbit osteoclasts, whereas the p17 fragment of active caspase-3 could be detected in human osteoclast-like cells by immunofluorescence staining. Caspase-3, therefore, appears to be the major effector caspase activated in osteoclasts by bisphosphonate treatment. Caspase activation and apoptosis induced by nitrogen-containing bisphosphonates are likely to be the consequence of the loss of geranylgeranylated rather than farnesylated proteins, because the ability to cause apoptosis and caspase activation was mimicked by GGTI-298, a specific inhibitor of protein geranylgeranylation, whereas FTI-277, a specific inhibitor of protein farnesylation, had no effect on apoptosis or caspase activity. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4843 mL | 7.4216 mL | 14.8432 mL | |
| 5 mM | 0.2969 mL | 1.4843 mL | 2.9686 mL | |
| 10 mM | 0.1484 mL | 0.7422 mL | 1.4843 mL |