AZD2858 is a novel, potent, orally bioactive, and selective GSK-3 (glycogen synthase kinase-3) inhibitor with the potential to be used in fracture healing. It inhibits GSK-3 with an IC50 of 68 nM. It can increase bone mass in rats and activate the Wnt signaling. After a two-week treatment period, treatment rats given AZD2858 orally experienced a dose-dependent increase in trabecular bone mass. Cortical sites also noticed the notable effect. In biomechanical testing, the diaphyseal strength of femora and the vertebral compression strength both increased.
Physicochemical Properties
| Molecular Formula | C21H23N7O3S |
| Molecular Weight | 453.5174 |
| Exact Mass | 453.158 |
| Elemental Analysis | C, 55.62; H, 5.11; N, 21.62; O, 10.58; S, 7.07 |
| CAS # | 486424-20-8 |
| Related CAS # | 2108876-05-5 (HCl);486424-21-9 (xHCl);486424-20-8; |
| PubChem CID | 10138980 |
| Appearance | Green to khaki solid powder |
| Density | 1.4±0.1 g/cm3 |
| Index of Refraction | 1.663 |
| LogP | 2.97 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 32 |
| Complexity | 730 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | S(C1C([H])=C([H])C(C2=C([H])N=C(C(C(N([H])C3=C([H])N=C([H])C([H])=C3[H])=O)=N2)N([H])[H])=C([H])C=1[H])(N1C([H])([H])C([H])([H])N(C([H])([H])[H])C([H])([H])C1([H])[H])(=O)=O |
| InChi Key | FHCSBLWRGCOVPT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H23N7O3S/c1-27-9-11-28(12-10-27)32(30,31)17-6-4-15(5-7-17)18-14-24-20(22)19(26-18)21(29)25-16-3-2-8-23-13-16/h2-8,13-14H,9-12H2,1H3,(H2,22,24)(H,25,29) |
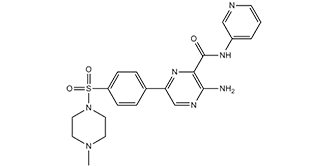
| Chemical Name | 3-amino-6-[4-[(4-methyl-1-piperazinyl)sulfonyl]phenyl]-N-3-pyridinyl-2-pyrazinecarboxamid |
| Synonyms | AZD-2858; AZD2858; AZD 2858 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
GSK-3α (IC50 = 0.9 nM); GSK-3β (IC50 = 5 nM); CDK5/p25 (IC50 = 356 nM); Haspin (IC50 = 366 nM); CDK5/p35 (IC50 = 387 nM); DYRK2 (IC50 = 491 nM); CDK2/cyclin A (IC50 = 810 nM); CDK1/cyclin B (IC50 = 1246 nM); PIM3 (IC50 = 1269 nM); TLK2 (IC50 = 1381 nM); PKD2 (IC50 = 2462 nM); CDK2/cyclin E (IC50 = 3310 nM); Aurora-A (IC50 = 4966 nM) Glycogen Synthase Kinase 3 (GSK3), including GSK3α and GSK3β isoforms. For GSK3β, the IC₅₀ value was ~10 nM; for GSK3α, the IC₅₀ value was ~20 nM. No inhibitory activity against other kinases (e.g., CDK2, ERK1, JNK2) was detected at concentrations up to 1 μM [2] |
| ln Vitro |
AZD2858 is a selective GSK-3 inhibitor with an IC50 of 68 nM, inhibits tau phosphorylation at the S396 site, activates Wnt signaling pathway. A 3-fold increase in -β-cateninlevels is seen in primary isolated human osteoblast-like cells after AZD2858 treatment (1 μM, 12 h). [1] In vitro, AZD2858 stimulates hADSC commitment to osteoblasts and osteogenic mineralization while causing -β-cateninto stabilize in human and rat mesenchymal stem cells. [2] 1. In primary rat bone marrow mesenchymal stem cells (BMSCs) induced for osteogenesis, treatment with AZD2858 (10 nM, 30 nM, 100 nM for 14 days) dose-dependently increased alkaline phosphatase (ALP) activity: at 100 nM, ALP activity was ~2.8-fold higher than the osteogenic induction control (measured via p-nitrophenyl phosphate colorimetry). It also upregulated the secretion of osteocalcin (OCN, a mature osteoblast marker) by ~3.2-fold at 100 nM (ELISA) and enhanced the expression of osteogenic genes (Runx2, ALP, OCN) by qPCR: Runx2 mRNA was ~2.5-fold higher, ALP mRNA ~3.0-fold higher, and OCN mRNA ~2.7-fold higher at 100 nM [2] 2. In primary rat chondrocytes cultured in chondrogenic medium, AZD2858 (10 nM-100 nM for 7 days) dose-dependently inhibited chondrogenic differentiation: the expression of type II collagen (a chondrocyte marker) was reduced by ~60% at 100 nM (Western blot), and the number of Alcian blue-positive chondrocyte clusters decreased by ~55% at 100 nM (Alcian blue staining) [2] |
| ln Vivo |
After two weeks of treatment, oral AZD2858 treatment in rats results in a dose-dependent increase in trabecular bone mass compared to control, with the maximum effect occurring at a dose of 20 mg/kg once daily (total BMC: 172% of control). Cortical sites exhibit a minor but noteworthy effect (total BMC: 111% of control).[1] AZD285 treatment (30 μmol/kg) on rats daily for up to 3 weeks shows an increase in both mineral density (of 28% at 2 weeks and 38% at 3 weeks) and mineral content (of 81% at 2 weeks and 93% at 3 weeks) in the calluses. AZD285 treatment makes the fractures heals more rapidly, with a bony callus without an obvious endochondral component.[3] Over the course of a 28-day exposure period, rats exposed to AZD2858 exhibit time-dependent changes in serum biomarkers of bone turnover and an increase in bone mass. Following 7 days, AZD2858 increases the bone formation biomarker P1NP and decreases the bone resorption biomarker TRAcP-5b in rats, indicating increased bone anabolism and decreased resorption.[2] 1. In male Sprague-Dawley rats (250-300 g) with tibial transverse fractures (stabilized with intramedullary pins), oral administration of AZD2858 (30 mg/kg, once daily for 21 days) accelerated fracture healing. Micro-CT analysis showed that the bone volume/total volume (BV/TV) of the fracture callus was ~2.3-fold higher than the vehicle group, and the bone mineral density (BMD) of the callus was ~1.8-fold higher. Histological staining (H&E, Masson’s trichrome) revealed no endochondral bone formation in the AZD2858-treated group (in contrast to the vehicle group, which showed obvious cartilage tissue in the callus) [2] 2. Biomechanical testing of the fractured tibias (day 21) showed that AZD2858 treatment increased the ultimate load (by ~45%) and stiffness (by ~38%) compared to the vehicle group, indicating improved mechanical strength of the healed bone [2] 3. Immunohistochemical staining of fracture callus tissue (day 14) showed that AZD2858 increased the number of osteocalcin-positive osteoblasts by ~2.6-fold and reduced the number of type II collagen-positive chondrocytes by ~70% vs. vehicle, confirming enhanced osteogenesis and suppressed chondrogenesis in vivo [2] |
| Enzyme Assay |
In vitro functional activity of AZD2858 is evaluated using NIH-3T3 cells that express 4-repeat Tau. In 6-well plates, the cells are plated at a density of 6 105 cells/well after being grown in DMEM media with 2 mM L-glut and 10% HiFCS. At concentrations of 1, 10, 100, 500, 1000, 2000, and 10,000 nM, AZD2858 is dosed in triplicates in each experiment. A 100 L volume of ice-cold lysis buffer containing 0.5% NP-40, 10 mM Tris, pH 7.2, 150 mM NaCl, and 2 mM EDTA is used to treat cells for 4 hours prior to cell lysis. Cocktail Protease Inhibitors, 50 mM NaF, 0.2 mM NaVO4, and other phosphatase and protease inhibitors are added to a suspension to create a reaction. After the solution has been snap frozen at 80 °C for at least one hour, it is thawed on ice, the lysate is clarified using centrifugation, and then a Western blot is performed in accordance with industry standards. After blocking, the blots are exposed overnight to the primary antibody, Phospho-Ser396-tau (1:1000), washed, and then incubated with the secondary antibody (donkey anti-rabbit, 1:5000). Re-probing is done using the secondary horseradish peroxidase linked antibody (sheep anti-mouse, 1:10000) and the primary antibody Tau5 (1:200). The ratio of S396 tau to total tau (tau5) is calculated after all blots have been quantified using densitometric analysis and developed using ECL Western blot detection reagents and Kodak X-ray films. 1. GSK3β kinase activity assay: Recombinant human GSK3β (5 ng) was incubated with a synthetic peptide substrate (sequence: YRRAAVPPSPSLSRHSSPHQpSEDEEE, 50 μM) in reaction buffer containing 20 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 1 mM DTT, and 10 μM [γ-³³P]-ATP. AZD2858 (1 nM-100 nM) was added, and the mixture was incubated at 30°C for 60 minutes. The reaction was terminated by spotting 20 μL of the mixture onto phosphocellulose paper, which was washed 3 times with 1% phosphoric acid to remove unincorporated [γ-³³P]-ATP. Radioactivity was measured via liquid scintillation counting, and the IC₅₀ for GSK3β was calculated from the dose-response curve (percent inhibition vs. log concentration) [2] 2. GSK3α kinase activity assay: The protocol was identical to the GSK3β assay, except recombinant human GSK3α (5 ng) was used. The IC₅₀ for GSK3α was determined using the same concentration range of AZD2858 and data analysis method [2] 3. Kinase selectivity assay: For other kinases (CDK2, ERK1, JNK2), recombinant enzymes (5-10 ng) were incubated with their respective peptide substrates, [γ-³³P]-ATP, and AZD2858 (0.1 nM-1 μM) in kinase-specific reaction buffers. Radioactivity was measured as above, and no inhibitory activity was detected for these kinases [2] |
| Cell Assay |
Rat MSCs (isolated from Sprague Dawley rat bone marrow at less than 8 weeks after gestation) and human adipose derived stem cells (HADSCs) are cultured in DMEM with 5% FBS and 2 mM GlutaMax as the basal medium. Before being treated with AZD2858 (0.3 nM to 20 mM), cells are seeded in 96-well plates with basal media (3–5000 cells/well) for 18 hours. -Catenin stabilization is assessed after 24 hours. 1. Rat BMSC osteogenesis assay: Primary rat BMSCs were isolated from femurs, seeded in 24-well plates at 2×10⁴ cells/well, and cultured in osteogenic medium (DMEM + 10% FBS + 50 μg/mL ascorbic acid + 10 mM β-glycerophosphate). AZD2858 (10 nM-100 nM) was added, and the medium was changed every 3 days. On day 14, cells were lysed for ALP activity detection (using p-nitrophenyl phosphate as substrate, absorbance measured at 405 nm); supernatant was collected for OCN ELISA. For qPCR, total RNA was extracted on day 7, cDNA synthesized, and primers for Runx2, ALP, and OCN were used (GAPDH as the reference gene) [2] 2. Rat chondrocyte chondrogenesis assay: Primary rat chondrocytes were isolated from articular cartilage of 2-week-old rats, seeded in 6-well plates at 5×10⁵ cells/well, and cultured in chondrogenic medium (DMEM + 10% FBS + 10 ng/mL TGF-β1). AZD2858 (10 nM-100 nM) was added, and the medium was changed every 2 days. On day 7, cells were fixed for Alcian blue staining (to count chondrocyte clusters) or lysed for Western blot analysis (primary antibody against type II collagen, β-actin as loading control) [2] |
| Animal Protocol |
Each rat is dosed orally with vehicle or AZD2858 using a plastic gavage tube. The dosage is 10 mL/kg. Deionized water that has been pH-adjusted to 3.50.1 makes up the vehicle. pH 3.50.1 is adjusted for formulations. The doses are 0, 0.2, 2, or 20 mg/kg administered either twice daily (TD), once daily (OD), every other day (O/2D), or every fourth day (O/4D) for a total of 14 days. The protocol yields 13 groups with an average of 8 animals each (104 animals total). Each animal receives a subcutaneous injection of a bicarbonate buffered calcein solution (8 mg/kg, 1 mL/kg)[1] at 7 days following the study's start and once more 2 days before the planned terminal necropsy. 1. Rat tibial fracture model: Male Sprague-Dawley rats (250-300 g) were anesthetized, and a transverse fracture was created in the right tibia using a bone saw, followed by intramedullary pin fixation. Rats were randomly divided into 2 groups (n=10/group): vehicle (0.5% methylcellulose, oral gavage) and AZD2858 (30 mg/kg, suspended in 0.5% methylcellulose, oral gavage). Dosing was performed once daily for 21 days. On days 7, 14, and 21, 3 rats per group were euthanized: the fractured tibias were harvested for Micro-CT analysis (to measure BV/TV and BMD), histological staining (H&E, Masson’s trichrome, immunohistochemistry for OCN and type II collagen), and biomechanical testing (three-point bending test to measure ultimate load and stiffness) [2] |
| ADME/Pharmacokinetics |
1. In male Sprague-Dawley rats, oral administration of AZD2858 (30 mg/kg) showed an oral bioavailability of ~40%. The peak plasma concentration (Cₘₐₓ) was ~250 ng/mL, achieved at ~1 hour (Tₘₐₓ) post-dosing. The elimination half-life (t₁/₂) was ~3.2 hours. Tissue distribution analysis (1 hour post-dosing) showed that the drug accumulated in the bone (femur, ~180 ng/g) and liver (~320 ng/g), with lower concentrations in the kidney (~80 ng/g) and muscle (~60 ng/g) [2] |
| Toxicity/Toxicokinetics |
1. In the 21-day rat fracture study (30 mg/kg, oral), AZD2858 caused no significant changes in body weight (weekly measurement: drug group gained ~15% vs. ~14% in vehicle group) or serum markers of liver (ALT, AST) and kidney function (creatinine, urea nitrogen) compared to the vehicle group [2] 2. Histopathological examination of major organs (liver, kidney, spleen, heart) from AZD2858-treated rats (day 21) showed no abnormal lesions (e.g., inflammation, necrosis) vs. vehicle [2] 3. Plasma protein binding of AZD2858 was ~90% in rat plasma (measured via equilibrium dialysis) [2] |
| References |
[1]. GSK-3 inhibition by an orally active small molecule increases bone mass in rats. Bone. 2012 Mar;50(3):619-27. [2]. Rats treated with AZD2858, a GSK3 inhibitor, heal fractures rapidly without endochondral bone formation. Bone. 2013 May;54(1):126-32. [3]. Human stem cell osteoblastogenesis mediated by novel glycogen synthase kinase 3 inhibitors induces bone formation and a unique bone turnover biomarker profile in rats. Toxicol Appl Pharmacol. 2013 Oct 15;272(2):399-407. [4]. Glycogen synthase kinase-3α/β inhibition promotes in vivo amplification of endogenous mesenchymal progenitors with osteogenic and adipogenic potential and their differentiation to the osteogenic lineage. J Bone Miner Res. 2011 Apr;26(4):811-21. |
| Additional Infomation |
AZD2858 is a member of the class of pyrazines that is pyrazine substituted by (pyridin-3-yl)aminocarbonyl, amino, and 4-(4-methylpiperazine-1-sulfonyl)phenyl groups at positions 2, 3 and 6, respectively. It is a potent inhibitor of GSK3alpha and GSK3beta (IC50 values of 0.9 and 4.9 nM, respectively) and increases bone mass (via Wnt activation) in rats. It has a role as an EC 2.7.11.26 (tau-protein kinase) inhibitor, an antineoplastic agent, a bone density conservation agent and a Wnt signalling activator. It is a member of pyrazines, a secondary carboxamide, a member of pyridines, a N-methylpiperazine, a sulfonamide and an aromatic amine. 1. AZD2858 exerts its osteogenic effects by inhibiting GSK3, which stabilizes β-catenin and activates the Wnt/β-catenin signaling pathway. This activation promotes the differentiation of mesenchymal stem cells into osteoblasts and suppresses their differentiation into chondrocytes, leading to "direct" bone formation without endochondral ossification during fracture healing [2] 2. The ability of AZD2858 to accelerate fracture healing and increase bone mass (without cartilage intermediate) suggests potential therapeutic applications in conditions requiring rapid bone repair (e.g., traumatic fractures) and osteoporosis [2] 3. AZD2858 is an orally active GSK3 inhibitor, which offers advantages over injectable bone-forming agents (e.g., parathyroid hormone) in terms of patient compliance [2] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~7 mg/mL (15.43 mM) Water: <1 mg/mL Ethanol: <1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: 1.25 mg/mL (2.76 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (2.76 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: 30% PEG400+0.5% Tween80+5% propylene glycol: 30 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2050 mL | 11.0249 mL | 22.0497 mL | |
| 5 mM | 0.4410 mL | 2.2050 mL | 4.4099 mL | |
| 10 mM | 0.2205 mL | 1.1025 mL | 2.2050 mL |