AZD1305 is a novel and potent potassium channel and sodium channel antagonist under investigation for the management and reversal of cardiac arrhythmias. It can inhibit rapidly the activating delayed-rectifier potassium current, L-type calcium current, and inward sodium current.
Physicochemical Properties
| Molecular Weight | 434.504348993301 |
| Exact Mass | 434.233 |
| CAS # | 872045-91-5 |
| PubChem CID | 15605092 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.066 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 31 |
| Complexity | 635 |
| Defined Atom Stereocenter Count | 0 |
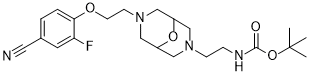
| SMILES | FC1C=C(C#N)C=CC=1OCCN1CC2CN(CCNC(=O)OC(C)(C)C)CC(C1)O2 |
| InChi Key | BLLNYXOLLAVTRF-HDICACEKSA-N |
| InChi Code | InChI=1S/C22H31FN4O4/c1-22(2,3)31-21(28)25-6-7-26-12-17-14-27(15-18(13-26)30-17)8-9-29-20-5-4-16(11-24)10-19(20)23/h4-5,10,17-18H,6-9,12-15H2,1-3H3,(H,25,28)/t17-,18+ |
| Chemical Name | (2-(7-(2-(4-Cyano-2-fluorophenoxy)ethyl)-9-oxa-3,7-diazabicyclo(3.3.1)non-3-yl)ethyl)carbamic acid tert-butyl ester |
| Synonyms | AZD-1305 AZD 1305 AZD1305 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Atrial potential PRR is induced in isolated coronary vascular access by AZD-1305 (1,3 μM) [1]. In isolated coronary vascular access, atrial potential PRR is conducted by AZD-1305 (1,3 μM) [1]. Greater use-induced declines are produced by the maximal rising velocity of the beat (Vmax) [1]. 1-10 μM) significantly reduces Vmax, action potential triggering and onset potential of canine pulmonary vein cuff preparations; the action potential duration increases significantly at 90% repolarization. AZD-1305 (1,3 μM) induces sodium channel-mediated parameters in atrial coronary myocardial conduction compared to the ventricle. AZD-1305 (5 μM) tonicizes and modifies ventricular sodium channels in isolated coronary vascular access. Stronger blockade of ventricular sodium channels [1] AZD-1305 (and greater increase in diastolic excitation threshold [1]. Amiodarone greatly strengthened the action of AZD-1305, and AZD-1305 (1-10 μM) canine pulmonary vein catheter dramatically extended the basal cycle duration of the pulmonary vein cuff preparation while maintaining a 1:1 activator [2]. |
| ln Vivo | AZD-1305 (braid concentration 1.2 μM and 4.5 μM, intravenous infusion) has longer ERP prolongation duration and repolarization time than the ventricles of Beagle dogs [1]. Comparing the short-term duration and excitability of canine atria with Beagle canine ventricles, AZD-1305 (braid concentration 1.2 μM and 4.5 μM, intravenous administration) is more effective [1]. In the Beagle canine model of atrial fibrillation mediated by guinea choline, AZD-1305 (mid-mid 1-3 μM, intravenous infusion) stops persistent atrial fibrillation and inhibits its formation [1]. |
| References |
[1]. AZD1305 Exerts Atrial Predominant Electrophysiological Actions and Is Effective in Suppressing Atrial Fibrillation and Preventing Its Reinduction in the Dog. Cardiovasc Pharmacol. 2010, Volume 56,Number 1. [2]. Electrophysiologic and antiarrhythmic effects of AZD1305 in canine pulmonary vein sleeves. THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS. 2010, Volume 334,Number 1. [3]. AZD1305 exerts atrial predominant electrophysiological actions and is effective in suppressing atrial fibrillation and preventing its reinduction in the dog. J Cardiovasc Pharmacol. 2010 Jul;56(1):80-90. |
| Additional Infomation |
AZD1305 has been used in trials studying the treatment and basic science of Atrial Flutter, Atrial Fibrillation, and Left Ventricle Function. See also: Azd-1305 (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3015 mL | 11.5075 mL | 23.0150 mL | |
| 5 mM | 0.4603 mL | 2.3015 mL | 4.6030 mL | |
| 10 mM | 0.2301 mL | 1.1507 mL | 2.3015 mL |