Physicochemical Properties
| Molecular Formula | C32H35F3N4O5S |
| Molecular Weight | 644.704317331314 |
| Exact Mass | 644.228 |
| Elemental Analysis | C, 59.62; H, 5.47; F, 8.84; N, 8.69; O, 12.41; S, 4.97 |
| CAS # | 929890-64-2 |
| PubChem CID | 57339445 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 45 |
| Complexity | 1090 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | MAQDQJWCSSCURR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H35F3N4O5S/c1-2-19-45(42,43)39-17-15-38(16-18-39)21-22-3-11-26(12-4-22)36-30(40)24-7-5-23(6-8-24)28-20-27(37-31(41)25-9-10-25)13-14-29(28)44-32(33,34)35/h3-8,11-14,20,25H,2,9-10,15-19,21H2,1H3,(H,36,40)(H,37,41) |
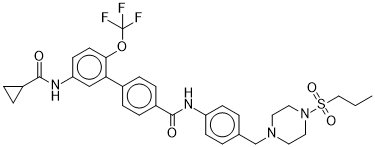
| Chemical Name | 4-[5-(cyclopropanecarbonylamino)-2-(trifluoromethoxy)phenyl]-N-[4-[(4-propylsulfonylpiperazin-1-yl)methyl]phenyl]benzamide |
| Synonyms | AZD-7295; AZD7295; AZD7295; AZD 7295; 102MC1467J; 4-[5-(cyclopropanecarbonylamino)-2-(trifluoromethoxy)phenyl]-N-[4-[(4-propylsulfonylpiperazin-1-yl)methyl]phenyl]benzamide; 5'-[(Cyclopropylcarbonyl)amino]-N-[4-[[4-(propylsulfonyl)-1-piperazinyl]methyl]phenyl]-2'-(trifluoromethoxy)[1,1'-biphenyl]-4-carboxamide; UNII-102MC1467J; AZD 7295 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | HCV NS5A protein |
| ln Vitro |
Similarly, AZD-7295 is potent against GT-1b replicon (EC50 value of 0.007 μM), but it is virtually inactive against GT-1a and GT-3a replicons (EC50 values of 1.24 μM and 1.3 μM, respectively) [35]. In an early clinical study with AZD-7295, a mean 2.1 log10 decline in viral load from baseline (BL) was observed in GT-1b infected patients after five days of dosing (233 mg three times per day (TID)), while no apparent antiviral effect was observed for GT-1a and GT-3 infected patients receiving the same treatment [35]. Since there were no marked differences in PK parameters among patients infected with different genotypes, and since the Ctrough values of AZD-7295 exceeded the GT-1b EC90 by 9.4-fold but did not reach the EC50 values of GT-1a and GT-3a, the HCV decline correlated well with the in vitro replicon potency. Table 1 lists the in vitro potency of selected NS5A inhibitors; the EC50 values are derived from published data using different assays (transient or stable cell lines) and do not represent a direct side-by-side comparison of inhibitors. Assuming that sufficient exposure can be achieved in vivo for each of these inhibitors, it is expected that the in vitro potency will be clinically meaningful.[2] The direct-acting antivirals (DAAs) currently in development for treatment of hepatitis C fall into four categories: (i) NS3/4A protease inhibitors: ABT-450/r, faldaprevir, asunaprevir, GS-9256, vedroprevir (GS-9451), danoprevir, MK-5172, vaniprevir, sovaprevir, ACH-2684, narlaprevir and simeprevir, in addition to those that are already developed [telaprevir (Incivek®) and boceprevir (Victrelis®)], (ii) NS5A protein inhibitors: ABT-267, daclatasvir, ledipasvir, ACH-2928, ACH-3102, PPI-668, AZD-7295, MK-8742, and GSK 2336805; (iii) NS5B (nucleoside-type) polymerase inhibitors: sofosbuvir (now approved by the FDA since 6 December 2013), GS-0938, mericitabine, VX-135, ALS 2158 and TMC 649128; (iv) NS5B (non-nucleoside-type) polymerase inhibitors: VX-222, ABT-072, ABT-333, deleobuvir, tegobuvir, setrobuvir, VCH-916, VCH-759, BMS-791325 and TMC-647055. Future drug combinations will likely exist of two or more DAAs belonging to any of the 4 categories, with the aim to achieve (i) pan-genotypic hepatitis C virus (HCV) activity, (ii) little or no risk for resistance; (iii) short duration (i.e. 12 weeks) of treatment, and (iv) a sustained viral response (SVR) and definite cure of the disease.[1] |
| ln Vivo | Robust HCV RNA declines have been reported for GT-1a, GT-1b, GT-3a and GT-4a infected patients treated with NS5A inhibitors (Table 2). All except AZD-7295 were dosed once daily (QD). One striking characteristic of the anti-HCV effect of NS5A inhibitors in patients is the sharp initial viral decline. For example, a ∼2 log10 decline in viral load was observed four hours after the first 60 mg dose of DCV in a patient infected with GT-1a HCV. Robust HCV RNA declines have also been observed in patients infected with other genotypes and treated with other NS5A inhibitors. The potency of IDX-719 on GT-1a, GT-3a and GT-4a in the replicon system (0.0062–0.024 nM, Table 1) translated to a 3.0–3.6 log10 decline in viral RNA in patients infected with these HCV genotypes in a three-day monotherapy study (Table 2). A similar correlation between replicon potency and clinical effect across genotypes was observed for PPI-668 (Table 1, Table 2)[2]. |
| References |
[1]. Current race in the development of DAAs (direct-acting antivirals) against HCV. Biochem Pharmacol. 2014 Jun 15;89(4):441-52. [2]. Antiviral activity and resistance of HCV NS5A replication complex inhibitors. Curr Opin Virol. 2013 Oct;3(5):514-20. |
| Additional Infomation | AZD7295 has been investigated for the treatment of Hepatitis C. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5511 mL | 7.7555 mL | 15.5111 mL | |
| 5 mM | 0.3102 mL | 1.5511 mL | 3.1022 mL | |
| 10 mM | 0.1551 mL | 0.7756 mL | 1.5511 mL |