Physicochemical Properties
| Molecular Formula | C33H38F3N7O11 |
| Molecular Weight | 765.69000 |
| Exact Mass | 765.258 |
| CAS # | 210345-03-2 |
| PubChem CID | 44134612 |
| SequenceShortening | LEHD |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 1211.9±65.0 °C at 760 mmHg |
| Flash Point | 686.7±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.587 |
| LogP | 1.45 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 54 |
| Complexity | 1480 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | HULKIXRFKRCRHD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C33H38F3N7O11/c1-15(2)8-22(39-16(3)44)31(52)41-21(6-7-26(45)46)29(50)42-23(9-18-13-37-14-38-18)32(53)43-24(12-27(47)48)30(51)40-17-4-5-19-20(33(34,35)36)11-28(49)54-25(19)10-17/h4-5,10-11,13-15,21-24H,6-9,12H2,1-3H3,(H,37,38)(H,39,44)(H,40,51)(H,41,52)(H,42,50)(H,43,53)(H,45,46)(H,47,48) |
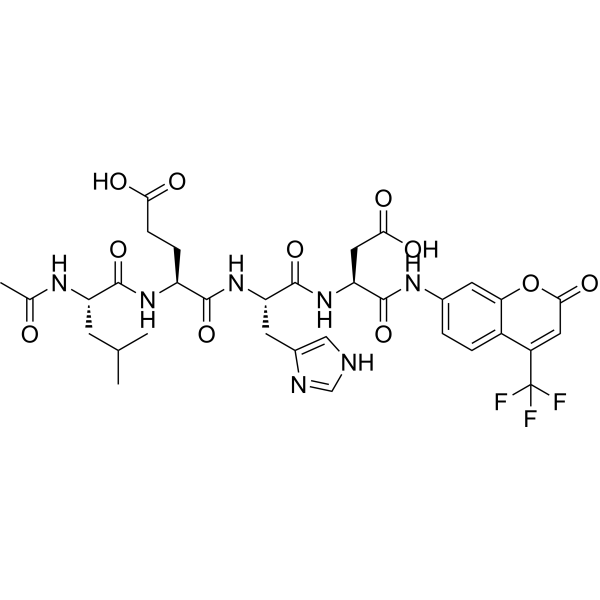
| Chemical Name | 4-[(2-acetamido-4-methylpentanoyl)amino]-5-[[1-[[3-carboxy-1-oxo-1-[[2-oxo-4-(trifluoromethyl)chromen-7-yl]amino]propan-2-yl]amino]-3-(1H-imidazol-5-yl)-1-oxopropan-2-yl]amino]-5-oxopentanoic acid |
| Synonyms | Ac-LEHD-AFC; 210345-03-2; (4S)-4-[[(2S)-2-acetamido-4-methylpentanoyl]amino]-5-[[(2S)-1-[[(2S)-3-carboxy-1-oxo-1-[[2-oxo-4-(trifluoromethyl)chromen-7-yl]amino]propan-2-yl]amino]-3-(1H-imidazol-5-yl)-1-oxopropan-2-yl]amino]-5-oxopentanoic acid; LEHD; Ac-LEHD-AFC? (trifluoroacetate salt); MFCD01862610; SCHEMBL4468806; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Caspase-9 |
| ln Vitro |
Protocol Guidelines (The following protocol serves as a recommended procedure. This is a general guideline that should be adapted based on your specific experimental requirements). Caspase Activity Assessment via Fluorogenic Substrate Assay[1]: 1. Preparation of Cell Lysis Buffer: o 1% Nonidet P-40 o 200 mM NaCl o 20 mM Tris/HCl (pH 7.4) o 10 mg/L Leupeptin o 0.27 kU/L Aprotinin o 100 mM PMSF 2. Incubation Procedure: o Use 25 mg of total protein in lysis buffer o Incubate with 50 mmol/L of: AC-DEVD-AMC (HY-P1003) for caspase-3 AC-VEID-AFC for caspase-8 AC-LEHD-AFC for caspase-9 o Reaction buffer composition: 10 mM HEPES (pH 7.4) 220 mM Mannitol 68 mM Sucrose 2 mM NaCl 2.5 mM KH2PO4 0.5 mM EGTA 2 mM MgCl2 5 mM Pyruvate 0.1 mM PMSF 1 mM Dithiothreitol 3. Activity Measurement: o Monitor fluorescent 7-amino-4-methylcoumarin release o Take measurements at 2-minute intervals for 1 hour o Use a spectrofluorometer for detection |
| Cell Assay |
The assessment of the caspase activity by fluorogenic substrate assay [1] Cell lysates were prepared by lysing the cells in a buffer containing 1 % Nonidet P-40, NaCl 200 mmol/L, Tris/HCl 20 mmol/L, pH 7.4, leupeptin 10 mg/L, aprotinin (trypsin inhibitor 0.27 kU/L) and 100 mm PMSF. Caspase protease activity was determined by incubating the lysate (25 mg of total protein) with 50 mmol/L of fluorogenic substrate, AC-DEVD-AMC (caspase-3), AC-VEID-AFC (caspase-8) or AC-LEHD-AFC (caspase-9) in the buffer (HEPES 10 mmol/L, pH 7.4, containing mannitol 220 mmol/L, sucrose 68 mmol/L, NaCl 2 mmol/L, KH2PO4 2.5 mmol/L, EGTA 0.5 mmol/L , MgCl2 2 mmol/L, pyruvate 5 mmol/L, PMSF 0.1 mmol/L, and dithiothreitol 1 mmol/L). The caspase activity was assessed by measuring fluorescent 7-amino-4-methylcoumarin released for 1 h at a 2-min intervals by a spectrofluorometer. |
| References |
[1]. Je-chun-jun induced apoptosis of human cervical carcinoma HeLa cells. Acta Pharmacol Sin. 2004 Oct;25(10):1372-9. |
| Additional Infomation |
Aim: To study the mechanism of je-chun-jun (JCJ)-inducing the apoptosis of the human cervical carcinoma, HeLa cells.
Methods: The cell viability was assessed using MTT assay. The optical density was measured at 570 nm. The caspase activity was measured using 50 mmol/L of fluorogenic substrate, AC-DEVD-AMC (caspase-3), AC-VEID-AMC (caspase-8) or AC-LEHD-AFC (caspase-9). To confirm the expression of proteins, Western blotting was performed. To detect the characteristic of apoptosis chromatin condensation, HeLa cells were stained with Hoechst 33258 in the presence of JCJ. For the cell cycle analysis, HeLa cells were incubated with Propidium iodide (PI) solution. Fluorescence intensity of cell cycle was measured using flow cytometry system. Results: The loss of viability occurred following the exposure of 10 g/L JCJ. Cells treated with 10 g/L JCJ for 3 d exhibited the apoptotic morphology (brightly blue-fluorescent condensed nuclei by Hoechst 33258-staining) and the reduction of cell volume. Cells incubated with JCJ for 48 h were arrested at the G1 phase of cell cycle and their G1 checkpoint related gene products such as cyclin D1 were transiently decreased. We showed that JCJ induced the p38 MAPK activation in HeLa cells. The p38 MAPK inhibitor, SB203580 protected Hela cells from the JCJ-induced death as well as intervened the JCJ-induced accumulation of cells at the G1 phase. In contrast, MEK1 (-ERK upstream) inhibitor, PD98059 had no effect on HeLa cells. Conclusion: JCJ induced cell cycle arrest and apoptosis of HeLa cells through p38 MAPK pathway. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3060 mL | 6.5301 mL | 13.0601 mL | |
| 5 mM | 0.2612 mL | 1.3060 mL | 2.6120 mL | |
| 10 mM | 0.1306 mL | 0.6530 mL | 1.3060 mL |