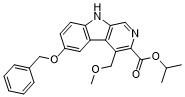
Abecarnil (ZK112119; ZK-112119) is a positive allosteric modulator of GABAA receptor, acting as a ligand or a partial agonist for benzodiazepine (BZ) receptor with anxiolytic and anticonvulsant effects.
Physicochemical Properties
| Molecular Formula | C24H24N2O4 |
| Molecular Weight | 404.46 |
| Exact Mass | 404.174 |
| Elemental Analysis | C, 71.27; H, 5.98; N, 6.93; O, 15.82 |
| CAS # | 111841-85-1 |
| PubChem CID | 65914 |
| Appearance | Solid powder |
| Density | 1.246g/cm3 |
| Boiling Point | 620.2ºC at 760mmHg |
| Flash Point | 328.9ºC |
| Vapour Pressure | 2.62E-15mmHg at 25°C |
| Index of Refraction | 1.647 |
| LogP | 5.006 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 30 |
| Complexity | 565 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C([H])([H])C1C([H])=C([H])C([H])=C([H])C=1[H])C1C([H])=C([H])C2=C(C=1[H])C1=C(C([H])([H])OC([H])([H])[H])C(C(=O)OC([H])(C([H])([H])[H])C([H])([H])[H])=NC([H])=C1N2[H] |
| InChi Key | RLFKILXOLJVUNF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H24N2O4/c1-15(2)30-24(27)23-19(14-28-3)22-18-11-17(29-13-16-7-5-4-6-8-16)9-10-20(18)26-21(22)12-25-23/h4-12,15,26H,13-14H2,1-3H3 |
| Chemical Name | 6-(Benzyloxy)-4-(methoxymethyl)-9H-pyrido[3,4-b]indole-3-carboxylic acid 1-methylethyl ester |
| Synonyms | ZK-112119Abecarnil ZK 112119 ZK112119 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Rat cortical membranes are better able to bind t-[35S]butylbicyclic phosphorothioate when bevacarnal is present [1]. Compared to diazepam (DZP), abelanil has a 3–6 times greater affinity for forebrain BZ receptors [1]. |
| ln Vivo | Abecarnil (0.3 mg/kg, IP, once) antagonizes the rise in neuroactive hormones caused by foot shock [2]. Abecarnil (0-2.5 mg/kg, IP, once) dose-dependently decreases epileptic activity [3]. Abecarnil is efficacious against sound-induced convulsions in DBA/2 mice, air blast-induced generalized seizures in gerbils, and myoclonus in baboon baboons [4]. Abecarnil is 2-10 times more powerful than DZP in most tests of anxiolytic activity in rodents and in lowering locomotor activity in mice and rats completely accustomed to the testing room [1]. |
| Animal Protocol |
Animal/Disease Models: Male SD (SD (Sprague-Dawley)) CD rats (200-250 g) [2] Doses: 0.3 mg/kg Route of Administration: IP, once, 30 minutes before sacrifice Experimental Results: failed to change basal pregnenolone and Progesterone only slightly diminished elevated THDOC levels but antagonized the foot-shock-induced increase in neuroactive steroids. Animal/Disease Models: WAG/Rij rats (male and female, 190-380 g each, 13-19 weeks old, 8 rats per group) [3] Doses: 0, 0.16, 0.4, 1.0, 2.5 mg/kg; 1 mL/400 g Route of Administration: IP, once Experimental Results: diminished duration of spike discharges and increased immobility behavior. Dose-dependent reduction in epileptic activity, whether measured as number, mean duration, or total duration of spike discharges. The ED50 for reducing the number of sharp wave discharges in the second hour is 0.4 mg/kg. |
| References |
[1]. Abecarnil, a metabolically stable, anxioselective beta-carboline acting at benzodiazepine receptors. J Pharmacol Exp Ther. 1990 Apr;253(1):334-43. [2]. Stress-induced increase in brain neuroactive steroids: antagonism by abecarnil. Pharmacol Biochem Behav. 1996 May;54(1):205-10. [3]. Effects of the beta-carboline abecarnil on epileptic activity, EEG, sleep and behavior of rats. Pharmacol Biochem Behav. 1992 Jul;42(3):401-5. [4]. Anticonvulsant action of the beta-carboline abecarnil: studies in rodents and baboon, Papio papio. J Pharmacol Exp Ther. 1990 Apr;253(1):344-52. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4724 mL | 12.3622 mL | 24.7243 mL | |
| 5 mM | 0.4945 mL | 2.4724 mL | 4.9449 mL | |
| 10 mM | 0.2472 mL | 1.2362 mL | 2.4724 mL |