Physicochemical Properties
| Molecular Formula | C31H41N5O12 |
| Molecular Weight | 675.683548688889 |
| Exact Mass | 675.275 |
| CAS # | 348079-17-4 |
| PubChem CID | 24799009 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 1157.6±65.0 °C at 760 mmHg |
| Flash Point | 653.9±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.585 |
| LogP | 1.24 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 48 |
| Complexity | 1290 |
| Defined Atom Stereocenter Count | 6 |
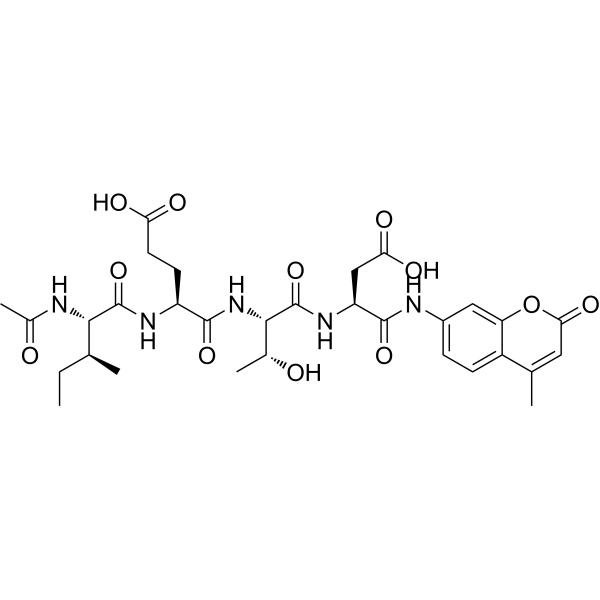
| SMILES | CC1=CC(=O)OC2=CC(NC(=O)[C@H](CC(=O)O)NC(=O)[C@H]([C@H](O)C)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@@H](NC(=O)C)[C@@H](C)CC)=CC=C12 |
| InChi Key | ZYTCBVQZQSFKHQ-HGZCCCDHSA-N |
| InChi Code | InChI=1S/C31H41N5O12/c1-6-14(2)26(32-17(5)38)30(46)34-20(9-10-23(39)40)28(44)36-27(16(4)37)31(47)35-21(13-24(41)42)29(45)33-18-7-8-19-15(3)11-25(43)48-22(19)12-18/h7-8,11-12,14,16,20-21,26-27,37H,6,9-10,13H2,1-5H3,(H,32,38)(H,33,45)(H,34,46)(H,35,47)(H,36,44)(H,39,40)(H,41,42)/t14-,16+,20-,21-,26-,27-/m0/s1 |
| Chemical Name | (4S)-4-[[(2S,3S)-2-acetamido-3-methylpentanoyl]amino]-5-[[(2S,3R)-1-[[(2S)-3-carboxy-1-[(4-methyl-2-oxochromen-7-yl)amino]-1-oxopropan-2-yl]amino]-3-hydroxy-1-oxobutan-2-yl]amino]-5-oxopentanoic acid |
| Synonyms | Ac-IETD-AMC; Ac-Ile-Glu-Thr-Asp-AMC; 348079-17-4; (4S)-4-[[(2S,3S)-2-acetamido-3-methylpentanoyl]amino]-5-[[(2S,3R)-1-[[(2S)-3-carboxy-1-[(4-methyl-2-oxochromen-7-yl)amino]-1-oxopropan-2-yl]amino]-3-hydroxy-1-oxobutan-2-yl]amino]-5-oxopentanoic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Fluorogenic agent for caspase-8/granzyme B |
| ln Vitro | Expression of the cyclin dependent kinase inhibitor p27(KIP1) is intimately linked to the control of proliferation, and is itself regulated by transcription, translation, phosphorylation, protein stability or sequestration. p27(KIP1) is also regulated during apoptosis; cleavage occurs at DPSD(139)S and ESQD(108)V, by a sub-set of Z-VAD-fmk-sensitive caspases. We have identified a related but distinct mechanism that regulates p27(KIP1) in proliferating lymphoid cell lines. In a B-lymphoid cell line (BJAB), the abundance of p27(KIP1) oscillates inversely to proliferation; loss of full-length p27(KIP1) correlates with the appearance of a truncated version corresponding to cleavage at DPSD(139)S. A direct correlation exists between the appearance of truncated p27(KIP1) and the presence of an activity able to cleave peptides representing DPSD(139)S and a caspase-8 substrate (Ac-IETD-AMC) in vitro. This activity is inhibited by Ac-IETD-CHO but not Z-VAD-fmk in vitro. Furthermore a requirement for caspase-8 has been excluded. The activity differs from the apoptosis related p27(KIP1)-cleaving activity; indeed few cells undergoing apoptosis are present in the population of proliferating cells. The activity is further distinguished by its inability to cleave a peptide based on ESQD(108)V in vitro, together with the lack of a corresponding cleavage product in vivo. Inhibition of the caspase activity in vivo promotes an accumulation of full length p27(KIP1), as well as a decrease in cell proliferation. Together these studies highlight the importance of non-apoptotic caspases in regulating p27(KIP1) in transformed lymphoid cells [1]. |
| Cell Assay |
Caspase assays [1] Extract preparation and fluorogenic caspase assays were carried out based on the method supplied with the caspase-3 assay kit although buffer recipes were modified to include inhibitors of serine-, aspartic- and metallo-proteases as indicated. Briefly, cells were washed with PBS and lysed (1×107 cells/ml) in cold lysis buffer (130 mM NaCl, 1% Triton X-100, 10 mM NaPPi, 10 mM Tris-HCl, 1 mM EDTA, 1 mM PMSF, 0.25 μg/ml pepstatin A, 10 mM NaH2PO4/Na2HPO4, pH 7.5). Debris was removed from the resultant extracts by centrifugation. Caspase assays were carried out in 96 well plates by mixing 50 μl of extract with 200 μl reaction buffer (10% glycerol, 2 mM DTT, 1 mM EDTA, 1 mM PMSF, 0.25 μg/ml pepstatin A, 200 mM HEPES, pH 7.5 and 25 μg/ml tetrapeptide-AMC substrate). The caspase inhibitors Ac-IETD-CHO (2 μg/ml) and Z-VAD-fmk (3 μM) were also added where indicated. Reactions were allowed to proceed for between 1 and 2 h at 37°C. Measurement of AMC liberated from the tetrapeptide-AMC substrates was carried out using a SpectraMax Gemini spectrofluorometer with an excitation wavelength of 380 nm and an emission wavelength of 440 nm. Background measurements generated from the reaction completed in the absence of substrate were assessed and subtracted from experimental values. |
| References |
[1]. Exploitation of a non-apoptotic caspase to regulate the abundance of the cdkI p27(KIP1) in transformed lymphoid cells. Oncogene. 2001 May 17;20(22):2737-48. |
| Additional Infomation |
BJAB cells contain caspase or caspase-like activities that are inversely regulated with respect to p27KIP1 abundance [1] In order to discover whether or not BJAB cells contained a caspase-like activity that could be responsible for the different extent of p27KIP1-cleavage seen in proliferating and arrested cells, we carried out in vitro cleavage assays on a panel of fluorogenic caspase substrates (Ac-YVAD-AMC, Ac-IETD-AMC, Ac-LEHD-AMC). Proliferating BJAB cells contained an activity that was capable of inducing a 10-fold increase in the cleavage of the tetrapeptide substrate Ac-IETD-AMC relative to a control lacking cell extract (Figure 4a). Furthermore, activity was reduced almost to control levels in extracts from arrested cells. Ac-IETD-AMC is frequently used to measure caspase-8 activity, suggesting that the activity present in BJAB cells is caspase-8-like in terms of substrate specificity. However, it is known that the specificity of caspase family members for tetrapeptide substrates overlaps (Talanian et al., 1997; Thornberry et al., 1997), and indeed the caspase-1 substrate (Ac-YVAD-AMC) also shows a high degree of cleavage in the presence of extract from proliferating cells. Again, the cleaving activity was greatly reduced in extracts from arrested cells. The caspase-9 substrate (Ac-LEHD-AMC) showed only a small amount of cleavage in the presence of growing BJAB extract. At present it is not clear whether these cleavage events reflect the presence of multiple caspase activities in growing BJAB cells, or whether a single caspase is responsible that has a low specificity for Ac-LEHD-AMC, and a much higher specificity for Ac-YVAD-AMC and Ac-IETD-AMC. In all cases proliferating cells contained more tetrapeptide cleaving activity than their arrested counterparts. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4800 mL | 7.4000 mL | 14.7999 mL | |
| 5 mM | 0.2960 mL | 1.4800 mL | 2.9600 mL | |
| 10 mM | 0.1480 mL | 0.7400 mL | 1.4800 mL |