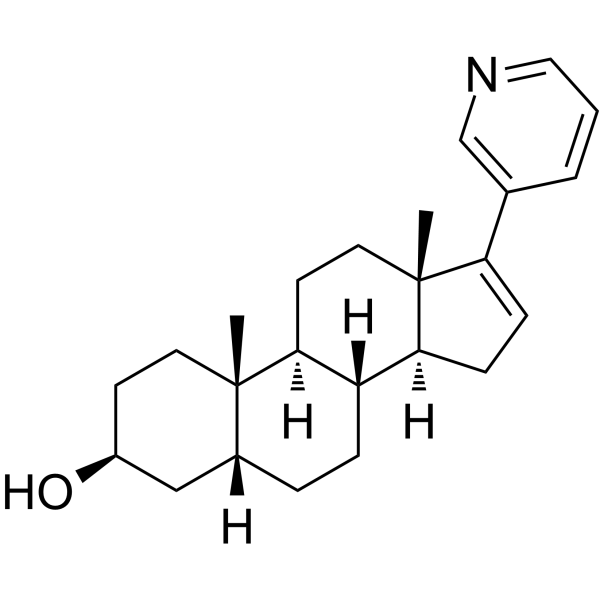
Abiraterone metabolite 1 is the 5β-reduced form/metabolite of abiraterone (CB7598; Zytiga), which is an approved anticancer drug acting as an irreversible and selective CYP17 inhibitor.
Physicochemical Properties
| Molecular Formula | C24H33NO |
| Molecular Weight | 351.524926900864 |
| Exact Mass | 351.256 |
| CAS # | 1940176-03-3 |
| PubChem CID | 122638979 |
| Appearance | White to off-white solid powder |
| LogP | 5.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 26 |
| Complexity | 585 |
| Defined Atom Stereocenter Count | 7 |
| SMILES | C[C@@]12C(C3C=NC=CC=3)=CC[C@H]1[C@@H]1CC[C@@H]3C[C@H](CC[C@]3(C)[C@H]1CC2)O |
| InChi Key | UNJQRCXVHBZVTM-JSIIKIRASA-N |
| InChi Code | InChI=1S/C24H33NO/c1-23-11-9-18(26)14-17(23)5-6-19-21-8-7-20(16-4-3-13-25-15-16)24(21,2)12-10-22(19)23/h3-4,7,13,15,17-19,21-22,26H,5-6,8-12,14H2,1-2H3/t17-,18+,19+,21+,22+,23+,24-/m1/s1 |
| Chemical Name | (3S,5R,8R,9S,10S,13S,14S)-10,13-dimethyl-17-pyridin-3-yl-2,3,4,5,6,7,8,9,11,12,14,15-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol |
| Synonyms | Abiraterone metabolite 1; 1940176-03-3; (3S,5R,8R,9S,10S,13S,14S)-10,13-dimethyl-17-pyridin-3-yl-2,3,4,5,6,7,8,9,11,12,14,15-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol; (3beta,5beta)-17-(3-Pyridinyl)androst-16-en-3-ol; SCHEMBL18170331; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CYP17 |
| ln Vitro | Abiraterone blocks androgen synthesis and prolongs survival in patients with castration-resistant prostate cancer, which is otherwise driven by intratumoral androgen synthesis. Abiraterone is metabolized in patients to Δ(4)-abiraterone (D4A), which has even greater anti-tumour activity and is structurally similar to endogenous steroidal 5α-reductase substrates, such as testosterone. Here, we show that D4A is converted to at least three 5α-reduced and three 5β-reduced metabolites in human serum. The initial 5α-reduced metabolite, 3-keto-5α-abiraterone, is present at higher concentrations than D4A in patients with prostate cancer taking abiraterone, and is an androgen receptor agonist, which promotes prostate cancer progression. In a clinical trial of abiraterone alone, followed by abiraterone plus dutasteride (a 5α-reductase inhibitor), 3-keto-5α-abiraterone and downstream metabolites were depleted by the addition of dutasteride, while D4A concentrations rose, showing that dutasteride effectively blocks production of a tumour-promoting metabolite and permits D4A accumulation. Furthermore, dutasteride did not deplete the three 5β-reduced metabolites, which were also clinically detectable, demonstrating the specific biochemical effects of pharmacological 5α-reductase inhibition on abiraterone metabolism. Our findings suggest a previously unappreciated and biochemically specific method of clinically fine-tuning abiraterone metabolism to optimize therapy.[1] |
| References |
[1]. Redirecting abiraterone metabolism to fine-tune prostate cancer anti-androgen therapy. Nature. 2016 May 26;533(7604):547-51. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 25 mg/mL (~71.12 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (7.11 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.11 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.11 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8448 mL | 14.2239 mL | 28.4479 mL | |
| 5 mM | 0.5690 mL | 2.8448 mL | 5.6896 mL | |
| 10 mM | 0.2845 mL | 1.4224 mL | 2.8448 mL |